Students must start practicing the questions from CBSE Sample Papers for Class 9 Science with Solutions Set 2 are designed as per the revised syllabus.
CBSE Sample Papers for Class 9 Science Set 2 with Solutions
Time Allowed :3 Hours
Maximum Marks: 80
General Instructions:
Read the following instructions carefully.
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section – A
(Select and write the most appropriate option out of the four options given for each of the questions 1 -20. There is no negative mark for incorrect response:)
Question 1.
Alia mixed Bacl2 solution and CuSO4 solution in a closed conical flask. [1]
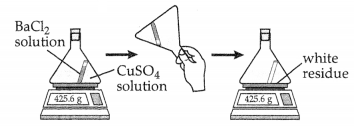
What can he concluded from the result of the experiment?
(A) Total mass of the chemicals remains the same.
(B) Total volume of the chemicals remains the same.
(C) State of matter of the chemicals remains the same.
(D) Composition of the chemicals remains the same.
Answer:
Option (A) is correct.
Explanation: As per the law of conservation of mass, total mass of the chemicals remains the same.
Question 2.
During summer, water kept in an earthen pot becomes cool because of the phenomenon of: [1]
(A) Diffusion
(B) Transpiration
(C) Osmosis
(D) Evaporation
Answer:
Option (D) is correct.
Explanation: It is because of the phenomenon called evaporation. An earthen pot has a large number of tiny pores in its walls and some of the water molecules continuously keep seeping through these pores to outside the pot. This water evaporates continuously and takes the latent heat required for vaporization from the remaining water. In this way, the remaining water loses heat and gets cooled.
Question 3.
Which of the following are chemical changes? [1]
(i) Decaying of wood
(ii) Burning of wood
(iii) Sawing of wood
(iv) Hammering of a nail into a piece of wood
(A) (i) and (ii)
(B) (ii) and (iii)
(C) (iii) and (iv)
(D) (i) and (iv)
Answer:
Option (A) is correct.
Explanation. Decaying of wood and burning of wood are chemical changes because in these processes, the chemical composition of wood is changed and new substances are formed. which cannot be converted back into their original form. Sawing of wood and hammering of a nail into a piece of wood are physical changes. I
Question 4.
The picture shows the symbol for sodium. [1]
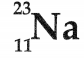
What can be concluded about sodium from the symbol?
(A) It contains 11 neutrons
(B) It contains 12 protons
(C) It contains 12 neutrons
(D) It contains 34 electrons
Answer:
Option (C) is correct.
Explanation: Atomic number, i.e., the number of protons in the element is written at the bottom and atomic mass number which is the sum of protons and neutrons is written at the top of the symbol. It means the sodium atom contains 11 protons and 12 (23-11) neutrons.
Question 5.
Rutherford’s alpha particle scattering experiment resulted into the discovery of: [1]
(A) Electron
(B) Proton
(C) Nuclens in the atom
(D) Atomic mass
Answer:
Option (C) is correct.
Explanation: Rutherfords’ alpha particle experiment led to the discovery of the nucleus in the atom.
Question 6.
Which condition out of the following will increase the evaporation of water? [1]
(A) Increase in temperature of water
(B) Decrease in temperature of water
(C) Less exposed surface area of water
(D) Adding common salt to water.
Answer:
Option (A) is correct.
Explanation: On increasing the temperature, kinetic energy of water molecules increases and more particles get enough kinetic energy to go into the vapour state- This increases the rate of evaporation. On the other hand, decrease in temperature of water, less exposed surface area of water, and addition of common salt to water decrease the rate of evaporation.
Question 7.
The boiling points of diethyl ether, acetone, and n-butyl alcohol are 35°C, 56°C and 118°C, respectively. Which one of the following correctly represents their boiling points in Kelvin scale? [1]
(A) 306K,329K,391K
(B) 308K,329K,392K
(C) 308K,329K,391K
(D) 329K,392K,308K
Answer:
Option (C) is correct.
Explanation: On applying the formula,
T°C + 273 = K,
The boiling point of diethyl ether 35°C + 273 = 308 K
Boiling point of acetone = 56° C + 273 = 329 K
and Boiling point of n-butvl alcohol = 118°C+273 = 391K.
Hence, the correct order of boiling points in Kelvin scale is 308 K, 329 K, and 391 K.
Question 8.
Girth of stem increases due to: [1]
(A) Apical meristem
(B) Lateral meristem
(C) vertical meristem
(D) Intercalary meristem
Answer:
Option (B) is correct.
Explanation: Girth of the Stem increases due to lateral meristematic tissue. They are found beneath the bark (called cork cambium) and in vascular bundles of dicot roots and sterns (called vascular cambium) as thin layers. This increase in the diameter or girth of the plant is called secondary growth.
Question 9.
The undefined nuclear region of prokaryotes is also known as: [1]
(A) Nucleus
(B) Nucleolus
(C) Nucleic acid
(D) Nucleoid
Answer:
Option (D) is correct.
Explanation: The undefined nuclear region of a prokaryotic cell is called nuclecid. The prokaryotic cells lack true nucleus. A circular
DNA lies naked in the cytoplasm.
Question 10.
The proteins and lipids, essential for building the cell membrane, are manufactured by: [1]
(A) Endoplasmic reticulum
(B) Golgi apparatus
(C) Plasma membrane
(D) Mitochondria
Answer:
Option (A) is correct.
Explanation: Rough endoplasmic reticulum (RER) is associated with the synthesis of proteins while smooth endoplasmic reticulum (SER) is associated with the synthesis of lipids.
Question 11.
Which muscles act involuntarily? [1]
(i) Striated muscles
(ii) Smooth muscles
(iii) Cardiac muscles
(iv) Skeletal muscles
(A) (i) and (ii)
(B) (ii) and (iii)
(C) (iii) and (iv)
(D) (i) and (iv)
Answer:
Option (B) is conect.
Explanation: The working of both smooth and cardiac muscles arc involuntarily while skeletal (also known as or striated) muscles move according to our will i.e. are voluntary in action.
Question 12.
Which of these properties qualifies Amoeba as eukaryote? [1]
(A) It is unicellular
(B) It needs food for energy
(C) It has a membrane-bound nucleus
(D) It is surrounded by a plasma membrane
Answer:
Option (C) is correct
Explanation: Amoeba has membrane-bound nucleus which is a characteristic found in eukaryotic cells only.
Question 13.
Area under the v – t graph represents a physical quantity, which has the unit: [1]
(A) m2
(B) m
(C) ms2
(D)ms-1
Answer:
Option (B) is correct.
Explanation: Area under v-t graph represents displacement whose unit is metro or (m).
It is because, unit of velocity v = m/s and unit of time = s.
Hence, unit of (v-t) graph m/s × s = m.
Hence, the unit of (v-t) graph is metre (m).
Question 14.
In case of negative work, the angle between the force and displacement is? [1]
(A) 0°
(B) 45°
(C) 90°
(D) 180°
Answer:
option (D) is correct
Explanation: In case of negative work, the angle between the force and displacement is 18o°.
Question 15.
Which one of the following nutrients is not available in fertilisers? [1]
(A) Nitrogen
(B) Phosphorus
(C) Iron
(D) Potassium
Answer:
Option (C) is correct
Explanation: Fertilisers supply nitrogen, phosphorus and potassium (NPK), but not iron.
Question 16.
The quality of honey differs from sample to sample. Which of these decides the quality of a honey sample? [1]
(A) Time of the day when the bees collect nectar.
(B) Time taken b’ the bees to build the beehive.
(C) type of flower from which the bees collect nectar.
(D) Size of the beehive from which the honey is collected.
Answer:
Option (C) is coned.
Explanation: The type of flower from which the bees collect nectar decides the quality of honey.
Assertion-Reason Based Questions
Question No. 17 to 20 consist of two statements – Assertion (A) and Reason (R). Answer these questions
by selecting the appropriate option given below:
(A) Both (A) and (R) are true, and (R) is the correct explanation of (A).
(B) Both (A) and (R) are true, and (R) is not the correct explanation of (A).
(C) (A) is true but (R) is false.
(D) (A) is false but (R) is true.
Question 17.
Assertion (A): è prefer to wear cotton clothes during summer.
Reason (R): Cotton clothes are good absorber of water. [1]
Answer:
Option (A) is correct.
Explanation: Cotton being good absorber of water helps in absorbing the sweat, which on evaporation gives a cooling sensation in the body.
Question 18.
Assertion (A): Mitochondria are semi-autonomous cell organdies.
Reason (R): Mitochondria generate energy. [1]
Answer:
Option (B) is correct.
Explanation: Mitochondria have their own DNA and ribosome to make proteins. They can replicate independently of nuclear DNA. Hence, they are known as semi-autonomous organelles. They are also associated with ATP production.
Question 19.
Assertion (A): On Moon, humans feel lighter than on Earth.
Reason (R): It is due to more gravitational force exerted by Moon on man. [1]
Answer:
Option (C) is correct.
Explanation: On Moon, humans feel lighter than on Earth. It is due to less gravitational force exerted by Moon on man.
Question 20.
Assertion (A): Cattles are fed with roughage and concentrates.
Reason (R): Roughage provides fibers while concentrates provide proteins and other nutrients. [1]
Answer:
Option (A) is correct.
Explanation: Roughage produces fibres while concentrates provide proteins and other nutrients. Hence cattle are fed with roughage and concentrates.
Section – B
(Questions No. 21 to 26 are very short answer questions)
21. Write the chemical formula for: [2]
(a) Zinc phosphate
(b) Lead carbonate
Answer:
(a) Zn3(PO4)2
(b) PbCO3
Question 22.
Distinguish between cell wall and cell membrane. [2]
Answer:
Differences between cell wall and cell membrane:
| Cell wall | Cell membrane |
| (1) It is present only in plant cells. | It occurs both in animal cells and plant cells. |
| (2) It is dead in nature and permeable. | It is a living membrane and is semi-permeable. |
Question 23.
Why is the plasma membrane called a selectively permeable membrane? [2]
OR
Differentiate between voluntary and involuntary muscles. Give one example of each type.
Answer:
The plasma membrane is called a selectively permeable membrane because it allows only some substances to pass through it.
OR
Differences between voluntary and involuntary muscles:
| Voluntary muscles | Involuntary muscles |
| (1) Those muscles whose action is normally controlled by an individual’s will, | Those muscles that contract without conscious control. |
| (2) Example: Skeletal muscles which are present in limbs (quadriceps, biceps, and pectoralis. etc.) | Example: Smooth muscles which are present in iris or the eye, bronchi of the lungs, and ureter. |
Question 24.
The table below shows the speed of a bus in three hours of its travel.
| Time | First hour | Second-hour | Third hour |
| Speed of the bus | 35 km/h | 60 km/h | 40 km/h |
Calculate the average speed of the bus.
Answer:
Average speed = \(\frac{\text { Total distance covered }}{\text { Total Time taken }}\)
Here is the total distance covered
35 km +60 km + 40 km 135 km
Total time taken = 3 hours
Therefore.
Average speed = \(\frac{135}{3} \) = 45 km/hr
Hence, the average speed of the bus is 45 knVhr.
Question 25.
The following diagram shows a simple pendulum consisting of a bob of mass 100 g. Initially, the bob of the pendulum is at rest at ‘O’. Ills then displaced to one side at A. The height of ‘A’ above ‘O’ is 5 cm. What is the value of kinetic energy and potential energy of the bob at the position P’ whose height above O is 2 cm? [2]
OR
When an object is immersed in the fluid name the two forces acting on it? [2]
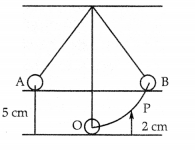
Answer:
We know that potential energy RE. is given by formula,
P.E. = mgh
Therefore, at height 5 cm
P.E. = 0.1 kg × 10 m/s2 × 0.05m
P.E. = 0.05 J
Now, at point A,
Total energy = P.E. + K.E.
E = 0.05 + 0 = 0.05 J (As the bob starts (rom
rest, K.E. will be zero.)
Now, at point P (at height 2cm),
P.E = 0.1 kg ×10 m/s2 × 0.02 m
P.E = 0.02 J
Now
K.E. = Total energy -PE.
K.E. = 0.05-0.02 = 0.03 J
OR
When an object is immersed in the fluid name the two forces acting on it are downward gravitational force and upward buoyant force.
Question 26.
The diagram shows a composite fish culture pond. [2]
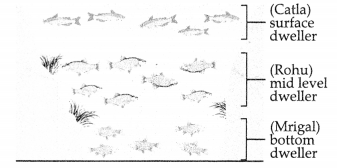
(a) What is composite fish culture?
(b) What is the advantage of such composite fish culture?
Answer:
(a) Composite hsh culture is a process of growing different types of fish in the same pond.
(b) Its advantage is, all areas of the pond are utilized for better fish production.
Section – C
(Questions No. 27 to 33 are short answer questions)
Question 27.
(a) Why path of light is not visible in a solution when a beam of light is passed through it?
(b) Classify each of following as solution, colloid or suspension:
(i) A mixture whose particles are big enough to scatter a beam of light passing through it.
(ii) A mixture whose particles settle down when it is left undisturbed. [3]
Answer:
(a) Because of small size, the particles cannot scatter a beam of light that is why path of light is not visible in a solution when a beam of light is passed through it.
(b)
- Colloids
- Suspension.
Question 28.
What is the effect of change of pressure on physical state of matter? Explain with an example of a gas. [3]
OR
There are. two elements A and B. Find the number of sub-atomic particles in each of these elements. What is the relationship between the two elements?
Answer:
The physical state of matter can be changed by changing the pressure. By lowering the temperature and increasing the pressure, gases can be changed into liquids and some solids can be changed into gases on decreasing the pressure. This happens with gases as there is lots of space between the particles of a gas and upon applying high pressure, particles come down to each other which upon cooling gets liquefied.
OR
A1326 electrons = 13 protons + 13 neutrons
Atomic number = 26-13 13
B1426 electrons = 14 protons + 12 neutrons
Atomic number = 26- 12 = 14
They are isobars.
Isobars are any member of a group of atomic or nuclear species all of which have the same mass number and have different atomic numbers.
Question 29.
Write three differences between prokaryotic and eukaryotic cells. [3]
Answer:
Differences between prokaryotic and eukaryotic cells:
| Prokaryotic cell | Eukaryotic cell |
| (1) Size: Generally small (1 – 10 µm) 1µm=10-6 m. |
Size: Generally large (5-100 µm) |
| (2) Nuclear region: Contains only nucleic acid and is undefined due to the absence of nuclear membrane and is known as nucleoid. |
Nuclear region: Well-defined and surrounded by a nuclear membrane. |
| (3) Membrane-bound cell organelles absent. | Membrane-bound cell organelles (e.g., chloroplasts, Golgi bodies, etc.) present. |
Question 30.
Explain in brief any three roles of epidermis in plants. [3]
Answer:
- The epidermis protects all parts of the plants.
- Epidermal cells on the aerial part of the plant often secrete a waxy, water-resistant layer which helps in protection against water loss and mechanical injury.
- The epidermis protects against invasion of parasitic fungi.
Question 31.
State the law of inertia. Why do we fall in forward direction if a moving bus stops suddenly and fall in the backward direction if it suddenly accelerates from rest? [3]
Answer:
Law of inertia: An object remains in its state of rest or of uniform motion in a straight line until an external unbalanced force acts on it. When a moving bus stops suddenly, the bus slows down but our body tends to remain in the state of motion due to inertia of motion, Sudden start of bus brings motion to the bus as well as our feet but rest of the body still has inertia of rest due to which we fall backward.
Question 32.
Name the physical quantities denoted by:
(a) the slope of the distance-time graph
(b) the area under velocity-time graph
(c) the slope of velocity-time graph. [3]
Answer:
(a) Speed
(b) Displacement
(c) Acceleration
Question 33.
In the musical instrument jal-tarang, the bowls contain different amounts of water.
(a) Which of the howls produces a low pitch sound?
(b) Which of the bowls produces a high-pitched sound?
(c) Which wave property determines the pitch? [3]
Answer:
(a) The bowl that contains the maximum quantity of water produces low pitch sound.
(b) The bowl that contains the least quantity of water produces high pitch sound.
(c) Frequency of sound waves determine the pitch.
Section – D
(Questions No. 34 to 36 are long answer questions)
Question 34.
(a) State two ways by which you can change a saturated solution to unsaturated solution.
(b) Distinguish between homogeneous and heterogeneous mixtures by giving one example of each. [5]
OR
When a solid melts the temperature of the system does not change after the melting point is reached even when we continue to supply heat. Give reason. Define latent heat of vaporisation. Which will cause more severe burns-boiling water or steam and why? [5]
Answer:
(a) Two ways by which we can change a saturated solution to unsaturated solution are:
- By increasing the temperature/by heating the solution.
- By increasing the amount of solvent.
| Homogeneous Mixture | Heterogeneous Mixture |
| (i) Uniform composition. | Non-uniform composition. |
| (ii) No distinct boundaries of separation. e.g., sugar ± water. |
Distinct boundaries of separation. e.g. sand + water. |
OR
When a solid melts the temperature of the system does not change after the melting point is reached even when we continue to supply heat because the supplied heat energy gets used up in changing the state by’ overcoming the forces of attraction between the particles. This is called latent heat.
Latent heat of vaporisation: The amount of heat energy that is required to change 1 L of a liquid into gas at atmospheric pressure at its boiling point. Steam will give more severe burns because particles of steam have extra energy in the form of latent heat of vaporisation.
Question 35.
(a) What are the consequences of the following conditions?
(i) A cell having higher water concentration than the surrounding medium.
(ii) A cell having lower water concentration than the surrounding medium.
(iii) A cell having equal water concentration to its surrounding medium.
(b) Name the materials of which the cell membrane and cell wall are composed of,
OR
The growth of plant occurs only in specific regions:
(a) Name the tissue which is responsible for this growth.
(b) State the different types of this tissue.
(c) Write one function of each of the above-mentioned tissue. [5]
Answer:
(a)
- When a cell possesses higher water concentration than the surrounding medium then exosmosis occurs in the cell due to the difference in concentration. As a result, the cell shrinks.
- When a cell has low water concentration than surrounding medium then endosmosis occurs that results in the swelling of the cell.
- A cell having equal water concentration to its surrounding medium will not show any change.
(b) Cell wall is composed of cellulose and cell membrane is composed of lipids and proteins.
OR
- Meristematic tissue
- The different types of meristematic tissue arc:
- Apical Meristem
- Iritercalary Meristem
- Lateral Meristem.
(c) Apical meristem: Helps in growth of the stem and the root.
- Intercalary meristem: Helps in elongating internodes of plants like sugarcane.
- Lateral meristem: Helps in growth and development of plant’s girth (both shoot and root).
Question 36.
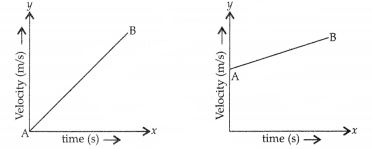
(a) Give one similarity and one dissimilarity between the two graphs.
(b) What do you understand by the term acceleration? What is meant by its being positive or negative? Explain with example. Write its SI units. [5]
OR
(a) Write the formula to find the magnitude of gravitational force between the Earth and an object on the Earth’s surface.
(b) Derive how does the value of gravitational force ‘F’ change between two objects when the:
(i) distance between them is reduced to half, and
(ii) mass of one object is increased four times.
Answer:
(a) Similarity: Both the graphs show uniform acceleration. 2
Dissimilarity: In first graph, the body starts from rest (u = O) while in the second graph the initial velocity is non-zero (u ≠ 0).
(b) Acceleration of a body is defined as the rate of change of its velocity with time Acceleration being positive means the velocity of the body is increasing while it being negative means the velocity is decreasing.
The Si unit of the acceleration is m/s2.
OR
(a) F = \(\frac{\mathrm{GMm}}{\mathrm{R}^2}\)
(b) According to the law of gravitation, the force of attraction acting between two bodies is given by,
F = \(\frac{\mathrm{GMm}}{\mathrm{R}^2}=\mathrm{G} \frac{\mathrm{Mm}}{\left(\frac{\mathrm{R}}{2}\right)^2} \)
= \(4 \mathrm{G} \frac{\mathrm{Mm}}{\mathrm{R}^2}\) = 4F
Thus, when the distance between the objects is reduced to half, the gravitational force increases by four times the original force
F=GM x \(\frac{4 \mathrm{~m}}{\mathrm{R}^2}\) =4F
So, as the mass of any one of the objects is increased four times, the force is also increased by four times.
Section – E
(Questions No. 37 to 39 are case-based! data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.)
Question 37.
In the following table, the mass number and the atomic number of certain elements are given. Study the given data and answer the following questions:
| Elements | Mass No. | Atomic No. |
| A | 1 | 1 |
| B | 7 | 3 |
| C | 14 | 7 |
| D | 40 | 18 |
| E | 40 | 20 |
(a) Which of the elements A, B, C, D will tend to form a cation?
(b) Which of the above elements is a noble gas?
(c) Which of the elements A, B, C, and D will tend to form an anion? [4]
OR
Which two elements are isobars of each other?
Answer:
(a) Element B has number of protons greater than number of electrons. So, it will tend to form a cation.
(b) The atomic number of element D is 18. So, its electronic configuration will be 2. 8, 8. The outermost shell is complete, so, it is a noble gas.
(c) Element A has number of electrons greater than number of protons. So, it will tend to form an anion.
OR
Element D and E both have same mass number, i.e., 40 but different atomic numbers so they are isobars of each other.
Question 38.
Observe the following diagram which shows some Localised tissues and answer the questions:
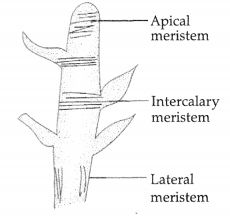
(a) Identify the diagram.
(b) What is apical meristem? Where do we find it?
(c) Which type of meristem, is found at the base of leaves and internodes? [4]
OR
which part helps in growth and development of plant’s girth?
Answer:
(a) The diagram shows meristematic tissues.
(b) Meristem present at the growing tips of stem and root is called apical meristem. It brings about increase in length of the stem and the root.
(c) Intercalary meristem is found at the base of leaves and internodes.
OR
Meristem located on the lateral portion of plant (lateral meristem) is responsible for increasing the girth of root and stem.
Question 39.
Waves of frequency 200 Hz are produced in a string as shown in the figure. Answer the following questions as given:
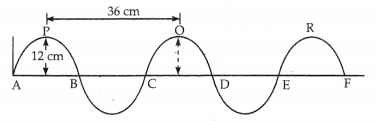
(a) Find the amplitude of the wave.
(b) Find the velocity of the wave.
(c) Find the wavelength of the wave.
OR
What is the frequency of a sound wave? [4]
Answer:
(a) Amplitude = Maximum displacement = 12 cm
(b) Frequency (n) = 200 Hz, Wavelength (λ)= 0.36m,
Now, ν = nλ
ν = 200Hz × 0.36m
ν=72m/s
(c) Wavelength = Distance between two crests = 36 cm = 0.36m
OR
The frequency of a sound wave is the number of oscillations per second.