Structure of the Atom Class 9 Notes – Here We have provided summary and revision notes for Class 9 Science Chapter 4. This CBSE notes contains CBSE Key Notes, CBSE Revision Notes, Short Key Notes, images, diagrams of the complete Chapter 4 titled Structure of the Atom of Science taught in class 9. If you are a student of class 9 who is using NCERT Textbook to study Science, then you must come across Chapter 4 Structure of the Atom. After you have studied lesson, you must be looking for notes to memorize. Here you can get complete Chapter 4 Structure of the Atom class 9 notes in one place. For a better understanding of this chapter, you should also see NCERT Solutions for Class 9 Science Chapter 4 Structure of the Atom.
CBSE Class 9 Science Notes Chapter 4 Structure of the Atom Pdf free download is part of Class 9 Science Notes for Quick Revision. Here we have given NCERT Class 9 Science Notes Chapter 4 Structure of the Atom.
CBSE Class 9 Science Notes Chapter 4 Structure of the Atom
Facts that Matter
- John Dalton assumed that atom is indivisible.
- In 1866 E. Goldstein discovered the presence of new radiations in a gas discharge tube and called them canal rays. These rays were positively charged radiations which led to the discovery of sub-atomic particle called proton.
In 1900 J.J. Thomson discovered the sub-atomic particle—the electron with a negative charge.
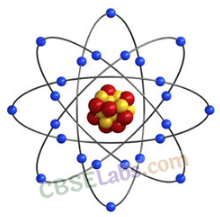
Structure of an Atom

2.Rutherford’s model of an atom
α-Particles: (+ 2 charge and 4 mass) when fast-moving a-particles are bombarded on very thin gold foil, following observations were made:
- Most of the α-particles passed straight through the gold foil.
- Some of the α-particles were deflected by the foil by small angles.
- One out of 12000 particles appeared to rebound.
Conclusions made by Rutherford based on his observations:
- Most of the space inside the atom is empty because a-particles passed through the gold foil.
- Very few particles were deflected from their path because +ve charge of the atom occupies a very little space.
- A very small fraction of a-particles were rebounded back, shows all + ve charge and mass of the gold atom is concentrated in a very small volume within an atom.
- The radius of the nucleus calculated was 105 times less than the radius of the atom.
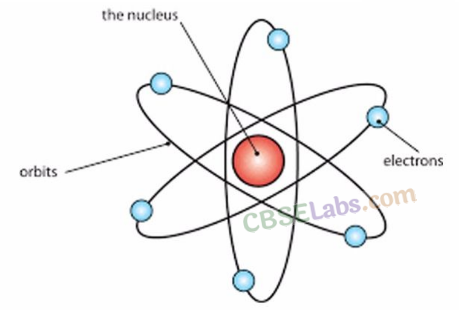
Nuclear Model of an Atom
- Centre → +ve charge → called nucleus. All mass resides in nucleus.
- Electrons → revolve around the nucleus in orbits.
- Size of the nucleus is very small as compared to the size of the atoms.
Drawbacks of Rutherford’s model of the atom: When an electron undergoes acceleration, it radiates energy. Thus revolving electron would lose energy and finally fall into the nucleus. Due to this atom should be highly unstable and hence matter would not exist in the form that we know.
But we know that atoms are quite stable.
Bohr’s Model of Atom
Postulates of Neil Bohr
- Only special orbits known as discrete orbits of electrons are allowed inside the atom.
- While revolving in discrete orbits the electrons do not radiate energy. These orbits are called energy levels.
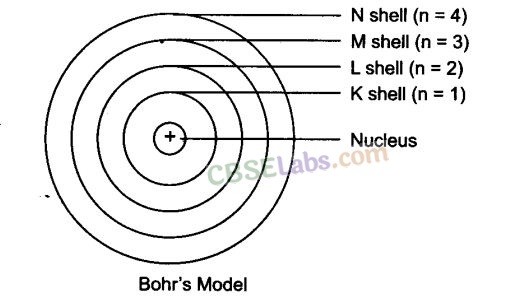
Orbits or shells are represented by K, L, M, N or the numbers, n = 1, 2, 3, 4
Neutrons
Distribution of electrons in different orbits (Shells) given by Bohr and Bury: Rules:
- Maximum number of electrons present in a shell is given by 2n2 (n = shell number)
E.g., n = 1 (K shell) 2(1)2 = 2 electron - The maximum number of electrons that can be accommodated in the outermost orbit is 8.
- Electrons are not accommodated in a given shell unless the inner shells are completely filled.
Definitions
- Valency: The combining capacity of an atom is called its valency,
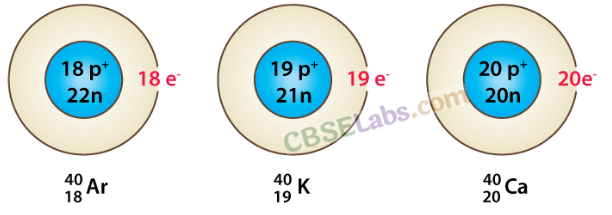
- Atomic number: It is equal to a number of protons.
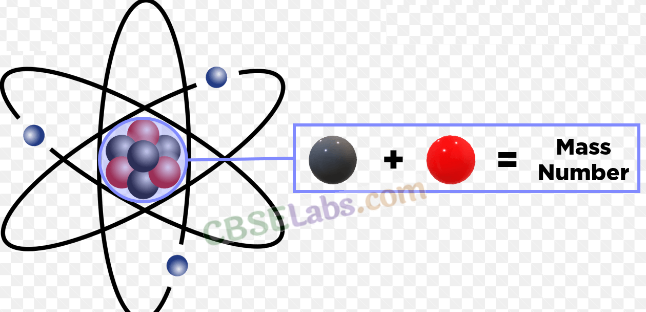
- Mass number: It is equal to the sum of protons and neutrons.
Isotopes: Atoms of the same element with same atomic number but a different mass number, are called isotopes.
Chemical properties → same but Physical properties → different
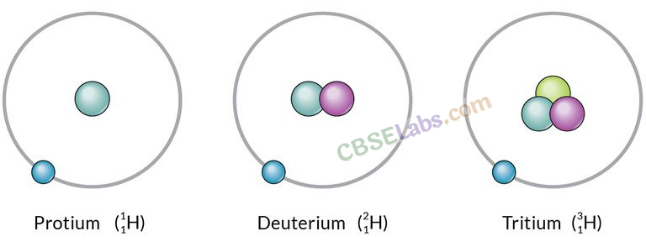
Applications of isotopes:
- Anisotope of Uranium used as fuel.
- Anisotope of Cobalt is used in the treatment of cancer
- Anisotope of Iodine is used in the treatment of goitre.
Isobars: Atoms of different elements with same mass number but different atomic numbers are called isobars.
NCERT Notes for Class 9 Science
- Chapter 1 Matter in Our Surroundings Class 9 Notes
- Chapter 2 Is Matter Around Us Pure Class 9 Notes
- Chapter 3 Atoms and Molecules Class 9 Notes
- Chapter 4 Structure of the Atom Class 9 Notes
- Chapter 5 The Fundamental Unit of Life Class 9 Notes
- Chapter 6 Tissues Class 9 Notes
- Chapter 7 Diversity in Living Organisms Class 9 Notes
- Chapter 8 Motion Class 9 Notes
- Chapter 9 Force and Laws of Motion Class 9 Notes
- Chapter 10 Gravitation Class 9 Notes
- Chapter 11 Work, Power And Energy Class 9 Notes
- Chapter 12 Sound Class 9 Notes
- Chapter 13 Why Do we Fall ill Class 9 Notes
- Chapter 14 Natural Resources Class 9 Notes
- Chapter 15 Improvement in Food Resources Class 9 Notes
We hope the given CBSE Class 9 Science Notes Chapter 4 Structure of the Atom Pdf free download will help you. If you have any query regarding NCERT Class 9 Science Notes Chapter 4 Structure of the Atom, drop a comment below and we will get back to you at the earliest.