NCERT Exemplar Class 8 Science Chapter 14 Chemical Effects of Electric Current are part of NCERT Exemplar Class 8 Science. Here we have given NCERT Exemplar Class 8 Science Chapter 14 Chemical Effects of Electric Current.
NCERT Exemplar Class 8 Science Chapter 14 Chemical Effects of Electric Current
Multiple Choice Questions (MCQs)
Question 1
An electric current can produce
(a) heating effect (b) chemical effect
(c) magnetic effect (d) chemical, heating and magnetic effects
Answer.
(d) When an electric current is passed through a conducting solution, it causes chemical reactions. The resulting effect is called chemical effect of current.
When an electric current is passed through a bulb, its filament gets heated to a high temperature and bulb starts glowing. The resulting effect is heating effect of current. Whenever an electric current te passed through a circuit, a magnetic field is produced around it. The resulting effect is magnetic effect of current. Thus, an electric current can produce chemical, heating and magnetic effects. •
Question 2
Boojho and Paheli performed experiments taking similar bulbs and cells but two different solutions A and B as shown in the figure.
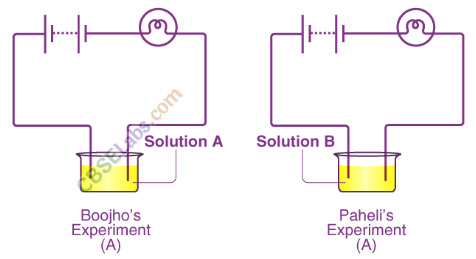
They found that the bulb in the set up A glows more brightly as compared to that of the set up B. You would conclude that
(a) higher current is flowing through the circuit in set up A
(b) higher current is flowing through the circuit in set up B
(c) equal current is flowing through both the circuits
(d) the current flowing through the circuits in the two set up cannot be compared in this manner
Answer.
(a) Bulb in the set up A glows more brightly because higher current is flowing through the circuit in set up A, as solution AIs better conductor of electricity than that of B.
Question 3
Boojho’s unde has set up an electroplating factory near his village. He should dispose off, the waste of the factory
(a) in the nearby river
(b) in the nearby pond
(c) in the nearby corn field
(d) according to the disposal guidelines of the local authority
Answer.
(d) In electroplating factory, the disposal of the used conducting solution is a major concern because it is a polluting waste, therefore it should be disposed off according to the disposal guidelines of the local authority.
Question 4
When electric current is passed through a conducting solution, there is a change of colour of the solution. This indicates
(a) the chemical effect of current (b) the heating effect of current
(c) the magnetic effect of current (d) the lightning effect of current
Answer.
(a) The passage of an electric current through a conducting solution causes chemical reactions. As a result, change of colour of solution occurs. This indicates the chemical effect of current.
Question 5
Which one of the following solutions will not conduct electricity?
(a) Lemon juice (b) Vinegar
(c) Tap water (d) Vegetable oil
Answer.
(d) Vegetable oil will not conduct electricity because it does not make ions easily.
Question 6
Which of the following metals is used in electroplating to make objects appear shining?
(a) Iron (b) Copper
(c) Chromium (d) Aluminium
Answer.
(c) Chromium is used for electroplating to make objects appear shining because chromium has a shiny appearance and it resists scratches.
Question 7
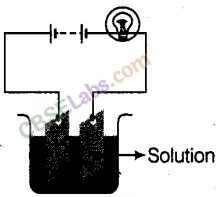
Which of the following solutions will not make the bulb glow in figure?
(a) Sodium chloride (b) Copper sulphate
(c) Silver nitrate (d) Sugar solution in diluted water
Answer.
(d) Sugar solution in diluted water does not make ions and is a poor conductor of electricity. Therefore, it will not make the current to flow through it and the bulb will not glow.
Very Short Answer Type Questions
Question 8
Fill in the blanks.
(i) The object to be electroplated is taken as……….electrode.
(ii)One of the most common applications of chemical effect of electric
current is……….
(iii)Small amount of a mineral salt present naturally in water makes it a………. of electricity.
(iv)Electroplating of………. is done on objects like water taps and
cycle bell to give them a shiny appearance.
Answer.
(i) Negative The object to be electroplated is taken as negative electrode or cathode, so that the free ions get deposited on it.
(ii) Electroplating It is thaprocess of depositing a layer of any desired metal on another material by means of electricity.
(iii) Good conductor Small amount of a mineral salt present naturally in water makes it a salt solution, therefore it becomes good conductor of electricity.
(iv) Chromlum Because chromium has a shiny appearance, it resists scratches and does not corrode.
Question 9
Why is a layer of zinc coated over iron?
Answer.
Layer of zinc is coated over iron because zinc prevents it from rust and corrosion.
Question 10
Will the solution of sugar in distilled water conduct electricity?
Answer.
No, the solution of sugar in distilled water is a poor conductor of electricity and therefore current cannot pass through it.
Question 11
Name the effect of current responsible for the glow of the bulb in an electric circuit.
Answer.
Heating effect of electric current is responsible for the bulb to glow in an electric circuit. Due to heating effect, the filament of the bulb gets heated to a high temperature and it starts glowing.
Short Answer Type Questions
Question12
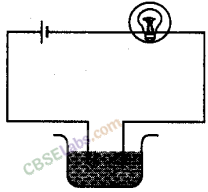
Boojho made the circuit given in the figure and observed that the bulb did not glow. On Paheli’s suggestion, he added one more cell in the circuit. The bulb now glows. Explain.
Answer.
Initially, the bulb was not glowing because the voltage required to make the bulb glow was not sufficient. With the addition of another cell, the voltage increases, i.e. the current through the bulb increases more than a particular value and the bulb starts glowing because it gets sufficient energy to heat its filament.
Question 13
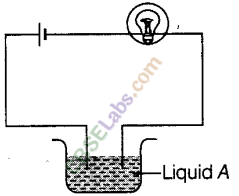
Paheli set up an experiment using liquid A in the beaker as shown in the figure. She observed that the bulb glows. Then, she replaced the liquid A by another liquid B. This time the bulb did not glow. Boojho suggested replacing the bulb by an LED. They observed that the LED glows. Explain.
Answer.
Liquid A is a good conductor of electricity and it allows the maximum current to pass through it which is sufficient to glow the bulb.
But when it is replaced by another liquid B, bulb does not glow because the current through liquid B could be weak and therefore unable to make the bulb glow.
But the small current which is passing through B is sufficient to glow a low voltage LED, so it glows.
Question 14
Paheli wants to deposit silver on an iron spoon. She took silver nitrate (AgNO3) solution in a beaker and set up a simple circuit for electroplating. Which terminal of the battery should the spoon be connected to? What mjjterial should the other electrode be made of?
Answer.
Spoon should be connected to the negative terminal of the battery. The other electrode should be made of silver, so that silver ions get deposited on an iron spoon.
Question 15
Why is tin electroplated on iron to make cans used for storing food?
Answer.
Electroplating of tin is done on the iron to make cans used for storing food because tin is less reactive than iron. Coating of tin prevents food from coming in contact with iron and thus, prevents it from getting spoiled.
Question 16
Observe figure.
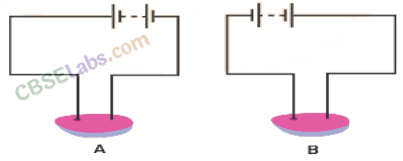
Which of these two circuits A or B shows the correct observation?
Answer.
It is not clear from the diagram that what is A and B. So, we cannot predict the actual solution. But if we compare with NCERT activity, this seems to be a potato and the correct diagram is A because positive terminal makes the greenish blue spot on the potato, due to the chemical effect of current in potato.
Question 17
Observe the following circuits carefully. In which circuit will the bulb glow? Write Yes or No in the blank space provided along each of the circuit given in the figure.
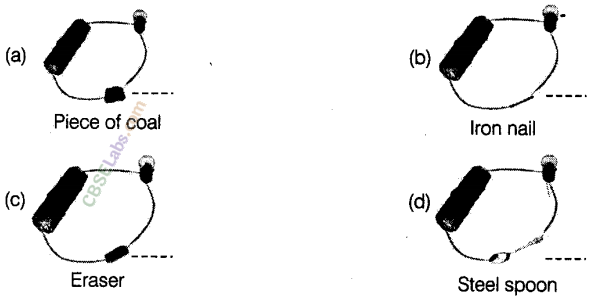
Answer.
(a) No, because piece of coal is a poor conductor of electricity.
(b) Yes, because iron nail is a good conductor of electricity.
(c) No, because eraser is a poor conductor of electricity.
(d) Yes, because steel spoon is a good conductor of electricity.
Long Answer Type Questions
Question18
An electric current is passed through a conducting solution. List any three possible observations.
Answer.
Three’ possible observations are *
(i) The colour of the solution may change depending upon the electrodes.
(ii)The temperature of the solution may increase.
(iii)Bubbles near the electrodes can be seen.’
Question19
In the circuit given in the figure,
Boojho observed that copper is deposited on the electrode connected to the negative terminal of the battery.
Paheli tried to repeat the same experiment. But she could find only one copper plate. Therefore, she took a carbon rod as negative electrode. Will copper be still deposited on the carbon rod? Explain your answer.
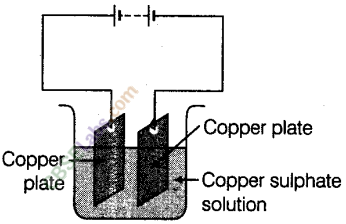
Answer.
Yes, Gopper from the copper sulphate solution will be deposited on the carbon rod. When electric current is passed through the copper sulphate solution, copper sulphate dissociates into copper and sulphate.
The free copper gets drawn to the electrode connected to the negative terminal of the battery, i.e. carbon rod and gets deposited on it. Thus, Paheli will obtain a coating of copper on carbon rod.
Question 20
Observe the circuit cjiven in the figure.
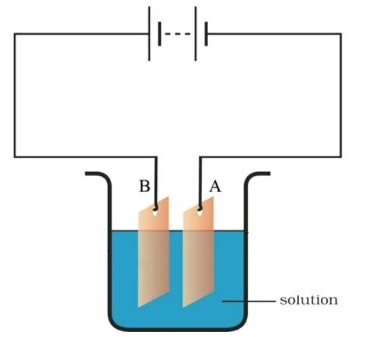
Boojho set up this circuit for purification of copper. What will be the nature of – (a) plate A (b) plate B (c) and the solution.
Explain the process of purification
Answer.
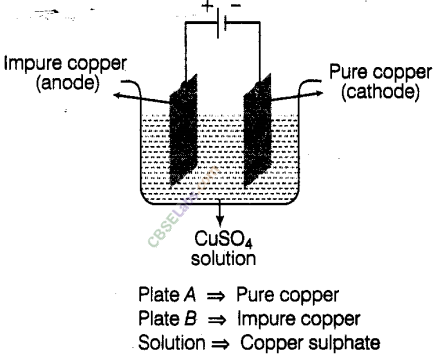
We take CuSO4as solution “and dip a pure copper plate as cathode (-ve) and impure copper plate as anode (+ve) in it. The copper from impure copper plate is transferred to the pure copper plate by the process of electroplating and impurities deposit at the bottom. In this way, the copper plate beoomes purified.
Question 21
Observe the following circuit given in the figure.
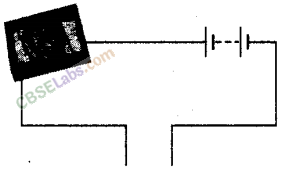
Current does not flow in the circuit, if there is a gap between the two wires. Does it indicate that air is a poor conductor of electricity? Does air never conduct electricity? Explain.
Answer.
Yes, if current is not flowing through the air gap between the wires, it means that air is a poor conductor of electricity. No, air conducts electricity only when there is great voltage or there is some moisture in air, such as during lightning.
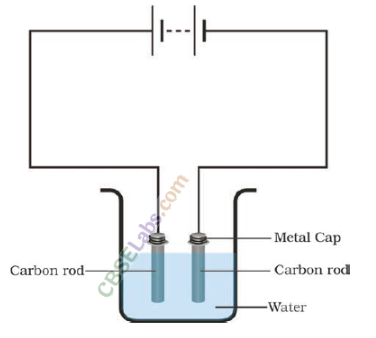
Question 22
Boojho made the circuit as shown in the figure. He wanted to observe what happens when an electric current is passed through water. But he forgot to add a few drops of lemon juice to water. Will it make any difference to his observations? Explain
Answer.
No current will pass through the circuit, if the water is distilled water and lemon juice is not added. If the water is saity, a small current will pass through the circuit. Adding few drops of lemon juice in water will increase the conductivity of the solution and large amount of current will flow easily.
Question 23
Observing that the bulb does not glow in the circuit shown in Fig. (A) Boojho changed the circuit as shown in Fig. (B). He observed deflection in the magnetic compass.
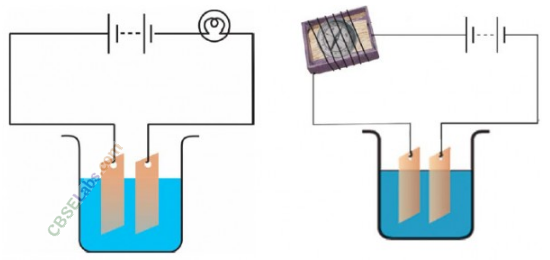
(a) What does the deflection in magnetic compass indicate?
(b) Why did the bulb not glow in Fig. (A)?
(c) What would be the effect of increase in the number of turns in the coil wound around the magnetic compass in Fig. (B)?
(d) What will be observed if the number of cells are increased in the circuit shown in Fig. (B)?
Answer.
(a) The deflection in the magnetic compass shows that the current is flowing in the circuit.
(b) The bulb did not glow, because the current passing through the circuit could be weak to make it glow. Also, it may happen because of other reasons like bulb may be fused or the connections may be loose.
(c) Increase in the number of turns in the coil wound around the magnetic compass •would increase the magnetic field around the magnetic compass which will result in the increase in the deflection in magnetic compass.
(d) Increase ‘in the number of cells will further increase the deflection in magnetic compass.
Question 24
You are provided with a magnetic compass, an empty matchbox, a battery of two cells and connecting wires. Using these objects, how will you make a tester for testing an electric circuit? Draw the necessary circuit diagram and explain.
Answer.
Take the tray from the inside of a discarded matchbox. Wrap an electric wire a few times around the tray. Place a’small compass needle inside it. Now, connect one free end of the wire to the one terminal of a battery. Leave the other end free.
Take another piece of wire and connect it to the other terminal of the battery. Join the free ends of two wires momentarily.
The compass needle shows deflection due to the magnetic effect of current. Your tester with two free ends of the wire is ready.
NCERT Exemplar Class 8 Science Solutions
- Chapter 1 Crop Production and Management
- Chapter 2 Microorganisms: Friend and Foe
- Chapter 3 Synthetic Fibres and Plastics
- Chapter 4 Materials : Metals and Non-Metals
- Chapter 5 Coal and Petroleum
- Chapter 6 Combustion and Flame
- Chapter 7 Conservation of Plants and Animals
- Chapter 8 Cell Structure and Functions
- Chapter 9 Reproduction in Animals
- Chapter 10 Reaching the Age of Adolescence
- Chapter 11 Force and Pressure
- Chapter 12 Friction
- Chapter 13 Sound
- Chapter 14 Chemical Effects of Electric Current
- Chapter 15 Some Natural Phenomena
- Chapter 16 Light
- Chapter 17 Stars and the Solar System
- Chapter 18 Pollution of Air and Water
NCERT Exemplar SolutionsNCERT Exemplar MathsNCERT Exemplar Science
We hope the NCERT Exemplar Class 8 Science Chapter 14 Chemical Effects of Electric Current help you. If you have any query regarding NCERT Exemplar Class 8 Science Chapter 14 Chemical Effects of Electric Current, drop a comment below and we will get back to you at the earliest.