Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 8 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 8 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Consider the following figure,
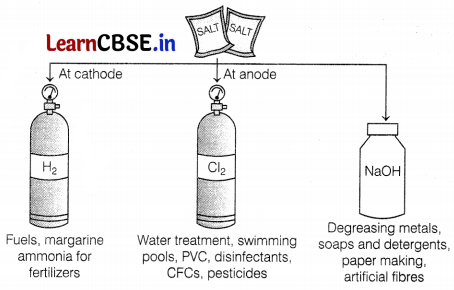
Which of the following two products are obtained from the process shown in the given figure?
(i) HCl
(ii) Bleach
(iii) Litmus
(iv) H2SO4
(a) (i) and (ii)
(b) (i) and (iii)
(c) (i) and (iv)
(d) (ii) and (iv)
Answer:
(a) (i) and (ii)
Hydrochloric acid and bleach are important products that are obtained from the chlor-alkali process.
Question 2.
Identify ‘X’ and ‘Y’ in the following reaction.
Ca(OH)2 (aq) + X (g) → CaCl2 (s) + Y + 2NH3 (g) ↑
(a) X = NH4Cl, Y = O2
(b) X = NH4Cl, Y = H2O
(c) X = (NH4)2SO4, Y = SO2
(d) X = NH4OH, Y = O2
Answer:
(b) X = NH4Cl, Y = H2O
When base Ca(OH)2 combines with ammonium salt, NH4Cl, then it liberates ammonia gas and forms calcium chloride, CaCl2, and water.
Ca(OH)2 (aq) + 2NH4Cl (g) → CaCl2 (s) + 2H2O (l) + 2NH3 (g)↑
So, X = NH4Cl and Y = H2O
![]()
Question 3.
Which of the following gives the correct increasing order of acid strength?
(a) Water < acetic acid < hydrochloric acid
(b) Water < hydrochloric acid < acetic acid
(c) Acetic acid < water < hydrochloric acid
(d) Hydrochloric acid < water < acetic acid
Answer:
(a) Water < acetic acid < hydrochloric acid
Hydrochloric acid is a mineral acid and ionizes completely in water, that’s why it is a strong acid. Acetic acid is an organic acid and ionizes only partially in the water, hence, it is a weak acid. Water is neutral. Thus, the order of acidity is water < acetic acid < hydrochloric acid.
Question 4.
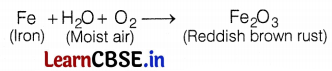
An element X on exposure to moist air turns reddish brown and a new compound Y is formed. The substance X and Y are
| X | Y | |
| (a) | Fe | Fe2O3 |
| (b) | Cu | CuO |
| (c) | Ag | Ag2S |
| (d) | Al | Al2O3 |
Answer:
(a) X = Fe, Y = Fe2O3
Iron (Fe) on exposure to moist air (H2O + O2), forms a reddish brown compound (Fe2O3) which is called rust.
Question 5.
Which of the following is not the use of graphite?
(a) It is used as a lubricant
(b) It is used in the manufacturing of lead-pencils
(c) It is used in manufacturing of artificial diamond
(d) It is used for making insulated plates
Answer:
(d) It is used for making insulated plates
Graphite can not be used for making insulated plates, as it is a good conductor of electricity.
Question 6.
Dolly added dilute sulphuric acid to iron sulphide. She observed a gas evolved which is _________
(a) hydrogen sulphide
(b) sulphur dioxide
(c) sulphur trioxide
(d) vapours of sulphuric acid
Answer:
(a) hydrogen sulphide
When dilute sulphuric acid is treated with iron sulphide, then hydrogen sulphide gas is evolved and iron sulphate is formed.
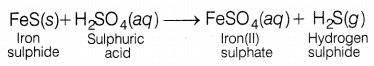
Question 7.
The structural formula of benzene is
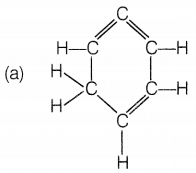
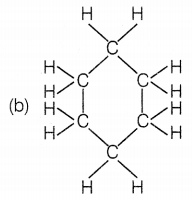
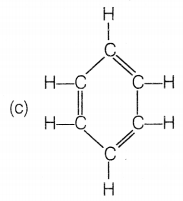
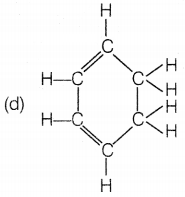
Answer:

Benzene molecule contains alternate single and double bonds. Its formula is C6H6. In structure (a), double bonds are not at alternate positions. In structure (b), the formula is C6H12 and in structure (d), the formula is C6H8.
Question 8.
Carefully study the diagram of the cross-section of the leaf with labels (i) to (iv). Select the correct option which gives correct identification and main function and/or characteristics.
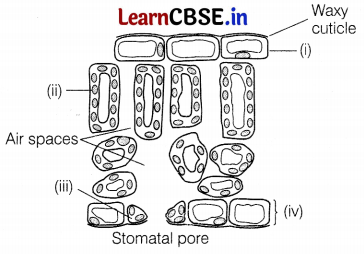
(a) (i) Upper epidermis – It contains a large number of stomata
(b) (ii) Chloroplast – Site for photosynthesis
(c) (iii) Guard cell – Protective layer of cells
(d) (iv) Lower epidermis – It has no stomata
Answer:
(b) (ii) Chloroplast – Site for photosynthesis
The correct labelling is as follows:
(i) Upper epidermis
(ii) Chloroplast
(iii) Guard cell
(iv) Lower epidermis
Chloroplasts are plant cell organelles, which are the site for the photosynthesis. It contains the photosynthetic pigment, chlorophyll that captures sunlight.
![]()
Question 9.
Identify the option that indicates the correct substrate for the enzyme that is secreted in the mouth, stomach, and small intestine respectively.
(a) Proteins, Proteins, Lipids
(b) Starch, Proteins, Lipids
(c) Starch, Lipids, Starch
(d) Starch, Lipids, Proteins
Answer:
(b) Starch, Proteins, Lipids
Mouth secretes salivary amylase which breaks down starch into sugars. Stomach secretes pepsin enzyme which breaks down proteins into smaller peptides and amino acids. In the small intestine, bile juice emulsifies fats while lipase breaks them.
Question 10.
If pea plants having round green seeds and wrinkled yellow seeds are crossed, what phenotypic ratio will be obtained in F2 progeny plants?
(a) 1 : 2 : 1
(b) 3 : 1
(c) 9 : 3 : 3 : 1
(d) 9 : 3 : 4
Answer:
(c) 9 : 3 : 3 : 1
If pea plants having round green seeds and wrinkled yellow seeds are crossed, the phenotypic ratio of 9 : 3 : 3 : 1 will be obtained in F2 progeny.
Question 11.
Select the correct option regarding the movement shown by Mimosa Pudica.
(a) The movement is non-directional and occurs due to turgor changes.
(b) The movement is an immediate response to a stimulus.
(c) The movement is in response to touch and is called xyctinasty.
(d) The movement is non-directional and involves growth.
Answer:
(a) The movement is non-directional and occurs due to turgor changes.
The leaves of Mimosa pudica responds to stimuli such as touch, blow or mechanical shack by folding their leaflets and lowering their leaves. This effect is caused by a change in the turgidity of the leaflets brought about by the movement of water into and out of the parenchymatous cells of the swollen leaf base.
Question 12.
Refer to the following figure that Represents the female reproductive system.
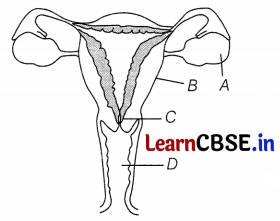
Choose the correct pair of label A, B, C, D.
(a) A – Ovary, B – Uterus, C – Cervix, D – Vagina
(b) A – Ovary, B – Fallopian tube, C – Uterus, D – Vagina
(c) A – Vagina, B – Fallopian tube, C – Uterus, D – Cervix
(d) A – Ovary, B – Cervix, C – Uterus, D – Vagina
Answer:
(a) A – Ovary, B – Uterus, C – Cervix, D – Vagina
Question 13.
Which of the following phenomena of light are involved in the formation of a rainbow?
(a) Reflection, refraction and dispersion
(b) Refraction, dispersion, and total internal reflection
(c) Refraction, dispersion, and internal reflection
(d) Dispersion, scattering, and total internal reflection
Answer:
(c) Refraction, dispersion and internal reflection
A rainbow is caused by dispersion, refraction, and internal reflection of sunlight by tiny water droplets, present in the atmosphere and always formed in a direction opposite to that of the sun. The water droplets act like small prisms. They refract and disperse the incident sunlight, then reflect it internally, and finally refract it again when it comes out of the raindrop.
Question 14.
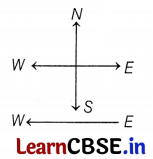
A constant current flows in a horizontal wire in the plane of the paper from East to West as shown in the given above figure. The direction of the magnetic field at a point from North to South.
(a) Directly above the wire
(b) Directly below the wire
(c) At a point located in the plane of the paper, on the north side of the wire
(d) At a point located in the plane of the paper on the south side of the wire
Answer:
(b) Directly below the wire
According to the right-hand thumb rule, when the conductor is held in the right hand, keeping the thumb from East to West as shown in the given figure, the curve of the finger will be from North to South at a point lying directly below the wire.
Question 15.
The development of a seedling from an embryo under appropriate conditions is called
(a) regeneration
(b) germination
(c) vegetative propagation
(d) pollination
Answer:
(b) germination
Germination is a process occurring in plants in which the embryo develops into a seedling under appropriate conditions.
![]()
Question 16.
Growth of the pollen tube towards the ovule during fertilization is an example of
(a) phototropism
(b) geotropism
(c) chemotropism
(d) hydrotropism
Answer:
(c) chemotropism
The growth of the pollen tube towards the ovule during fertilization is an example of chemotropism.
Direction (Q. Nos. 17-20) consists of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): Carbon shows maximum catenation property in the periodic table.
Reason (R): Carbon has a small size and thus, forms a strong C—C bond.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Catenation is the bonding of atoms of the same element into a series called a chain. Catenation occurs more readily with carbon, which forms strong covalent bonds with other C-atoms to form long chains and structures.
Question 18.
Assertion (A): All the human female gametes will have only X-chromosomes.
Reason (R): Females are homogametic with two X chromosomes.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Females are homogametic with two X chromosomes. That is why, all human female gametes will have only X-chromosomes.
Question 19.
Assertion (A): Amoeba an omnivore organisms.
Reason (R): A lion is a carnivore organism.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
Amoeba is an omnivore organism, It feeds upon both plant and animal matter. Its mode of nutrition is holozoic. Lion is a carnivore organism because it eats other animals (meat eaters).
Question 20.
Assertion (A): The magnetic field is stronger at a point that is nearer to the conductor and goes on decreasing on moving away from the conductor.
Reason (R): The magnetic field B produced by a straight current-carrying wire is inversely proportional to the distance from the wire.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
The magnitude of the magnetic field is
- directly proportional to the current I passing through the wire.
- inversely proportional to the distance r from the wire.
The magnetic field is stronger at a point that is nearer to the conductor and goes on decreasing on moving away from the conductor.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
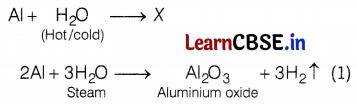
A metal taken does not react with cold as well as hot water, but it reacts with steam. Identify the metal and write the chemical equations involved.
Answer:
Aluminium only reacts with steam but does not react with hot or cold water. The chemical equations involved are as follows: (2)
Question 22.
A close coordination exists between the nerves and hormones. Discuss.
Answer:
The endocrine and neural system works in coordination for the normal functioning of our body. The synthesis and release of certain hormones is regulated by the neural system. Also, the release of certain hormones influences the activity of nerves. For example, The presence of food in our stomach distends the gastric wall. This results in the secretion of gastric hormone which stimulates gastric juice secretion in the stomach. Likewise, an increase in the concentration of adrenaline stimulates the respiratory centre of the brain. This in turn leads to an increase in the breathing rate of an individual. (2)
![]()
Question 23.
List the components and functions of transport systems in highly organized plants.
Or
Explain, why the transportation of materials is necessary for animals.
Answer:
The main components of the transport system in highly organized plants are the xylem and phloem. These can be explained as
- The xylem consists of tracheids and vessels. It conducts water and minerals (obtained from the soil) to the leaves. (1)
- Phloem consists of sieve tubes and companion cells. It helps to transport food materials, etc. from leaves to various parts of the plant. (1)
Or
The distribution of all essential substances such as food, oxygen, and water throughout the body is carried out through the system of transportation. It also displaces excretory wastes collected from the cells of the body to the excretory organs from where they are expelled from the body. Thus, transportation of materials is necessary to carry out various life processes. (2)
Question 24.
Electrical resistivities of some substances at 20°C are given as below:
| Silver | 1.60 × 10-8 Ω-m |
| Copper | 1.62 × 10-8 Ω-m |
| Tungsten | 5.2 × 10-8 Ω-m |
| Iron | 10.0 × 10-8 Ω-m |
| Mercury | 94.0 × 10-8 Ω-m |
| Nichrome | 100 × 10-6 Ω-m |
Answer the following questions using the above data:
(a) Among silver and copper, which one is a better conductor and why?
(b) Which material would you advise to be used in electrical heating devices and why?
Answer:
(a) Silver is a better conductor due to its lower resistivity. (1)
(b) Nichrome should be used in electrical heating devices due to very high resistivity. (1)
Question 25.
A convex lens forms a real and inverted image of a needle at a distance of 50 cm from it. Where is the needle placed in front of a convex lens, if the image is equal to the size of the object? Also, find the power of the lens.
Or
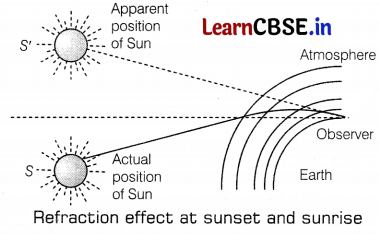
From the figure given above, explain the phenomena of advanced sunrise and delayed sunset.
Answer:
Given, image distance, v = +50 cm
Magnification, m = -1 [∵ image is inverted]
m = \(\frac{v}{u}\)
⇒ u = \(\frac{v}{m}=\frac{50}{-1}\) = -50 cm (1)
So, the needle is placed 50 cm in front of the lens.
By lens formula, \(\frac{1}{f}=\frac{1}{v}-\frac{1}{u}\)
⇒ \(\frac{1}{50}-\frac{1}{(-50)}\)
⇒ \(\frac{1}{f}=\frac{1}{25}\)
⇒ f = 25 cm = 0.25 m
∴ Power of convex lens, P = \(\frac{1}{f}=\frac{1}{0.25}\) = 4 D (1)
Advanced sunrise and delayed sunset: The figure shows the actual position of the Sun S at the time of sunrise and S’ the apparent position of the Sun. The advanced sunrise and delayed sunset are because of atmospheric refraction. The light rays starting from the Sun travel from rarer to denser layers. They bend more and more towards the normal. (1)
However, an observer on the Earth sees an object in the direction of the rays reaching his eyes. The Sun which is actually in a position below the horizon, appears in the position S’ above the horizon for him. Thus, the Sun appears to rise early by about 2 minutes and set late by about 2 minutes. This increases the length of the day by about 4 minutes. (1)
Question 26.
Carbon monoxide is dangerous when inhaled. Explain
Answer:
Carbon monoxide has a great affinity with haemoglobin in our blood. Due to this, it gets mixed with haemoglobin, almost 300 times more than oxygen. Thus, cutting off the supply of oxygen leads to less oxygen available for our body. This is why, it is dangerous when inhaled. (2)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
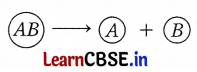
These given reactions require energy either in the form of heat light or electricity to break down the reactants. Identify and define the type of reaction. Write one equation each for this type of reaction where energy is supplied in the form of heat, light, and electricity.
Or
What is observed when sulphur dioxide is passed through (i) water and (ii) lime water? Also, write chemical equations for the reactions that take place.
Answer:
Decomposition reaction: A reaction in which a single reactant breaks down to form two or more products is known as a decomposition reaction.
(a) When a decomposition reaction is carried out by heating then it is known as a thermal decomposition reaction. (1)
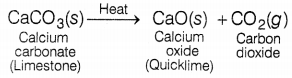
(b) A decomposition reaction in which energy is supplied in the form of light, is known as photochemical decomposition reaction. (1)
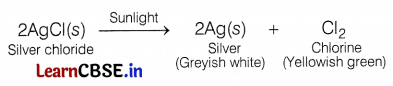
(c) A decomposition reaction in which energy is supplied in the form of electricity is known as an electrolytic decomposition reaction. (1)

Or
(a) When SO2 is passed through water, sulphurous acid is formed. Due to the formation of acid, the blue litmus is turned red. (1)
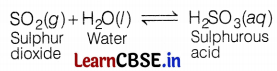
(b) When SO2 is passed through lime water, calcium sulphite (white ppt.) is formed, which reacts with excess SO2 to form calcium hydrogen sulphite. The chemical equation for the reactions are
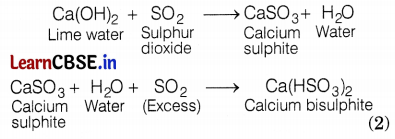
Question 28.
State the property utilized in the following.
(a) Graphite in making electrodes.
(b) Electrical wires are coated with polyvinyl chloride (PVC) or a rubber-like material.
(c) Metal alloys are used for making bells and strings of musical instruments.
Answer:
(a) Graphite is an allotrope of carbon which is a good conductor of electricity. It is cheap, insoluble in water, does not react with acids and bases, and is a non-corrosive material. Due to these properties, it is used in making electrodes. (1)
(b) Polyvinyl chloride (PVC) or a rubber-like material are insulators that do not allow electrons to flow. Hence, these are used in coating the electrical wires. (1)
(c) Metals and metal alloys are generally sonorous, i.e. they produce sound. Due to this property, they are used for making bells and strings of musical instruments. (1)
![]()
Question 29.
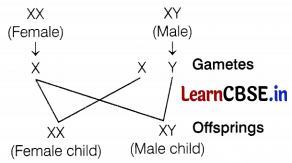
How is the sex of a child determined in human beings?
Answer:
A male germ cell that forms gametes carries one X and one Y chromosome, while a female germ cell carries two X chromosomes. Therefore, the sex of the child depends upon what happens during fertilization. (1)
(a) If a sperm carrying an X-chromosome fertilizes the egg, the child born will be a female (XX). (1/2)
(b) If a sperm carrying a Y-chromosome fertilizes the egg, the child born will be a male (XY). (1/2)
Thus, the sperm (the male gamete) determines the sex of the child. (1)
Question 30.
Two resistors X and Y of resistances 2 Ω and 3 Ω respectively are first joined in parallel and then in series. In each case, the supplied voltage is 5 V.
(a) Draw circuit diagrams to show the combination of resistors in each case.
(b) Calculate the voltage across the 3 resistors in the series combination of resistors.
Answer:
Given, X = 2 Ω, Y = 3 Ω and V = 5V
(a) Circuits are given below,
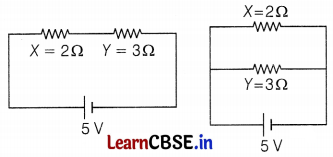
(b) Current in series combination circuit,
I = \(\frac{V}{R_{\mathrm{eq}}}=\frac{V}{X+Y}=\frac{5}{2+3}\) = 1A
Since the current in the series circuit is the same through all resistors, so potential drop across 3 Ω resistance
VY = IY = 1 × 3 = 3V (3)
Question 31.
A 6 cm tall object is placed perpendicular to the principal axis of a convex lens of focal length 25 cm. The distance of the object from the lens is 40 cm. Then, determine (a) the position (b) and the size of the image formed.
Answer:
Given, the height of the object, h0 = 6 cm
The focal length of the lens, f = 25 cm
Distance of the object, u = -40 cm

Question 32.
A student has taken two different coils of copper wire having a large number of turns. He inserts them over a non-conducting cylindrical roll. He also connected coil-1 with the battery and coil-2 with a galvanometer. Then, he observes that there is a deflection in the galvanometer needle.
(a) Why is the needle of the galvanometer deflected?
(b) What is the name of coil-1 and coil-2?
(c) Which phenomenon is involved in this activity?
Answer:
(a) Whenever the electric current through coil-1 is changing (starting or stopping), a potential difference and hence an electric current is induced in the coil-2. This induced current is responsible for the deflection of the needle of the galvanometer. (1)
(b) The coil-1 in which there is a change in electric current is called the primary coil. The coil-2 in which there is an induced current is called the secondary coil. (1)
(c) In the given activity, an electric current is produced by magnetism. This phenomenon is known as electromagnetic induction. (1)
Question 33.
Some modern insecticide has been introduced each having different properties like accumulation in the bodies of predators, broken down by soil bacteria, easily washed into lakes and rivers, and taken up by plant roots. Among all these insecticides, which one will help in reducing or keeping the level of environmental pollution to the lowest?
Answer:
Insecticides are non-biodegradable chemicals added to crop fields to stop the growth of insects infecting the crops. Modern insecticides are being developed keeping in mind, the harm they cause to the environment and its components. (1)
Biodegradable insecticides can be decomposed into harmless substances, which will subsequently be dispersed in their specific pathways and cause no pollution. Non-biodegradable insecticides build up in the fat tissues of the body and pass on to organisms that feed on them. (1)
Hence, they accumulate along the food chain resulting in significant amounts in the tissues of consumers at the highest trophic level. This is called biomagnification. The property of newly developed insecticides which includes that they can easily get decomposed into simpler components by soil bacteria, will help in reducing or keeping the level of environmental pollution to the lowest. (1)
Section D
Questions No. 34 to 36 are long answer questions.
Question 34.
(a) An organic compound A is a constituent of many medicines used as an antifreeze and has the molecular formula C2H6O. Upon reaction with alk. KMnO4, the compound A is oxidized to another compound B with the formula C2H4O2. Identify the compounds A and B. Write the chemical equation for the reaction that leads to the formation of B. Also, write the chemical equation, when A and B react with each other.
(b) Abhishek cut a lump of element X with a knife. During its reaction with water, X floats and melts. What is X? Explain.
Or
Two ores ‘X’ and Y were taken. On heating these ores, it was observed that heating of ore ‘X’ gives CO2 gas and ore ‘Y’ gives SO2 gas. Write steps to convert these ores into metals, giving chemical equations of the reactions that take place.
Answer:
(a) The organic compound A which is a constituent of many medicines and acts as antifreeze with the molecular formula C2H6O is ethanol (CH3CH2OH). Ethanol is oxidized to ethanoic acid (B) upon reaction with alk. KMnO4.

A and B react with each other to give ester, the reaction involved is a follows:
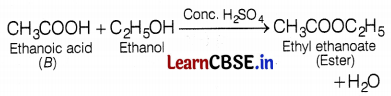
(b) X is potassium (K).
Potassium being soft can be cut with a knife and being lighter than water, it will float at the surface. The heat produced during the reaction with water will melt it. (2)
Or
(a) The ore X which gives CO2 gas is a carbonate ore.
(b) The ore Y which gives SO2 gas is a sulphide ore. (1)
The steps involved in the conversion of these ores into metals is given below:
(i) Enrichment of ores: Removal of unwanted material (gangue) from the ore is called enrichment or concentration of ore. The undesirable impurities like soil, sand, etc. are gangue or matrix. (1)
(ii) Conversion of ores into oxides
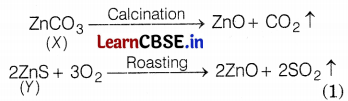
(iii) Reaction of oxide ore (smelting) is as follows.
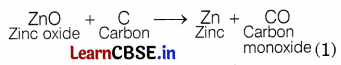
(iv) Refining: It is the process of purification of the metal obtained after reduction. Various refining methods are employed but the most common method is electrolytic refining. (1)
![]()
Question 35.
(a) Write differences between pollination and fertilization
(b) ‘Reproduction helps in providing stability to the population of a species’. Justify this statement.
Or
(a) Name the plant, different characteristics, and contrasting pairs, Mendel used for his experiments. What type of progeny was obtained by Mendel in F1 and F2 generations when he crossed the tall and short plants? Write the ratio he obtained in F2-generation plants.
(b) How do Mendel’s experiments show that traits may be dominant or recessive? Also, explain how gametes maintain their purity during reproduction.
Answer:
(a) Differences between pollination and fertilization are as follows: (\(\frac{1}{2}\) × 6)
| Pollination | Fertilisation |
| (i) It is the transfer of pollen grains from anther to the stigma of the same or different flower. | (i) It is the fusion of a male and a female gamete. |
| (ii) It carries male gamete-producing pollen grain to the female sex organs. | (ii) It leads to the formation of a zygote. |
| (iii) It is a physical process. | (iii) It is a biological process. |
| (iv) It occurs in seed plants only. | (iv) It occurs in both plants and animals. |
| (v) It is an external process. | (v) It can either be internal or external. |
| (vi) It leads to the formation of seeds. | (vi) This process leads to fertilization. |
(b) Reproduction is the process of producing new individuals of the same species by existing organisms of a species. It ensures the transfer of genetic materials from the first generation to the next generation. (1)
It is important for the continuity of the generation of an organism or species as DNA copying during reproduction helps to produce similar individuals as their parents to maintain the stability of a species. (1)
Or
(a) Mendel used the pea plant for his experiments. Seven pairs of contrasting characters in pea plants were studied by Mendel. (1)
| Character | Dominant trait | Recessive trait |
| Seed shape | Round | Wrinkled |
| Seed colour | Yellow | Green |
| Flower colour | Violet | White |
| Pod shape | Full | Constricted |
| Pod colour | Green | Yellow |
| Flower position | Axial | Terminal |
| Stem height | Tall | Dwarf |
The progeny produced from them, F1-generation plants were all tall. (1)
Then Mendel allowed F1 progeny plants to undergo self-pollination. In the F2 generation, he found that all plants were not tall, three quarters were tall and one-quarter of them were short. The ratio he obtained in F2-generation plants is 3 : 1. (1)
(b) When plants with two contrasting characters (e.g. tall and dwarf) are crossed, only one character is visible in F1-generation and the other character is suppressed. It shows the dominance of one character over another. The F1 hybrid when selfed, produced plants with both dominant and recessive phenotypes. (1)
It showed that the two unit factors of a character that remain together in an individual do not get mixed up, or get contaminated and keep their distinct identity. They separate or segregate during gamete formation. (1)
Question 36.
(a) When two ends of a metallic wire are connected across the terminals of a cell, then some potential difference is set up between its ends. In which direction electrons are flowing through the conductors?
(b) State the relation between potential difference, work done and charge moved.
(c) What is the power of a lamp, if it is connected to a 12 V battery and draws a current of 0.5 A?
Or
An electric lamp of resistance 20 Ω and a conductor of resistance 4 Ω are connected to a 6V battery as shown in the circuit given below.
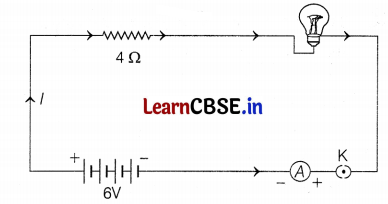
Calculate
(a) the total resistance of the circuit.
(b) the current through the circuit.
(c) the potential difference between the electric lamp and conductor.
Answer:
(a) Electrons are flowing through the conductors from their lower potential end to their higher potential end. (1)
(b) The relation between potential difference, work done and charge moved is given by
Potential difference = \(\frac{Work done}{Charge moved}\)
i.e. V = \(\frac{W}{q}\) (2)
(c) Given, V = 12V and I = 0.5A
As power, P = V × I = 12 × 0.5 = 6W (2)
Or
Given, the resistance of lamp (R1) = 20 Ω
Resistance of conductor (R2) = 4 Ω
A potential difference of battery (V) = 6V
(a) Total resistance, R = R1 + R2 = 20 + 4 = 24 Ω
(b) Current through the circuit
I = \(\frac{V}{R}=\frac{6}{24}\) = 0.25A (2)
(c) Potential difference across electric lamp = IR1
= 0.25 × 20
= 5V
The potential difference across conductor = IR2
= 0.25 × 4
= 1V (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) Substance ‘A’ is used as a refrigerant and food additive. |
| (ii) ‘A’ on reduction gives ‘B’. |
| (iii) ‘B’ is mainly used to produce ethylene. |
| (iv) ‘B’ on reaction with chlorine in the presence of sunlight gives ‘C’. |
| (v) ‘C’ is a colorless gas with a pungent odour. |
Based on the above hints answer the following questions.
(a) Name the compounds A and C.
(b) Write the chemical equation for the conversion of A to B and B to C.
Or
What is the industrial name of ‘A’? Give its industrial uses.
Answer:
(a) A – Ethene, C – Chloroethane (2)
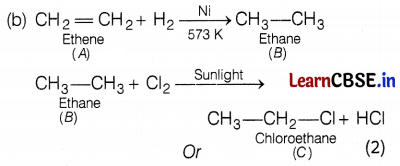
The industrial name of ethyne is acetylene.
Uses of ethyne are
(i) It is used in the manufacture of polythene.
(ii) It is used as an illuminant. (2)
![]()
Question 38.
(a) Some situations in our day-to-day life require a quick response from our body. Illustrate the sentence with the help of some examples.
(b) With the help of be labelled diagram explain the general scheme of how nerve impulses travel in the body.
Or
What is the function of receptors in our body think of a situation where receptors do not work properly. What problems are likely to arise?
Answer:
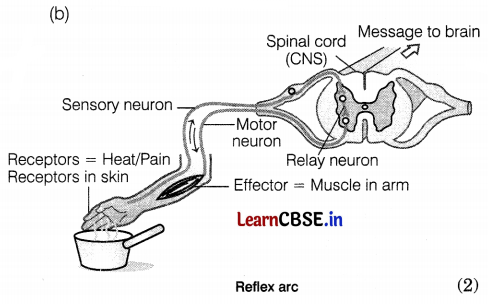
(a) Sudden quick action in response to something occurring in the environment is known as a reflex action. Reflex action is a rapid, automatic response to a stimulus, that is not under the voluntary control of the brain, i.e. it is an involuntary action. It is a simple form of behavior in which the same stimulus produces the same response every time, e.g.
- If we unknowingly touch a hot plate, we immediately move our hand away from it.
- moving our foot away on stepping something sharp.
Other examples are knee jerks, coughing, yawning, sneezing, etc. (2)
Or
The function of receptors in our body is very important as they collect information about changes in the environment around us. Receptors then pass the same information in the form of nerve impulses to the central nervous system, where the information is processed and the ultimate response is given. Now, for example, if the gustatory receptors of our tongue do not work properly, we will not be able to know the taste of different types of foods (whether it is sweet, salty, sour or bitter, etc.) (2)
Question 39.
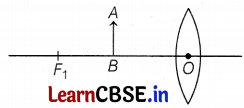
In an experiment, the formation of an image for an object AB placed in front of a convex lens is shown above in the figure, with an incomplete ray diagram.
(a) Based on text data given in the above paragraph, if the object is placed at infinity, then where is the position of the image formed by the lens?
(b) What is the nature and size of the image formed by the convex lens?
(c) For the given position of an object in the figure, where will be the image formed?
Or
If the focal length of the lens is 8 cm and an object is placed at 12 cm from the optical center, then find the position of the image formed.
Answer:
(a) The convex lens has the property to converge the parallel beam of light rays at 4 points, i.e., the focus of the lens. Hence, the image will be formed at the focus. (1)
(b) In this case, the nature of the image formed by a convex lens is virtual and erect. The size of the image is larger than that of the object, i.e. magnified image will be formed. (1)
(c) The formation of the image is as shown below:
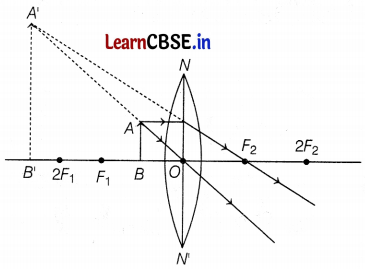
Hence, the position of the image is beyond 2F1 on the same side of the object. (2)
Or
Given, the focal length of the lens, f = 8 cm
Object distance, u = -12 cm
Using lens formula, \(\frac{1}{f}=\frac{1}{v}-\frac{1}{u}\)
⇒ \(\frac{1}{8}=\frac{1}{v}-\frac{1}{-12}\)
⇒ \(\frac{1}{v}=\frac{1}{24}\)
⇒ v = 24 cm
Hence, the image will be formed at a distance of 24 cm from the optical centre. (2)