Students must start practicing the questions from CBSE Sample Papers for Class 11 Chemistry with Solutions Set 5 are designed as per the revised syllabus.
CBSE Sample Papers for Class 11 Chemistry Set 5 with Solutions
Time Allowed : 3 hours
Maximum Marks: 70
General Instructions:
- There are 33 questions in this question paper with internal choice.
- Section – A consists of 16 multiple -choice questions carrying 1 mark each.
- Section – B consists of 5 short answer questions carrying 2 marks each.
- Section – C consists of 7 short answer questions carrying 3 marks each.
- Section – D consists of 2 case – based questions carrying 4 marks each.
- Section – E consists of 3 long answer questions carrying 5 marks each.
- All questions are compulsory.
- Use of log tables and calculators is not allowed.
Section-A
The following questions are multiple-choice questions with one correct answer. Each question carries 1 mark. There is no internal choice in this section.
Question 1.
How many significant figures are there in 3.070 & 0.0025?
(A) 2 & 5
(B) 4 & 2
(C) 1 & 2
(D) 6 & 4
Answer:
(B) 4 & 2
Question 2.
How many number of atoms are present in 52 u of He?
(A) 11
(B) 15
(C) 13
(D) 12
Answer:
(C) 13
Explanation:
4u of He = 1 atom of He
∴ 52 u of He = \(\frac{1}{4}\) × 52
= 13 atoms of He.
![]()
Question 3.
Ethyne on passing through red hot iron tube at 873K undergoes cyclic polymerization. Three molecules of ethyne polymerise to form:
(A) ethane
(B) ethene
(c) benzene
(D) ethanol
Answer:
(c) benzene
Explanation:
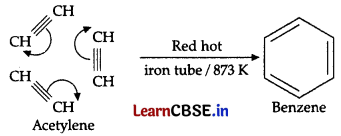
Question 4.
Which element is isoelectronic with Na+?
(A) Ne
(B) He
(C) K+
(D) C
Answer:
(A) Ne
Number of electrons in Na+ = 11 — 1 = 10
Number of another element having 10 electron = Ne Therefore, Ne is iso-electronic with Na+.
Question 5.
The element with atomic number 57 belongs to:
(A) s-block
(B) p-block
(C) d-block
(D) f-block
Answer:
(C) d-block
Question 6.
Hydrogen bonds are formed in many compounds, e.g., H20, HF, NH3. The boiling point of such compounds depends to a large extent on the strength of hydrogen bond and the number of hydrogen bonds. The correct decreasing order of the boiling points of above compounds is:
(A) HF > H2O > NH3
(B) H20 > HF > NH3
(C) NH3 > HF > H2O
(D) NH3 > H2O > HF
Answer:
(B) H20 > HF > NH3
The size and electronegativity of an atom both affect the strength of hydrogen bonding. The size of an atom an
electronegativity are inverseiy related, smaller the size, higher is the electronegativity there are two molecules of HF attcahed through hydrogen bonding , whereas four molecules in water, conjugative stability.
Question 7.
The pressure-volume work for an ideal gas can be calculated by using the expression W = \(\int_{\mathrm{V}_i}^{\mathrm{V}_i} \mathrm{P}_{e x} d \mathrm{~V}\) The work can also be calculated from the pV- plot by using the area under the curve within the specified limits. When an ideal gas is compressed (a) reversibly or (b) irreversibly from volume V, to Vf choose the correct option.
(A) w (reversible) = w (irreversible)
(B) w (reversible) < w (irreversible) (C) w (reversible) > w (irreversible)
(D) w (reversible) = w (irreversible) + pexDV
Answer:
(B) w (reversible) < w (irreversible)
Explanation:
Area under the curve is always more in irreversible compression as can be seen from fig (a) and (b)
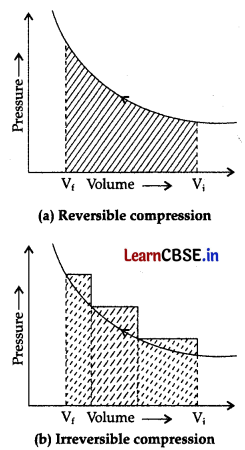
Question 8.
The entropy change can be calculated by using the expression ΔS = qrevT. When water freezes in a glass beaker, choose the correct statement amongst the following:
(A) AS (system) decreases but AS (surroundings) remains the same. –
(B) AS (system) increases but AS (surroundings) decreases.
(C) AS (system) decreases but AS (surroundings) increases.
(D) AS (system) decreases and AS (surroundings) also decreases.
Answer:
(C) AS (system) decreases but AS (surroundings) increases.
Explanation:
Freezing is exothermic process. The heat released increases the entropy of surroundings.
Question 9.
During hearing of a court case, the judge suspected that some changes in the documents had been carried out. He asked the forensic department to check the ink used at two different places. According to you, which technique can give the best results?
(A) Column chromatography
(B) Solvent extraction
(C) Distillation
(D) Thin layer chromatography
Answer:
(D) Thin layer chromatography
Question 10.
What will be the value of pH of 0.01 mol dm-3 CH3COOH (Ka = 1.74 × 10-5)?
(A) 3.4
(B) 3.6
(C) 3.9
(D) 3.0
Answer:
(A) 3.4
Explanation:
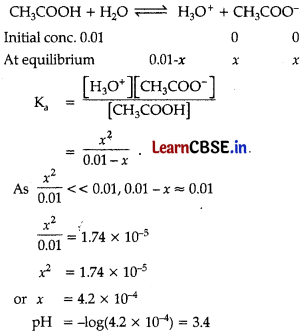
Question 11.
Which one is more stable cation?
(A) CH3)33C+
(B) CH3C+H2
(C) C+H3
(D) All of these
Answer:
(A) (CH3)33C+
Explanation:
Hyperconjugation interaction in (CH3)3C+ is greater than in CH3C+H2 as the (CH3)3C+ has nine C-H bonds. In C+H3, vacant p orbital is perpendicular to the plane in which C-H bonds lie, hence cannot overlap with it. Thus, C+H3 lacks hyper conjugative stability.
![]()
Question 12.
Which of the following is not an example of redox reaction?
(A) CuO + H2 → Cu + H2O
(B) Fe2O3 + 3CO → 2Fe + 3CO2
(C) 2K + F2 2KF
(D) BaCl2 + H2SO<sub4 → BaSO4 + 2HC1
Answer:
(D) BaCl2 + H2SO<sub4 → BaSO4 + 2HC1
Explanation:
It is not a redox reaction as there is no change in oxidation number of any of the reactants. It is an example of double displacement reaction.
Question 13.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Among the two O-H bonds in H20 molecule, the energy required to break the first O-H bond and the other O-H bond is the same.
Reason (R): This is because the electronic environment around oxygen is the same even after breakage of one O-H bond.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(D) A is false but R is true.
Answer:
(D) A is false but R is true.
Explanation:
In case of H2O molecule, the enthalpy needed to break the two O – H bonds is not the same. The difference in the AaH° value shows that the second O-H bond undergoes some change because of changed chemical environment.
Question 14.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Work and internal energy of a system are state functions.
Reason (R): The sum of q + w is a state function.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(D) A is false but R is true.
Explanation:
Work is a path function while internal energy is a state function. It is true that (q + w) is a state function.
Question 15.
Given below are two statements labelled as Assertion (A) and Reason (R). Assertion (A): Pent-1-ene and pent-2-ene are position isomers.
Reason (R): Position isomers differ in the position of functional group or substituent. Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(A) Both A and R are true and R is the correct explanation of A
![]()
Question 16.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Cyclobutane is less stable than cyclopentane Reason (R): Presence of bent bonds causes loss of orbital overlap.
Select the most appropriate answer from the options given below:
(A) Both A and R are true and R is the correct explanation of A
(B) Both A and R are true but R is not the correct explanation of A.
(C) A is true but R is false.
(D) A is false but R is true.
Answer:
(C) A is true but R is false.
Section-B
This section contains 5 questions with internal choice in one question. The following questions are very short answer type and carry 2 marks each.
Question 17.
(a) Which is more informative? Empirical formula or molecular formula.
(b) Calculate the number of carbon atoms in 34.20g of C6H,206.
Answer:
(a) As molecular formula gives exact number of atoms present in the compound, so molecular formula is more informative.
(b) Number of particles in a mole = 6.022 × 1023
Molecular mass of C6H2O6 = 180
Number of carbon atoms = 6
∴ 180 g of C6H12O6 contains
= 6 × 6.022 × 1023 carbon atoms
34.20 g of C6H12O6 contains
= \(\frac{6 \times 6.022 \times 10^{23} \times 34.20}{180}\)
= 6.865 × 1023 carbon atoms
Question 18.
How would you explain the fact that first ionisation enthalpy of sodium is lower than that of magnesium but its second ionisation enthalpy is higher than that of magnesium? :
Answer:
The first electron in both the cases has to be removed from 3s-orbitals, but nuclear charge of Na is less than that of Mg. Hence, I.E. of Na is lower than that of Mg.
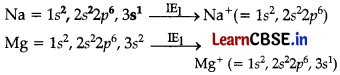
After the loss of first electron, the electronic configuration ofNa+ is 1s2,2s22p6, i.e., that of noble gas which is very stable and hence the removal of second electron from Na+ is very difficult. In the case of Mg, after the loss of first electron, electronic configuration of Mg+ ions is 1s2, 2s22p6, 3s1. The second electron to be removed is from 3s orbital which is easier. Hence, IE2 of sodium is much larger than IE2 of Mg.
Question 19.
Consider the following species:
N3-, O2-, F– , Na+, Mg2+ and Al3+
(a) What is common in them?
(b) Arrange them in the order of increasing ionic radii.
Answer:
(a) Since, all these species have equal number of electrons, i.e., 10 therefore, these are isoelectronic species. (1)
(b) In isoelectronic species, greater the nuclear charge, lesser will be the ionic radius therefore, increasing order of ionic radii:
Al3+ < Mg2+< Na+ < F– < O2- < N3- (1)
![]()
Question 20.
(a) Name the hybridisation involved in:
(i) C2H2
(ii) SF6
(b) Why dipole moment of CO2 is zero while H2O is a polar though both have similar formula?
OR
(a) Calculate the bond order for N2+ molecule.
(b) Water molecule has bent structure whereas BeCl2 has a linear structure. Give Reason
Answer:
(a) (i) C2H2 – sp hybridisation
(ii) SF6 – sp3d2 hybridisation
(b) Due to sp hybridisation, geometry of CO2 molecule is linear. So the resultant dipole moment becomes zero.

But H2O has sp3 hybridisation and possesses bent structure due to which the dipole moment adds up. Hence, H2O is polar molecule.
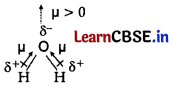
Or
Bond order on N2+:
N2+: No. of electrons = 14 — 1 = 13
Molecular orbital configuration:

(b) Be (Atomic number 4) has a total of 4 electrons. Its electronic configuration is Is2 2s2, It has two valence electrons and no lone pair of electrons. Each of the two electrons can form bonds with chlorine atoms. Chemical bonds involve electrons and because all electrons have a negative charge, the two bonds between the Be and Cl will arrange themselves around the central atom (Be) so that the electrons in the bonds are as far apart as possible. Thus, the molecule is linear with the chlorines on opposite sides of the Be.
![]()
In case of water, oxygen has a total of 8 electrons, with electronic configuration 1s2 2s2 2p4. Oxygen has two electron pairs in the outermost shell, as well as two valence electrons available for bonding. The two electrons can form bonds with two single electrons of hydrogen atoms and would like to again form a linear shape. However, the two electrons pairs exert a strong repulsive force on the O — H bonds, distorting like to again form a linear shape. However, the two electrons pairs exert a strong repulsive force on the O — H bonds, distorting them into the bent shape of the water molecule. This shape maximizes the distance between all the electrons surrounding the central oxygen atom. That’s why water molecule is bent in shape.
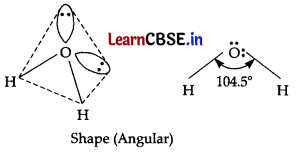
Question 21.
(a) What will be equilibrium constant for the reverse reaction
(b) At equilibrium, the concentrations of N2 = 4.2 × 10-3M, O2 = 5.3 × 10-3M and NO = 3.8 × 10-3M in a sealed vessel at 800K. What will be Kc for the reaction.
Answer:
(a) It is the inverse of the equilibrium constant for the reaction in the forward direction. For example
H2(g) + I2(g) >2HI(g)
Kc = [HI]2 / [H2] [I2]
The equilibrium constant for the reverse reaction,
2HI(g) → H2(g) + I2(g),
at the same temperature is,
K’c = [H2][I2]/[HI]2 = 1/X, = 1/Kc
Thus, K’c = \(\frac{1}{\mathrm{~K}}\)
(b) N2(g) + O2(g) → 2NO(g) Solution
For the reaction equilibrium constant, Kc can be written as

Section-C
This section contains 7 questions with internal choice in one question. The following questions are short answer type and carry 3 marks each. (No internal choice given in Board SQP)
Question 22.
20g of CaCO3 and 20 g of H2SO4 react to give CaSO4along with water and CO2.
(a) Determine the limiting reagent for the above reaction.
(b) How much CaS04 will be formed?
(c) If 1 mole of gas occupies 22.4 L at STP then calculate the volume of CO2 evolved in the above reaction. [Ca = 40, C = 12, O = 16, H = 1, S = 32]
Answer:
CaCO3 + H2SO4 → CaSO3 + H2O + CO2
(a) 1 mole CaCO3 requires 1 mole H2SO4 100 g CaC03 require 98g H2SO4
∴ 20 g CaCO3 will require \(\frac{98}{100}\) × 20
= 19.6 g H2SO4
Hence, CaCO3 is the limiting reagent.
(b) 1 mole, CaCO3 produces 1 mole CaSO4 100g CaCO3produce 136g CaSO4
20g CaCO3 will produce \(\frac{136}{100}\) × 20
= 27.2 g CaSO4
(c) 1 mole CaCO3 gives 1 mole CO2
Number of moles = mass of CO2/molecular mass of CO2
= 20/100 = 0.2 moles
∴ 0.2 moles of CaCO3 will give 0.2 moles of CO2
= 0.2 × 22.4 L
= 4.48 L of CO2
![]()
Question 23.
Identify the reagents shown in bold in the following equations as nucleophiles or electrophiles:
(a) CH3COOH + HO– → CH3COO– + H20
(b) CH3COCH3 + CN– → (CH3)3C(CN )(OH)
(c) C6 H6 + CH3CN → C6H5COCH3
Answer:
(a) HO– acts as a nucleophile as it is an electron-rich species, i.e., it is a nucleus seeking species.
(b) CN– acts as a nucleophile as it is an electron-rich species, i.e., it is a nucleus seeking species.
(c) CH3\(\stackrel{+}{\mathrm{C}} \mathrm{N}\) acts as an electrophile as it is an electron-deficient species.
Question 24.
(a) Using the VSEPR theory, identify the type of hybridization and draw the structure of OF3. What are oxidation states of O and F?
(b) MgCl2 is linear, but SnCl2 is angular. Why?
Answer:
Electron dot structure of OF2 is
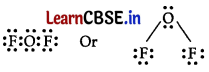
Thus, the central atom (O-atom) has 4 pairs of electrons (2 bond pairs and 2 lone pairs). Hence, oxygen in OF2 is sp3 hybridised and the molecule is V-shaped. Oxidation state of F = -1, oxidation state of O = +2.
Question 25.
Two moles of an ideal gas initially at 27°C and 1 atm pressure are compressed isothermally and reversibly till the final pressure of the gas 10 atm. Calculate the values of q, W and AU for this process.
Answer:
Given n = 2 moles
T = 27 + 273 = 300 K
Pi = 1 atm
Pf = 10 atm
∴ Work done, W = 2.303 nRT log \(\frac{p_f}{p_i}\)
= 2.303 × 2 × 8.314 JK-1 mol-1 × 300 K × log\(\frac{10}{1}\)
= 11488.28 J
For isothermal compression of ideal gas,
∆U = 0
Further, ∆U = q + W
0 = j + W .
q = -W = -11488.28 J
Question 26.
Fill up the blanks in the following table related to H20(l) → H2O(steam) at standard atmospheric pressure.
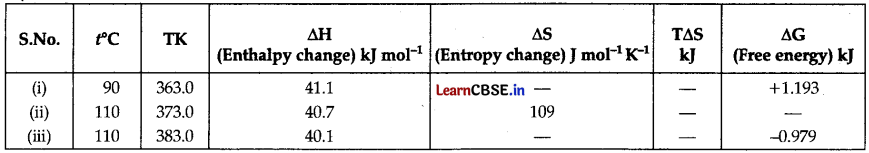
Answer:
(i) ∆G = ∆H – T∆s + 1.193
= 41.1 – 363 × ∆S
363 × ∆S = 41.1 – 1.193 = 39. 907
∴ ∆S = \(\frac{39.907}{363}\)
= 0.1099 KJ mol-1 K-1
= 110 J mol-1 K-1
(ii) T∆S = 373 × 109
= 40657 = 40.65 KJ mol-1
∆G = ∆H – T∆S
40.7 – 40.65
= -0.05 KJ mol-1
(iii) ∆G = ∆H – T∆s
– 0.979 = 40.1 – T∆s
∴ T∆s = 40.1 + 0.979
= 41.079 KJ mol-1
∆S = \(\frac{\Delta S}{T}=\frac{41 \cdot 079}{383}\)
= 0.1072 KJ mol-1 K-1
= 107.2 J mol-1 K-1.
Question 27.
Explain why?
(a) Reaction FeSO4(aq) + Cu(s) → CuSO4(aq) + Fe(s) does not occur.
(b) Zinc can displace copper from aqueous CuSO4 solution but Ag can not.
(c) Solution of AgNOa turns blue when copper rod is immersed in it.
Answer:
EθFe2+/Fe = -0.44 V
EθCu2+/Cu = +0.34 V
We know that a metal having more negative value of Ee can displace the metal having less negative or positive value of Eθfrom their salt solution. So, Fe can displace Cu from CuSO4 solution.
But, in the reaction, it is showing that Cu displaces Fe from FeSO4 solution which is not feasible.
So, the given reaction does not occur.
(b) EθZn2+/Zn = – 0.76 V
Eθcu2+/Cu = + 0.34 V
EθEAg+/Ag = + 0.80 V
Similarly, Zn has negative Eθ and Cu has positive Eθ. So, zinc can displace copper from aqueous CuSO4 solution. But silver has higher positive value of Ee than Cu, so, Ag cannot displace Cu from CuS04 solution.
(c) Cu has lesser Eθ than Ag. So, copper can displace Ag from AgNO3 solution due to which the solution turns blue.
![]()
Question 28.
Why do alkenes prefer to undergo electrophilic addition reaction while arenes prefer electrophilic substitution reaction? Explain.
Answer:
Due to the presence of a p-electron cloud above and below the plane of alkenes and arenes, these are electron rich molecules and, therefore, provide sites for the attack of electrophiles. Hence, they undergo electrophilic reactions. Alkenes undergo electrophilic addition reactions because they are unsaturated molecules.
Arenes, on the other hand, cannot undergo electrophilic addition reactions. This is because benzene has a large resonance energy of 150.4 kj mol-1. During electrophilic addition reactions, two new s-bonds are formed but the aromatic character of benzene gets destroyed and, therefore, resonance energy of benzene ring is lost. Hence, electrophilic addition reactions of arenes are not energetically favourable. Arenes, in contrast, undergo electrophilic substitution reactions in which s C – H bond is broken and new s C – X bond is formed: The aromatic character of benzene ring is not destroyed and benzene retains its resonance energy. Hence, arenes undergo electrophilic substitution reactions.
Section-D
The following questions are case -based questions. Each question has an internal choice and carries 4 (1 +1+2) marks each. Read the passage carefully and answer the questions that follow.
Question 29.
Read the following passage and answer the following questions:
James Maxwell (1870) suggested that when electrically charged particle moves under acceleration, alternating electrical and magnetic fields are produced and transmitted. These fields are transmitted in the form of waves called electromagnetic waves or electro magnetic radiation. These waves do not require any medium for their propagation and can move in vacuum. The increasing order of wavelengths of these radiations is given as below : Cosmic rays < g – rays < X – rays < Ultraviolet light < Visible light < Infrared rays < Micro waves < Radio waves.
(a) What is the relation between wavelength, wave number, frequency and velocity?
OR
The three dimensional structure of any orbital is given by which Quantum number?
(b) The radio 93.5 FM, broadcasts on a frequency of 1,370 kHz (kilo hertz). What will be the wavelength of the electromagnetic radiation emitted by transmitter. Also, predict which part of the electromagnetic spectrum does it belong to?
(c) When are Paschen and Brackett series displayed? Mention in which portion of electromagnetic spectrum they fall.
Answer:
(a) c = λ × u and 1/λ = \(\bar{v}\)
∴ u = c.\(\bar{v}\)
Explanation:
A wave has its wavelength denoted by lambda (λ) The number of waves that pass a given point in one second is the frequency denoted by neu (u). The number of wavelengths per unit length is the wave number neu bar (\(\bar{v}\)). The relation can be expressed as, when ‘c’ is the speed of electromagnetic radiation in vacuum.
OR
The three dimensional shape of orbital is given by Azimuthal quantum number
(b) The wavelength, λ, = c electromagnetic radiation /v frequency
On substituting the given values, we get λ = c/v
(3 × 108m/s)/1370 × 103/s
= 218.3 m
This value wavelength is a characteristic radio wave wavelength.
(c) When an electron transitions from a higher energy level (nh = 4, 5, 6, 7, 8, …) to an energy state with nl = 3, the Paschen series is seen. The Paschen series’ entire wavelength ranges inside the infrared portion of the electromagnetic spectrum. The Brackett series, which has the shortest wavelength, overlaps with the Paschen series.
When an electron transitions from a higher energy level (nh = 5, 6, 7, 8, 9,…) to an energy state with nl = 4, the Brackett series is seen. The electromagnetic spectrum’s infrared region encompasses the entire wavelength range of the Brackett series.
![]()
Question 30.
Read the following passage and answer the following questions:
The variables pressure, volume, concentration and temperature may change the state of Equilibrium. The change is governed by the Le-Chatelier’s principle. The decomposition of NH3(g) can be made spontaneous by increasing the temperature and lowering pressure. In the reaction, removal of any product from the reaction mixture makes the reversible reaction irreversible and therefore, reaction proceeds to completion.
(a) When will the equilibrium Solid → Liquid → Gas will shift in forward direction ?
(b) What kind of change in free energy for the equilibrium, in a gaseous reaction, PCl5 → PCl3 + Cl2takes place on addition of an inert gas at constant pressure and at constant volume?
(c) At 700 K, the equilibrium constant Kp for the reaction 2SO3(g) → 2SO2(g) + O2(g)
is(1.80 × 10-3kPa). What is the numerical value in moles per litre of Kc for this reaction at the same temperature?
OR
For the reaction, N2(g) + 3H2(g) → 2NH3(g), the partial pressures of N2 and H2 are 0.80 and 0.40 atmosphere respectively at equilibrium. The total pressure of the system is 2.80 atmosphere. What is Kp for the above reaction?
Answer:
(a) Increase in temperature will favour the forward reaction which is endothermic.
(b) Addition of inert gas at constant volume has no effect on any equilibrium reaction. When inert gas is added at constant pressure in the given reaction, it proceeds in forward direction. ∆G remains constant, but AG decreases as the reaction proceeds spontaneously in forward direction to attain equilibrium.
(c) Kp = Kc(RT)∆n
2SO3(g) → 2SO2(g) + O2(g)
∆n = np – nR
3 – 2 = 1
Kc = \(\frac{\mathrm{K}_{\mathrm{p}}}{(\mathrm{RT})^{\Delta n}}\)
Kc = \(\frac{1.80 \times 10^{-3}}{0.0821 \times 700}\)
= 3.1 × 10-5 mol L-1
Or
For the reaction,
N2(g) + 3H2(g) ⇌ 2NH3(g)
Equilibrium constant
Kp = \(\frac{\left(\mathrm{p}_{\mathrm{NH}_3}\right)^2}{\left(\mathrm{p}_{\mathrm{N}_2}\right) \times\left(\mathrm{p}_{\mathrm{H}_2}\right)^3}\)
Given,
PN2 = 0.80 atm
PN2 = 0.40 atm
Ptotal = 2.80 atm
PN2 + PN2 + PNH3 = Ptotal
0.80 + 0.40 4- PNH3 ~ 2.80
PN2 + PNH3 = 2.80 – 1.20
= 1.60 atm
∴ K2 = \(\frac{(1.60)^2}{(0.80) \times(0.40)^3}\)
= 50 atm-2
Section-E
The following questions are long answer type and carry 5 marks each. All questions have an internal choice.
Question 31.
Attempt any five of the following:
(a) What is de Broglie wavelength for an electron with light velocity?
(b) What is angular momentum of electron in 5th shell?
(c) What is the relation between probability of finding an electron and the orbital wave function?
(d) Can we apply Heisenberg’s uncertainty principle to a stationary electron? Why or why not?
(e) Write the maximum number of electron in a sub-shell with / = 3 and n = 4.
(f) Write the maximum number of electron that can be associated with the following set of quantum numbers?
n = 3, / = 1 and ml = -1
(g) Write the maximum number of electron that can be accommodated in an atom in which the highest principal
quantum number value is 4.
Answer:
(a) Mass of electron (m) = 9.1 × Kb31 kg
Velocity of light (v) = 3.0 × 108 m/s
According to de Broglie equation,
λ = \(\frac{h}{m v}\)
= \(\frac{6.626 \times 10^{-34} \mathrm{~J} \mathrm{~s}}{9.1 \times 10^{-31} \mathrm{~kg} \times 3.0 \times 10^8 \mathrm{~m} / \mathrm{s}}\)
= 0.2427 × 10-11
= 0.2427 × 10-12m
= 2.427 pm
(b) Angular momentum of electron is 5th shell
J = \(\frac{n h}{2 \pi}\)
= \(\frac{5 \times 6.626 \times 10^{-34} \mathrm{~J} \mathrm{~s}}{2 \times 3.14}\)
= \(\frac{33.13 \times 10^{-34}}{6.28}\)
= 5.275 × 10-34 kg m2/s
(c) The probability of finding an electron at a point within an atom is proportional to the square of the orbital wave function, i.e., | Ψ |2 at that point | Ψ |2.
(d) No, we cannot apply Heisenberg’s uncertainty principle to a stationary electron, because velocity = 0 and so, position cannot be measured accurately.
(e) n = 4, l = 3
∴ sub shell is 4f
for l = 3, ml = -3, -2, -1,0, +1, +2, +3.
Each orbital has maximum two electrons. So, maximum number of electron in 4f sub-shell is 14.
(f) n = 3, l = 1 and ml = -1
According to this set of quantum numbers, the outermost sub-shell is 3px.
∴ Electronic configuration is Is2 2s22p6 3s223px-1‘
Thus, maximum number of electrons is 13.
(g) n = 4
Maximum number Of electrons = 2n2 = 2(4)2 = 32
![]()
Question 32.
(a) Why is benzene extra ordinarily stable though it contains three double bonds?
(b) In the presence of peroxide, addition of HBr to prepare propene takes place according to anti Markovnikov’s rule but peroxide effect is not seen in the case of HC1 and HI. Explain.
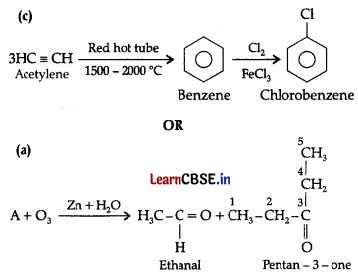
(c) How will you convert: Acetylene to chlorobenzene
OR
(a) An alkene ‘K on ozonolysis gives a mixture of ethanal and pentan-3- one. Write structure and IUPAC name of ‘A’.
(b) For the following compounds, write structural formulas and IUPAC names for all possible isomers having the number of double or triple bond as indicated:
(i) C4H8 (one double bond)
(ii) C5H8 (one triple bond)
Answer:
(a) Benzene is a hybrid of resonating structures given as:
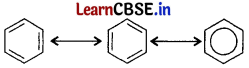
All six carbon atoms in benzene are sp2 hybridised. The two sp2 hybrid orbitals of each carbon atom overlap with the sp2 hybrid orbitals of adjacent carbon atoms to form six sigma bonds in the hexagonal plane. The remaining sp2 hybrid orbital on each carbon atom overlaps with the s-orbital of hydrogen to form six sigma C – H bonds. The remaining unhybridised p-orbital of carbon atoms has the possibility of forming three π bonds.
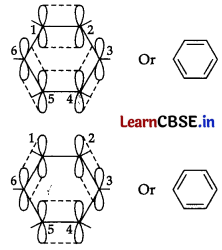
The six Jt-electrons are delocalized and can move freely about the six carbon nuclei. Even after the presence of three double bonds, these delocalized ir-electrons stabilize benzene.
(b) This may be due to the fact that the H-Cl bond being stronger (430.5 kj mol-1) than H-Br bond (363.7 kj mol-1), is not cleaved by the free radical, whereas the H-I bond is weaker (296.8 kj mol-1) and iodine free radicals combine to form iodine molecules instead of adding to the double bond.
During ozonolysis, an ozonide having a cyclic structure is formed as an intermediate which undergoes cleavage to give the final products. Ethanal and pentan-3-one are obtained from the intermediate ozonide. Hence, the expected structure of the ozonide is:
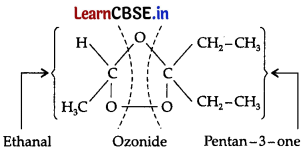
This ozonide is formed as an addition of ozone to ‘A’. The desired structure of ‘A’ can be obtained by the removal of ozone from the ozonide. Hence, the structural formula of ‘A’ is:
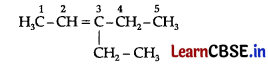
The IUPAC name of ‘A’ is 3-Ethylpent-2-ene
(b) (i) The following structural isomers are possible for C4H8 with one double bond:

The IUPAC name of
Compound (I) is But-l-ene,
Compound (II) is But-2-ene, and
Compound (III) is 2-Methylprop-l-ene.
(ii) The following structural isomers are possible for C5H8 with one triple bond:
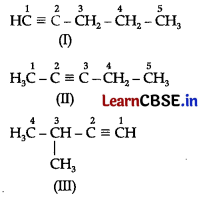
The IUPAC name of
Compound (I) is Pent-l-yne,
Compound (II) is Pent-2-yne, and
Compound (in) is 3-Methylbut-l-yne.
Question 33.
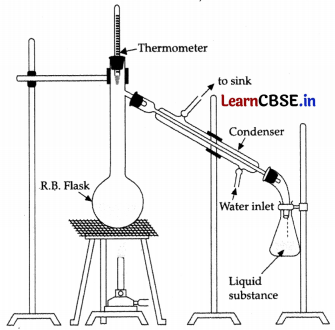
You have a mixture of three liquids A, B and C. There is a large difference in the boiling points of A and rest of the two liquids i.e., B and C. Boiling point of liquids B and C are quite close. Liquid A boils at a higher temperature than B and C and boiling point of B is lower than C. How will you separate the components of the mixture? Draw a diagram showing set up of the apparatus for the process.
OR
(a) (i) What is retardation factor?
(ii) Which elements are detected by Lassaigne’s test?
(b) (i) Show the polarization of carbon-magnesium bond in the following structure.
CH3—CH2—CH2—CH2—Mg—X
(ii) Give a brief description of the principles of one of the techniques of purification of hydrocarbons taking
an example.
(iii) Draw formulas for the first five members of each homologous series beginning with the given compound:
CH3COCH3
Answer:
difference in boiling point. Liquid B and C have boiling points very close to each other and cannot be separated by simple distillation hence, separated by fractional distillation. Liquid B is distilled first because the order of boiling points of A, B and C are as follows:
B < C < A
Or
(a) (i) The relative adsorption of each component
Rf value = Distance moved by the substance from base line (x)/Distance moved by the solvent from base line (y)
(ii) Nitrogen, sulphur, halogens and phosphorus present in organic compound are detected by ‘Lassaigne’s test
![]()
Distillation:
It is based on the fact that liquids having different boiling points vapourise at different temperatures. The vapours are then cooled and the liquids so formed are collected separately. This method is used to separate volatile liquids from non-volatile impurities or a mixture of those liquids that have a sufficient difference in their boiling points.
For example :
a mixture of chloroform (b.p = 334 K) and aniline (b.p = 457 K) can be separated by the method of distillation.
(iii) CH3COCH3 : Propanone
CH3COCH2CH3 : Butan-2-one
CH3COCH2CH2CH3 : Pentan-2-one
CH3COCH2CH2CH2CH3 : Hexan-2-one
CH3COCH2CH2CH2CH2CH3 : Heptan-2-one