Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 9 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 9 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Which of the following two combinations is correct?
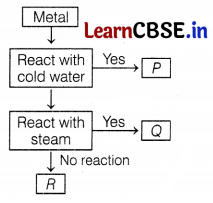
(a) P – Sodium, Q – Aluminium
(b) P – Aluminium, Q – Zinc
(c) Q – Potassium, R – Lead
(d) Q – Copper, R – Silver
Answer:
(a) P – Sodium, Q – Aluminium
Metal like potassium and sodium reacts violently with cold water.
![]()
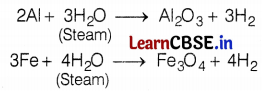
Metal like aluminium, iron, and zinc do not react either with cold or hot water. They react with steam and form the metal oxide and hydrogen gas.
Lead, copper, silver, and gold do not react with water at all.
Question 2.
The following equation of chemical reaction takes place when copper reacts with nitric acid. Identify x and y in the following reaction.
Cu + xHNO3 → Cu(NO3)2 + yNO2 + 2H2O
(a) 4 and 2
(b) 3 and 5
(c) 2 and 3
(d) 4 and 4
Answer:
(a) 4 and 2
By equating the number of atoms on both sides, the balanced equation will be
Cu + 4HNO3 → Cu(NO3)2 + 2NO2 + 2H2O
![]()
Question 3.
Chlorine forms a diatomic molecule, Cl2. The electron dot structure for this molecule is
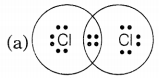
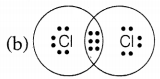
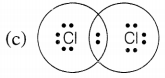
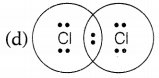
Answer:

In a chlorine molecule, both chlorine atoms contribute one electron and thus, share a single electron pair to form a single covalent bond. As electrons are shared by both atoms, they acquire the inert gas configuration of the argon atom in the valence shell.
Question 4.
A more reactive metal displaces a less reactive metal from the aqueous solution of the latter. From the following sets tell which one represents a correct picture of both the possible (✓) and impossible (✗) reactions between metals and the solutions of different salts.
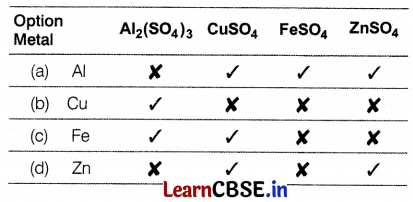
Answer:
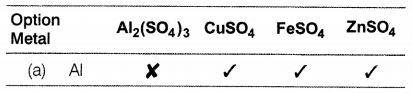
Al represents the correct picture of possible and impossible reactions. Aluminium is more reactive than copper, iron, and zinc. Hence, it displaces all of them from their salt solution whereas copper and iron are less reactive than aluminium and, therefore, give no reaction with them. Zinc being more reactive than iron, can displace iron from FeSO4 solution.
Question 5.
Which of the following are the correct structural isomers of butane?
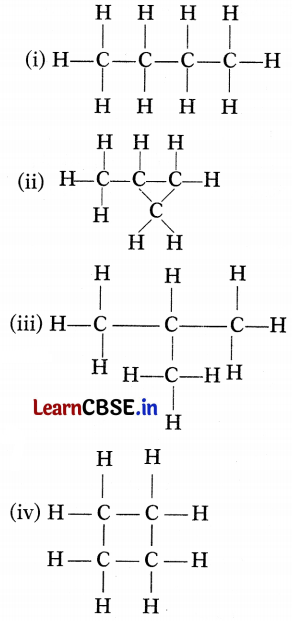
(a) (i) and (iii)
(b) (ii) and (iv)
(c) (i) and (ii)
(d) (iii) and (iv)
Answer:
(a) (i) and (iii)
Structure (i) is n-butane. Structure (iii) is iso-butane.
Since the molecular formula is the same, only the structures are different. So, (i) and (iii) are isomers while structures (ii) and (iv) have molecular formula C4H8.
Question 6.
Sarita heated the limestone and observed that a product was formed alongwith a gas. The product formed is
(a) slaked lime
(b) quick lime
(c) lime water
(d) caustic soda
Answer:
(b) quick lime
When limestone (also called calcium carbonate) is heated, then it decomposes into calcium oxide and carbon dioxide. Calcium oxide is also known as quick lime.

Question 7.
Which of the following reactions involved the combination of two elements?
(a) CaO + CO2 → CaCO3
(b) 4Na + O2 → 2Na2O
(c) SO2 + \(\frac{1}{2}\)O2 → SO3
(d) NH3 + HCl → NH4Cl
Answer:
(b) 4Na + O2 → 2Na2O
In this reaction, two elements sodium and oxygen participate and form sodium oxide. However, in other reactions, the compounds are combined and form a single product.
![]()
Question 8.
Observe the diagram of the nephron given below.
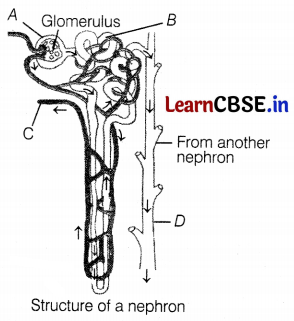
Match the labelling referred in Column I and correlate with the function in Column II.
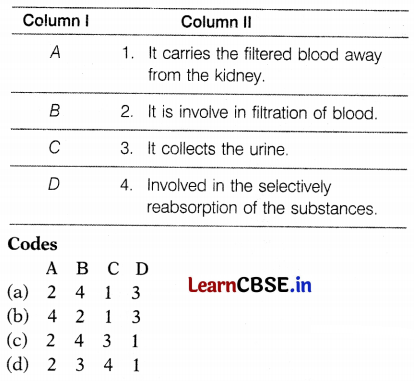
Answer:
(a) A – 2, B – 4, C – 1, D – 3
The correct labelling is as follows:
A – Bowman’s capsule
B – Tubular part of the nephron
C – Branch of renal vein
D – Collecting duct
Question 9.
Identify the phase of digestion which is represented in the diagram in Amoeba. The arrow indicates the flow of stages of the nutrition process.
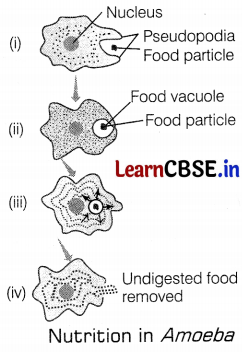
(a) The pseudopodia form the food vacuole for the egestion of undigested food
(b) The pseudopodia fuses over the food particle forming a food vacuole for the absorption of the food
(c) The pseudopodia forms the food vacuole for the excretion
(d) The pseudopodia fuses over the nucleus forming a food vacuole for the absorption of food
Answer:
(b) The pseudopodia fuses over the food particle forming a food vacuole for the absorption of the food.
Amoeba take in food using temporary finger-like extensions of the cell surface, which fuse over the food particle forming a food vacuole. Inside the vacuole, complex substances are broken down into simpler ones which then diffuse into the cytoplasm, The remaining undigested material is moved to the surface of the cell and thrown out.
Question 10.
A recessive homozygous is crossed with a heterozygous of the same gene. What will be the phenotype of the F1 generation?
(a) All dominant
(b) 75% dominant, 25% recessive
(c) 50% dominant, 50% recessive
(d) 25% dominant, 50% heterozygous, 25% recessive
Answer:
(c) 50% dominant, 50% recessive
Let’s take, the homozygous recessive as tt and heterozygous dominant as Tt. So, the cross will be
| T | t | |
| t | Tt | tt |
| t | Tt | tt |
So, the phenotype of the F.-generation is
Tt (tall) : tt (dwarf) (1 : 1)
i.e. 50% dominant and 50% recessive.
Question 11.
What is the correct sequence of components of the reflex arc?
(A) Sensory afferent nerve
(B) Relay neuron
(C) Motor nerve
(D) Receptor organ
(E) Effector, e.g. muscle
(a) D → C → B → A → E
(b) D → A → B → C → E
(c) E → B → E → A → D
(d) E → C → A → D → B
Answer:
(b) D → A → B → C → E
The sequence of events in the reflex arc is as follows:
(D) Receptor organ-like skin perceives the stimulus and activates a sensory nerve impulse.
(A) The sensory (afferent) nerve carries messages in the form of sensory impulses to the spinal cord.
(B) The neurons of the spinal cord (relay neurons) analyze sensory impulses and transmit them to motor neurons.
(C) The Motor (efferent) nerve conducts these impulses from the central nervous system to the effectors.
(E) Effector-like muscles respond by pulling back the organ away from the stimulus.
Question 12.
Identify the option that indicates the correct function or purpose served by cotyledons, plumule, and radicle respectively.
(a) Future shoot, Future root, Store food
(b) Future root, Future shoot, Store food
(c) Store food, Future shoot, Future root
(d) Future root, Store food, Future shoot
Answer:
(c) Store food, Future shoot, Future root
Cotyledons store food for seed germination. Plumule grows into a future shoot of a plant. Radicle grows into the future root of a plant.
Question 13.
No matter how far you stand from a mirror, your image appears erect. The mirror is likely to be
(a) plane
(b) concave
(c) convex
(d) either plane or convex
Answer:
(d) either plane or convex
Plane mirrors and convex mirrors always form an erect image.
Question 14.
To an astronaut the sky on the moon appears dark because
(a) there is no light on the moon
(b) there is no atmosphere on the surface of the moon
(c) moon is a non-luminous object
(d) the surface of the moon absorbs all the sunlight
Answer:
(b) there is no atmosphere on the surface of the moon
There is no atmosphere on the moon. Thus, light rays are not scattered and hence sky appears dark.
![]()
Question 15.
The main function of abscisic acid in plant is to
(a) increase the length of cells
(b) promote cell division
(c) inhibit growth
(d) promotes the growth of stems
Answer:
(c) inhibit growth
The main function of abscisic acid in plants is to inhibit growth.
Question 16.
In a flower, the parts that produce male and female gametes (green cells) are
(a) stamen and ovary
(b) filament and stigma
(c) anther and ovary
(d) stamen and style
Answer:
(c) anther and ovary
In a flower, the parts that produce male and female gametes are anther and ovary, respectively.
Direction (Q. Nos. 17-20) consists of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true, but R is false.
(d) A is false, but R is true.
Question 17.
Assertion (A): Denatured alcohol is not suitable for drinking purposes.
Reason (R): Because denatured alcohol does not same taste and smell as drinkable alcohol.
Answer:
(c) A is true, but R is false.
Denatured alcohol is not suitable for drinking purposes because it contains some poisonous substances like methanol, copper sulphate, etc. which results in coagulation of protoplasm causing nausea, blindness, and even death. Hence, A is true but R is false.
Question 18.
Assertion (A): Genes are present in every cell of an organism and control the traits of the organisms.
Reason (R): A gene is a specific segment of DNA occupying a specific position on a chromosome.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
Genes are present in every cell of organisms and control the traits of the organisms. It is a specific segment of DNA occupying a specific position on a chromosome. There is a specific gene for every protein.
Question 19.
Assertion (A): Photosynthesis is the opposite biochemical reaction of respiration.
Reason (R): Energy is utilized during respiration.
Answer:
(c) A is true, but R is false.
Respiration is defined as the process of biochemical oxidation of nutrients at the cellular level during which energy is released whereas photosynthesis is the synthesis of glucose molecules.
Question 20.
Assertion (A): A current-carrying conductor experiences a force in a magnetic field.
Reason (R): The force acting on a current-carrying conductor in a magnetic field is due to the interaction between the magnetic field produced by the conductor and the external magnetic field.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
When a current-carrying conductor is placed in a magnetic field, it experiences a force except when it is placed parallel to the magnetic field. The force acting on a current-carrying conductor depends on the magnetic field produced by the current-carrying conductor and the external magnetic field. Thus, both A and R are true, but R is not the correct explanation of A.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
Compound A oxidizes in the presence of a strong oxidizing agent to produce an acid that is present in ants. Write the formula and IUPAC names of both compounds. Give the formula and IUPAC names of the next two members of their homologous series.
Answer:
CH3OH; IUPAC name – Methanol
The general formula of an alcohol can be written as R—OH (where R is an alkyl group).
Here, CH2 and (CH2)2 are added to CH3OH, the next two members are
C2H5—OH; IUPAC name – Ethanol
C3H7—OH; IUPAC name – Propanol (1)
An acid present in ants is HCOOH; IUPAC name – Methanoic acid
The next two members are (in the same manner as before)
CH3COOH; IUPAC name – Ethanoic acid
C2H5—COOH; IUPAC name – Propanoic acid (1)
![]()
Question 22.
Name the hormones secreted by the thyroid and pituitary glands and specify one function of each.
Answer:
Thyroid gland: It secretes thyroxine hormone, which regulates the metabolism of carbohydrates, fats, and proteins in the body. Iodine is essential for the synthesis and secretion of thyroxine. (1)
Pituitary gland: It is known as the master gland of the human body as it controls the functioning and secretions of other glands.
It secretes many hormones, one of which is the Growth hormone. It regulates the growth and development of bones and muscles. (1)
Question 23.
What is the role of leaves, glomerulus, and ureter in excretion by various organisms?
Or
In birds and mammals, the left and right sides of the heart are separated. Give reasons.
Answer:
Leaves: Plants can accumulate some of their wastes in leaves. These leaves fall off and the plant gets rid of the waste. (1)
Glomerulus: In nephrons, it filters the blood passing through it.
Ureter: Transports urine from the kidney to the urinary bladder. (1)
Or
The heart of mammals and birds is four-chambered. It helps them in various ways as these are warm-blooded animals. Their metabolism is complex and the body temperature is to be maintained throughout. The separation keeps the oxygenated and deoxygenated blood away from the mixing, allowing a highly efficient supply of oxygen to the body. (2)
Question 24.
Find out from the table given below,
(a) the medium having the highest optical density.
(b) the medium with the lowest optical density.
Absolute Refractive Index of Some Material Media
| Material Medium | Refractive Index |
| Air | 1.0003 |
| Ice | 1.31 |
| Water | 1.33 |
| Alcohol | 1.36 |
| Kerosene | 1.44 |
| Canada Balsam | 1.53 |
| Rock Salt | 1.54 |
| Carbon Disulphide | 1.63 |
| Dense Flint Glass | 1.65 |
| Ruby | 1.71 |
| Fused Quartz | 1.46 |
| Turpentine Oil | 1.47 |
| Benzene | 1.50 |
| Crown Glass | 1.52 |
| Sapphire | 1.77 |
| Diamond | 2.42 |
Answer:
(a) The medium with the highest refractive index will have the highest optical density. So, the diamond has the highest optical density. (1)
(b) The medium with the lowest refractive index will have the lowest optical density. So, air has the lowest optical density. (1)
Question 25.
What are the advantages of connecting electrical appliances in parallel with the battery instead of connecting them in series?
Or
In the arrangement shown in the figure, there are two coils wound on a non-conducting cylindrical rod. Initially, the key is not inserted in the circuit. Later the key is inserted and then removed shortly after.
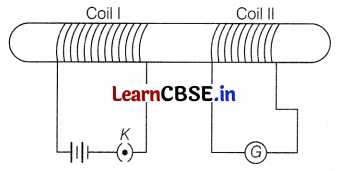
What are the two observations that can be noted from the galvanometer reading?
Answer:
The following are the advantages of connecting electrical devices in parallel with the battery:
(i) Parallel circuits divide the current among the electrical devices so that they can have the necessary amount of current to operate properly. (1)
(ii) If one of the devices in a parallel combination fuses or fails, then the other devices keep working without being affected. (1)
Or
The magnetic field lines around a straight current-carrying conductor are concentric circles whose centers lie on the wire (as shown in the figure).
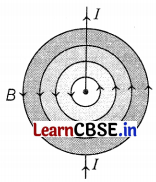
In the above figure, I and B represent the current and magnetic field, respectively. (2)
Question 26.
‘Effective segregation of wastes at the point of generation is very important’. Justify this statement.
Answer:
Effective segregation of waste is very important at the point of generation because the method of treating waste depends on the nature of waste, e.g. Biodegradable waste is used to make manure out of compost, whereas non-biodegradable waste could be recycled and reused for various purposes. Also, effective segregation of waste means that less waste goes to landfill, which makes it cheaper and better for people and the environment. (2)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
P, Q, and R are 3 elements that undergo chemical reactions according to the following equations.
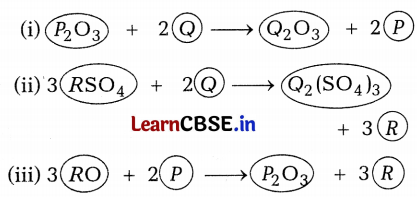
Answer the following questions.
(a) Which element is most reactive?
(b) Which element is least reactive?
(c) State the type of reaction listed above.
Answer:
(a) The most reactive element is Q as it has replaced both P and R from their compounds.
(b) Element R is least reactive as it has been replaced by both P and Q.
(c) Displacement reaction. (1 × 3)
![]()
Question 28.
What is meant by electrolytic reduction? How is sodium obtained from its molten chloride? Explain.
Or
A metal A, which is used in the thermite process, when heated with oxygen gives an oxide B, which is amphoteric. Identify A and B. Write down the reactions of oxide B with HCl and NaOH.
Answer:
In electrolytic reduction, the metals are extracted by the electrolysis of their salts. Sodium is obtained by the electrolysis of molten chlorides. The metals are deposited at the cathode (the negatively charged electrode), whereas chlorine is liberated at the anode (the positively charged electrode).
At cathode, Na++ e– → Na
At anode, 2Cl– → Cl2 + 2e–
Reaction, NaCl \(\rightleftharpoons\) Na+ + Cl– (3)
Or
Metal A is aluminium (Al) which is used in thermite reaction. Al reacts with oxygen to form aluminium oxide, Al2O3 (B), which is amphoteric.
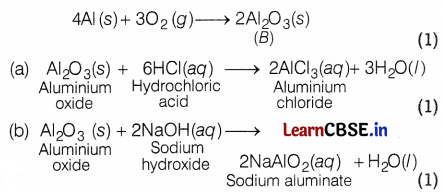
Question 29.
Mustard was growing in two fields A and B. Field A produced brown coloured seeds, while field B produced yellow-coloured seeds. It was observed that in field A, the offspring showed only the parental trait for consecutive generations, whereas in field B, the majority of the offspring showed a variation in the progeny. What are the probable reasons for these?
Answer:
In field A, the reason for parental traits in consecutive generations of the offspring is self-pollination. In self-pollination, the pollen from the stamen of a flower is transferred to the stigma of the same flower. The parent plant remains the same, so there will be no or very little variation seen. (1)
In field B, variation is seen to occur because of the recombination of genes as cross-pollination takes place. In cross-pollination, the pollen from the stamen of a flower is transferred to the stigma of a different flower. There is a difference in the genetic material of the parents, therefore, the offspring shows variation. (2)
Question 30.
What is ozone and how does it affect an ecosystem?
Answer:
Ozone is a triatomic molecule, i.e. made up of three atoms of oxygen joined together. Its molecular formula is O3. It can affect any ecosystem in the following ways:
- It protects against ultraviolet rays if, present in the stratosphere. (1)
- Ozone dissipates the energy of UV rays by undergoing dissociation followed by reassociation.
O2 \(\stackrel{U V}{\longrightarrow}\) O + O; O + O2 → O3 (Ozone) (1) - Because of its depletion, UV rays enter the atmosphere. It is highly toxic and causes injury to mucous membranes, eye irritation, and internal hemorrhages in animals and humans. (1)
Question 31.
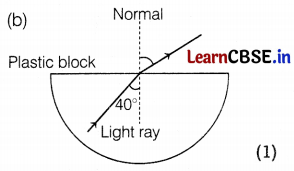
(a) In the figure below, a light ray travels from the air into the semi-circular plastic block. Give a reason why the ray does not deviate at the semi-circular boundary of the plastic block.
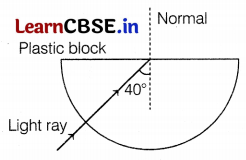
(b) Complete the ray diagram of the above scenario when the light ray comes out of the plastic block from the top flat end.
Answer:
(a) The ray of light is undergoing normal incidence at the air-plastic block interface. For normal incidence, there is no deviation. (1)
Question 32.
(a) State the relation between potential difference, work done and charge moved.
(b) Calculate the work done in moving a charge of 4 C from a point at 220 V to a point at 230 V.
Answer:
(a) The relation between potential difference, work done and charge moved is given by
Potential difference = \(\frac{Work done}{Charge moved}\)
V = \(\frac{W}{q}\) (1)
(b) Given, charge, q = 4C
Potential at point A, VA = 220 V
Potential at point B, VB = 230 V
Work done, W =?
∴ Potential difference, V = VB – VA
= 230 – 220
= 10 V
We know that
work done, W = V × q
= 10 × 4
= 40J (2)
![]()
Question 33.
A student fixed a straight thick copper wire between two terminals of a battery. He placed a magnetic compass near the current carrying copper wire. Then, he observed that the position of the compass needle changed slightly.
(a) What is the phenomenon by which the compass needle gets deflected?
(b) What does the change in the position of the compass needle indicate?
(c) If we change the direction of current through the wire, then how does the position of the compass needle change?
Answer:
(a) When a magnetic compass is placed near a current-carrying conductor, the position of the compass needle changes. This phenomenon is known as the magnetic effect of current. (1)
(b) The change in the position of the compass needle near a current-carrying conductor indicates that a magnetic field is produced around the conductor. (1)
(c) If we reverse the direction of current through the wire, then the change observed in the position of the compass needle is also in the opposite direction. (1)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
(a) Visha observed that an aldehyde as well as a ketone can Be represented by the same molecular formula, say C3H6O. Write their structures and name them. State the relation between the two in the language of science.
(b) Give two industrial uses of caustic soda.
Or
What is water crystallization? Write the common name and chemical formula of a commercially important compound that has ten water molecules as water of crystallization. How is this compound obtained? Write the chemical equation also. List any two uses of this compound.
Answer:
(a) The same molecular formula can represent an aldehyde as well as a ketone, say C3H6O.
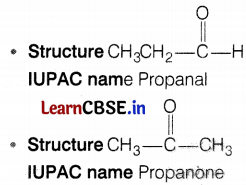
Such compounds with identical molecular formulas but different structures are called structural isomers. (2)
(b) Caustic soda (sodium hydroxide, NaOH) is used
- for mercerizing cotton fabrics in the textile industry.
- to manufacture the soap, paper, dyes, etc. (2)
- in petroleum refining.
Or
Crystals of some compounds seem to be dry but contain some water molecules attached to them. This water is called water of crystallization. The common name for a compound containing ten molecules of water of crystallization is washing soda and its chemical formula is Na2CO3. 10H2O. (2)
Preparation:
NaCl (s) + H2O (l) + CO2 (g) + NH3 (g) → NH4Cl (g) + NaHCO3 (s)
2NaHCO3 (s) \(\stackrel{Heat}{\longrightarrow}\) Na2CO3 (s) + H2O (l) + CO2 (g) ↑
Na2CO3 (s) + 10H2O (l) → Na2CO3 . 10H2O (s) (2)
Uses
- Used in glass, soap, and paper industries.
- Used for removing the permanent hardness of water. (1)
Question 35.
(a) Explain the barrier and chemical method of contraception by giving an example of each.
(b) Explain the structure of the carpel with the help of a labelled diagram.
Or
(a) Compare and contrast nervous and hormonal mechanisms for control and coordination in animals.
(b) How does chemical coordination occur in plants?
Answer:
(a) (i) Barrier method: It involves the usage of certain products or devices that prevent the meeting of gametes and help in birth control, e.g. condom, diaphragm, etc. (1)
(ii) Chemical method: It involves the usage of chemicals called spermicides, which are applied in the vagina to kill sperm. It can only be used with condoms or diaphragm (1)
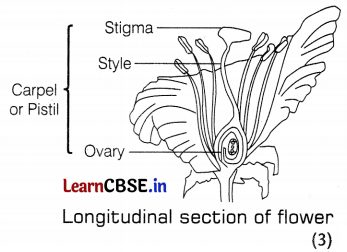
(b) Carpel or Pistil is the female reproductive part and is present in the centre of a flower. It is made up of the following three parts:
- Stigma: the sticky terminal part of the carpel which helps in receiving the pollen grains.
- Style: the middle elongated part of the carpel which helps in the attachment of stigma to the ovary.
- Ovary: the swollen bottom part of the carpel which contains ovules having an egg cell.
Or
(a) Comparison between hormonal mechanisms are as follows:
| Basis | Hormonal Mechanism/Endocrine System | Nervous Mechanism |
| Passage of information | It is through chemicals called hormones. | It is through electrical conduction. |
| Sensory Receptors | Absent | Present |
| Rapidity | The system is comparatively slower. | The system is rapid. |
| Connection | The system is not connected to target sites directly. | The system is directly connected to every part under its control. |
| Response | The response is slow and produced by all the cells of the target tissues. | The response is quick and limited to those cells that are innervated with nerves. |
| Role in growth and development | The system controls growth and development. | It has little role in growth and development. |
| Components | It consists of glands and their secretions. | It consists of neurons, nerves, and nervous organs. |
| Effects | The effect of chemical messages lasts for a longer period. | The effect of a nervous message is for a short duration. |
| Action | It is involuntary. | It can be voluntary or involuntary. |
(b) Chemical coordination in plants is achieved by the plant hormones. Plant hormones are the chemical compounds, which help the plant to coordinate the growth, development, and responses to the environment. The plant contains the following plant hormones:
- Auxins help in cell elongation.
- Gibberellins help in the growth of the stem.
- Cytokinins promote cell division.
- Abscisic acid inhibits plant growth. (2)
![]()
Question 36.
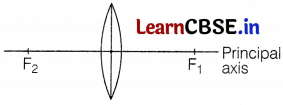
The above figure shows a thin lens with a focal length of 5 m.
(a) What is the kind of lens shown in the above figure?
(b) If a real inverted image is to be formed by this lens at a distance of 7 m from the pole, then show with calculation where should the object be placed.
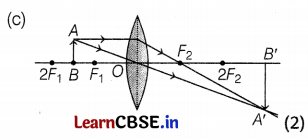
(c) Draw a neatly labelled diagram of the image formation mentioned in (b).
Or
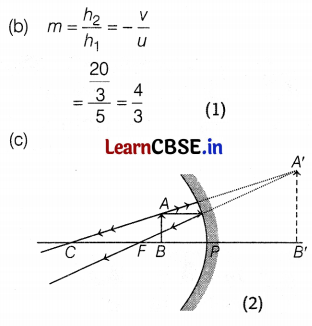
A 10 cm long pencil is placed 5 cm in front of a concave mirror having a radius of curvature of 40 cm.
(a) Determine the position of the image formed by this mirror.
(b) What is the size of the image?
(c) Draw a ray diagram to show the formation of the image as mentioned above.
Answer:
(a) Convex lens (1)
(b) Here, \(\frac{1}{f}=\frac{1}{v}-\frac{1}{u}\)
In this case, v = 7 m and f = 5 m
Putting the values in the equation we get,
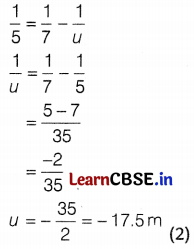
The object will be placed 17.5 m on the left of the convex lens.
Or

The image is obtained at 6.67 m behind the mirror. (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) Compound ‘A’ is widely used as a preservative in pickles. |
| (ii) ‘A reacts with ethanol to form a sweet-smelling compound ‘S’. |
Based on the above hints answer the following questions.
(a) Identify the compound A and compound B.
(b) Which gas is produced when compound ‘A’ reacts with washing soda? Write the chemical equation.
Or
How can we get the sodium salt of compound A from compound B? Name the process and write the corresponding chemical equation.
Answer:
(a) A = Ethanoic acid, B = Ester (2)
(b) CO2 gas is produced.
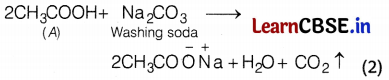
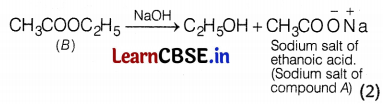
Or
The sodium salt of the compound ‘A’ can be obtained from compound ‘B’ by the action of a base. This process is called saponification.
Question 38.
Rubi crossed pure-breed pea plants having wrinkled green seeds and round yellow seeds. She found that the result obtained was an ‘AB’ type of seed. Where both alphabets denote two different traits of the plant, in the F1 generation. On further testing, she observed that when the result was self-closed, two new varieties, apart from the original ones were obtained. She named these varieties ‘AD ’ and ‘CB’.
(a) What are the phenotypes of AB seeds obtained? (1)
(b) State Mendel’s third law of inheritance. (1)
(c) Give the genotypic ratio of F2 -generation obtained. (2)
Or
What type of seeds will ‘AD’ and ‘CB’ turn as? (2)
Answer:
(a) AB-type seeds were round and yellow. (1)
(b) According to the third law, the segregation of a pair of alleles during the reduction division is independent of the segregation of the other pair of alleles. (1)
(c) The genotypic ratio obtained in F2-generation.
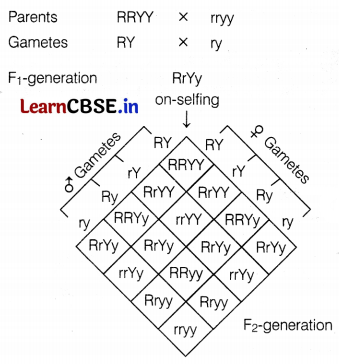
Genotypic ratio = 9 : 3 : 3 : 1
Round yellow = 9
Round green = 3
Wrinkled yellow = 3
Wrinkled green = 1 (2)
Or
AD and CB varieties will turn out to be round green and wrinkled yellow. (2)
![]()
Question 39.
A student experiments and plots the V-I graph of a sample of nichrome wire at three temperatures T1, T2, and T3 as shown in the figure.
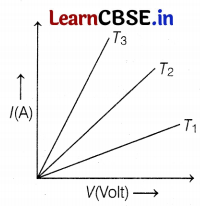
(a) Which temperature, V-I graph obeys Ohm’s law?
(b) Which temperature, the resistance of nichrome wire is minimum?
(c) What is the correct relation among temperatures T1, T2, and T3?
Or
Give two examples of the application of the heating effect of electric current.
Answer:
(a) The V-I curve of a given wire at temperatures T1, T2, and T3 obeys Ohm’s law because each V-I curve is a straight line. (1)
(b) Resistance of nichrome wire is directly proportional to temperature. Since, T1 < T2 < T3, hence, resistance of nichrome wire is minimum at temperature T1. (1)
(c) The slope of the V-I graph for the nichrome wire is directly proportional to its temperature. Hence, correct relation among T1, T2 and T3 is given as T3 > T2 > T1 (2)
Or
Electric iron and electric bulbs are two examples of the application of the heating effect of electric current. (2)