Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 7 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 7 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
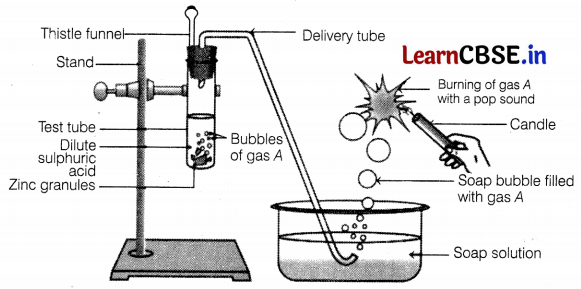
Identify gas A in the following experiment.
(a) Nitrogen
(b) Hydrogen
(c) Oxygen
(d) Carbon dioxide
Answer:
(b) Hydrogen
In the given experiment, the gas evolved (A) is hydrogen as zinc reacts with dil.H2SO4 to form zinc chloride and hydrogen gas.
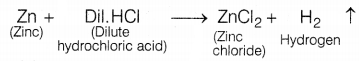
The presence of hydrogen is tested by bringing a burning candle near the soap bubbles filled with gas A (hydrogen). In doing so, hydrogen gas burns with a pop sound due to the reaction between hydrogen and oxygen present in the air.
Question 2.
Calcium phosphate is present in tooth enamel. Its nature is
(a) basic
(b) acidic
(c) neutral
(d) amphoteric
Answer:
(a) basic
Calcium phosphate Ca3(PO4)2 is a basic salt, as it is a salt of weak acid (phosphoric acid) and a slightly stronger base (calcium hydroxide).
![]()
Question 3.
A sample of soil is mixed with water and allowed to settle. The clear supernatant solution turns the pH paper yellowish-orange. Which of the following would change the colour of this pH paper to greenish-blue?
(a) Lemon juice
(b) Vinegar
(c) Common salt
(d) An antacid
Answer:
(d) An antacid
As pH paper turns greenish blue for weakly basic compounds and antacids contain weak bases like Mg(OH)2. So, an antacid would change the colour of this pH paper to greenish-blue. Other options (a) and (b) contain acids and option (c) is a neutral salt.
Question 4.
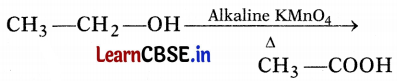
In the above-given reaction, alkaline KMnO4 acts as
(a) reducing agent
(b) oxidizing agent
(c) catalyst
(d) dehydrating agent
Answer:
(b) oxidizing agent
Alkaline KMnO4 acts as an oxidizing agent because it removes hydrogen from CH3CH2OH and adds one oxygen to it.
Question 5.
__________ will displace hydrogen from dilute acids.
(a) Copper
(b) Gold
(c) Zinc
(d) Silver
Answer:
(c) Zinc
Zinc will displace hydrogen from dilute acids as it has a higher reactivity than hydrogen.
Question 6.
Which of the following elements does not show tetravalency?
(a) Ge
(b) Si
(c) C
(d) O
Answer:
(d) O
C, Si, and Ge belong to group 14 and their valency is 4. But oxygen has electronic configuration 2, 6 and its valency is 2. Hence, it does not show tetravalency.
Question 7.
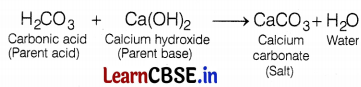
Which of the given options correctly represents the parent acid and base of calcium carbonate?
| Parent Acid | Parent Base | |
| (a) | HCl | NaOH |
| (b) | H2CO3 | Ca(OH)2 |
| (c) | H3PO3 | CaSO4 |
| (d) | H2SO4 | CaSO4 |
Answer:
(b) Parent Acid – H2CO3, Parent Base – Ca(OH)2
The terms ‘parent acid’ and ‘parent base’ are used to describe the original compounds that combine to make the salt. So, calcium carbonate salt can be obtained by the reaction between H2CO3 (parent acid) and Ca(OH)2 (parent base) to form calcium carbonate salt and water.
![]()
Question 8.
Which of the following represents the labelled parts X and Y correctly?
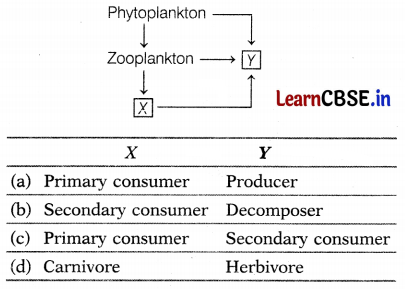
Answer:
(b) X – Secondary Consumer, Y – Decomposer
In the given food chain, X could be a small fish that feeds on zooplankton. Hence, it is a secondary consumer. Whereas Y acts on every trophic level which means it is a decomposer who acts on dead remains of living organisms.
Question 9.
Iodine is necessary for the synthesis of which hormone?
(a) Adrenaline
(b) Thyroxine
(c) Auxin
(d) Insulin
Answer:
(b) Thyroxine
Iodine is necessary for the synthesis of thyroxine hormone.
Question 10.
Identify the genotype of parent plants by observing the result of the cross given below.

(a) Both parents are homozygous
(b) Both parents are heterozygous
(c) One parent is homozygous and the other parent is heterozygous
(d) Cannot say
Answer:
(a) Both parents are homozygous
The given Punnett square represents the genotype of the F1 generation. Here, all the plants of the F1 generation are heterozygous, which means both parents are homozygous. One parent is homozygous dominant (AA), whereas the other is homozygous recessive (aa).
Question 11.
The figure given below shows three stages in the cardiac cycle.
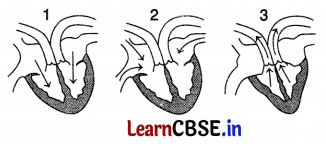
Which of the following sequences is correct regarding this
(a) 1, 2, 3
(b) 2, 1, 3
(c) 2, 3, 1
(d) 3, 1, 2
Answer:
(b) 2, 1, 3
In Figure (2), blood is entering into the right auricle through the superior and inferior vena cava and blood is entering into the left auricle through the pulmonary vein. Figure (1) shows the movement of blood from the auricles into the ventricles. Figure (3) shows the movement of blood from the right ventricle into the pulmonary artery and from the left ventricle into the aorta.
Question 12.
Which of the following represents reflex action?
(i) Beating of heart
(ii) Withdrawing your hand immediately upon touching a hot object
(iii) Riding a bicycle
(iv) Pulling leg immediately when footfalls on some sharp object
Choose the correct option
(a) (i) and (ii)
(b) (ii) and (iv)
(c) (i) and (iv)
(d) (ii) and (iii)
Answer:
(b) (ii) and (iv)
Reflex actions are rapid responses to a stimulus. E.g. Withdrawing your hand immediately upon touching a hot object and pulling up the leg immediately when your foot falls on some sharp object. Whereas, beating of the heart is an involuntary action, and riding a bicycle is a voluntary action.
![]()
Question 13.
What is the maximum resistance that can be made using five resistors each of \(\frac{1}{5}\) Ω?
(a) 1 Ω
(b) 5 Ω
(c) 2 Ω
(d) 2.5 Ω
Answer:
(a) 1 Ω
The maximum resistance is obtained when resistors are connected in a series combination. Thus, equivalent resistance,
Rs = n × R = 5 × \(\frac{1}{5}\) = 1 Ω
where, Rs = equivalent resistance for series combination. (1)
Question 14.
The angle of incidence from air to glass at point O on the hemispherical glass slab is
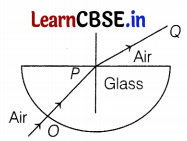
(a) 45°
(b) 0°
(c) 90°
(d) 180°
Answer:
(b) 0°
Since the light ray suffers no deviation. Thus, the angle of incidence and refraction must be equal to zero. (1)
Question 15.
During a Mendelian experiment, cross-breeding is done between tall pea plants bearing violet flowers and dwarf pea plants with white flowers. The F1 generation produced all violet flowers but half of them were short. What can be the genotype of the tall parent?
(a) TTww
(b) TTWW
(c) TtWW
(d) TtWw
Answer:
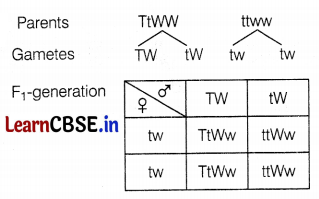
Since all the flowers are purple so tall parent is homozygous dominant for this character. Half of the plants are tall and half are dwarf, so a tall parent is heterozygous dominant for this character. Thus, the genotype of tall parent-TtWW.
Question 16.
A zygote having an X-chromosome inherited from the paternal side will be
(a) boy
(b) girl
(c) X-chromosomes from paternal side cannot determine the sex of the child
(d) either boy or girl
Answer:
(b) girl
Girl, the sex of the children is determined by chromosomes they inherit from their father. A child who inherits an X chromosome will be a girl and the one who inherits Y chromosome will be a boy.
Directions (Q.Nos. 17-20) Consist of two statements – Assertion (A) and Reason (R) Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true, but R is false.
(d) A is false, but R is true.
Question 17.
Assertion (A): Generally the candle flame is yellow.
Reason (R): The flame of a candle is yellow due to the presence of unburnt carbon particles.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
A candle flame is generally yellow due to the presence of unburnt carbon particles when light falls on these particles, they scatter a yellow colour. This shows, that the combustion of hydrocarbons in wax (candle) is not complete.
Question 18.
Assertion (A): Removal of apical bud promotes lateral bud growth.
Reason (R): The apical bud is the source of auxin.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
The apical bud is the source of auxins. It inhibits lateral bud growth. Thus, the presence of an apical bud does not allow the nearby lateral buds to grow, this is called apical dominance. So, when the apical bud is removed, the lateral bud sprouts.
![]()
Question 19.
Assertion (A): Ureter forms the common passage for both the sperm and urine.
Reason (R): It never carries sperm.
Answer:
(d) A is false, but R is true.
In males, the urethra forms the common passage for both the sperms and urine, whereas ureters are tubes – that propel urine from the kidneys to the urinary bladder. It never carries sperm.
Question 20.
Assertion (A): Myopia is due to the increased converging power of the eye lens.
Reason (R): Myopia can be corrected by using spectacles made from concave lenses.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
In myopia, due to the increased converging power of the eye lens, the image of a far object is formed in front of the retina. Myopia defects can be corrected by using a concave lens of suitable power.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
A metal forms two types of oxide and rust in moisture. Write the formulas of oxides and name the metal. Also, give the name of the metal used in the hot water apparatus.
Answer:
Iron forms two oxides iron (II) oxide and iron (III) oxide, i.e. ferrous and ferric oxide respectively.
Formula of ferrous oxide = FeO
Ferric oxide = Fe2O3
Copper is used in hot water apparatus since it is a good conductor of heat. (2)
Question 22.
Why is the progeny always tall when a tall pea plant is crossed with a short pea plant?
Answer:
The trait that represents the tallness in a pea plant is dominant over the other trait, i.e. shortness (dwarf). So progeny becomes tall when a tall pea plant is crossed with a short pea plant. (2)
Question 23.
State the functions of the following plant hormones.
(a) Abscisic acid
(b) Cytokinin
Or
Mention the correct positions of the pancreas, thyroid gland, pituitary gland, and adrenal gland in the human body. Also, mention the hormones released by them.
Answer:
(a) Function of Abscisic acid:
(i) It inhibits growth.
(ii) It causes dormancy of seeds, wilting of leaves,
(iii) Abscisic acid leads to stomatal closure. (1)
(b) Function of Cytokinin:
(i) Cytokinin promotes cell division and reduces apical dominance.
(ii) It delays aging in leaves. (1)
Or
(a) The pancreas is found just below the stomach. It secretes insulin.
(b) The thyroid gland is found just below the neck and it secretes thyroxine.
(c) The pituitary gland is the master gland. It is present at the base of the brain. It growth hormone.
(d) Adrenal glands are present on top of kidneys it secrete steroid hormones like aldosterone. (2)
Question 24.
| Name of the Appliances | Power |
| Television | 230 W |
| Light bulbs | 60 W, 100 W |
| Amplifier | 180 W |
| Refrigerator | 150 W |
| Electric iron | 750 W |
| Electric kettle | 2 kW |
| Immersion heater | 3 kW |
| Cooker | 8 kW |
From the above data:
(a) Find the current required by the cooker at 230V.
(b) What is the maximum number of 60W bulbs that can be run from the mains supply of 220 volts, if you do not want to overload a 5A fuse?
Answer:
(a) Power, P = 8 kW = 8000 W (from given table)
Voltage, V = 230 V
The current (I) required by a cooker of power 8 kW is given by
I = \(\frac{P}{V}=\frac{8000}{230}\) = 35A (1)
(b) Suppose, x numbers of bulbs can be used safely.
Power of 1 bulb = 60 W
So, power of x bulbs, P = 60 × x
The potential difference, V = 220V
P = V × I
⇒ 60 × x = 220 × 5
⇒ x = \(\frac{220 \times 5}{60}\) = 18 (1)
![]()
Question 25.
Beams of light are incident through holes A and B and emerge out of box through the holes C and D respectively, as shown in the figure.
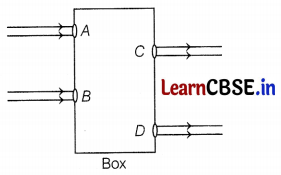
What could be inside the box?
Or
Two lamps, one rated at 100W-220V and the other at 60W-220V are connected in parallel to the electric mains supply. What current is drawn from the line, if the supply voltage is 220V?
Answer:
Here, the emergent rays are parallel to the direction of the incident ray. Therefore, a rectangular glass slab could be inside the box as the extent of bending of the light ray at the opposite parallel faces AB (air-glass interface) and CD (glass-air interface) of the rectangular glass slab are equal and opposite. This is why the ray emerges parallel to the incident ray. (2)
Or
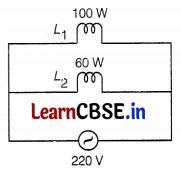
Given, potential, V = 220 V
Power, P1 = 100 W
Power, P2 = 60 W
∴ Current, I1 = \(\frac{P_1}{V}\) = \(\frac{100}{220}\) = 0.45 A
Current, I2 = \(\frac{P_2}{V}\) = \(\frac{60}{220}\) = 0.27 A
∴ Total current drawn, I = I1 + I2
= 0.45 + 0.27
= 0.72 A (2)
Question 26.
Name the organs by which sexual reproduction in floral plants takes place. What are the unisexual and bisexual flowers, explain with example.
Answer:
Stamens and carpels (pistils) are the reproductive organs of a flower, i.e. organs by which sexual reproduction in floral plants takes place. Most plants have both male and female reproductive organs in the same flower they are known as bisexual flowers, e.g. lily, rose, etc. while others have either male or female reproductive parts in a flower and are known as unisexual flowers, e.g. papaya, watermelon, etc. (2)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
Answer the following questions.
(a) State the functional group present in alcohols.
(b) Give the general formula of alcohol.
(c) What is meant by denatured alcohol?
Answer:
(a) -OH (1)
(b) CnH2n+1OH (1)
(c) To prevent the misuse of alcohol produced for industrial purposes, it is made unfit for drinking by adding poisonous substances like methanol to it. This is called denatured alcohol. (1)
Question 28.
Explain the process of electrolytic refining for copper with the help of a labeled diagram.
Or
An element A reacts with water to form compound B which is used in whitewashing. The compound B on heating forms an oxide C which on treatment with water gives back B. Identify A, B, and C and give the reactions involved.
Answer:
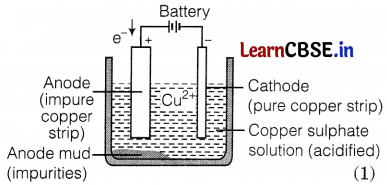
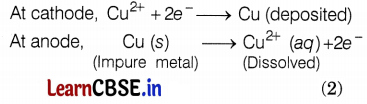
In the electrolytic process, the impure metal is made of the anode, and a thin strip of pure metal is made of the cathode. A solution of the metal salt is used as an electrolyte. On passing the current through the electrolyte, the pure metal from the anode dissolves into the electrolyte. An equivalent of pure metal from the electrolyte is deposited on the cathode.
Or
Element A is calcium (Ca), When it reacts with water, it forms calcium hydroxide. Thus, compound B is calcium hydroxide [Ca(OH)2], which is used in whitewashing.

Question 29.
Why is the flow of signals in a synapse from the axonal end of one neuron to the dendritic end of another neuron, but not the reverse?
Answer:
The synapse acts, like a one-way valve because the chemical substance is present only on one side of the gap. This chemical diffuses towards the dendrite end of the next neuron where it generates an electrical signal. Since the chemicals are absent at the dendritic end of the neuron, the nerve impulse can go across only from one side (which contains the chemical substance). In this way, it is ensured that nerve impulses travel in only one direction (through a particular set of neurons). (3)
![]()
Question 30.
Naveen wants to study the type of mirror and lens of a spherical mirror and a thin spherical lens each having a focal length of -15 cm.
(a) Specify the type of mirror.
(b) What type of lens is used?
(c) State one use of a mirror in the above activity.
Answer:
(a) For mirror, f = -15 cm
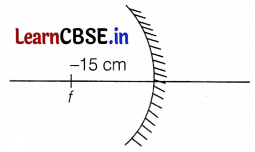
Hence, the mirror is a concave mirror according to the cartesian system. (1)
(b) For spherical lens, f = -15 cm
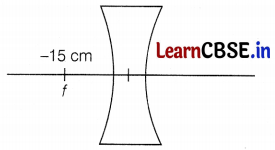
Hence, it is a concave lens. (1)
(c) The mirror used in the above activity is concave. It is used in a torch to get a powerful beam of parallel rays of light. (1)
Question 31.
Figure shows three identical bulbs A, B, and C which are connected to a battery of supply voltage V. When the switch S is closed, discuss the change in
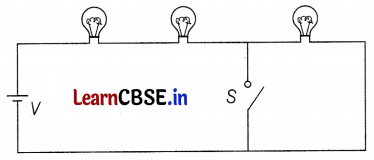
(a) the illumination of the three bulbs.
(b) the power dissipated in the circuit.
Answer:
When the switch is open,
VA = VB = VC = \(\frac{V}{3}\)
and PA = PB = PC = \(\frac{(V / 3)^2}{R}=\frac{V^2}{9 R}\) = P (say)
(a) When the switch closed, then the bulb C is short-circuited and will be no current through C, PC = 0
VA = VB = \(\frac{V}{2}\)
⇒ PA = PB = \(\frac{(N / 2)^2}{4 R}=\frac{9}{4} P\) (1\(\frac{1}{2}\))
(b) Power dissipated, P1 = PA + PB + PC = 3P
Thus, Pt = PA + PB + PC
= \(\frac{9}{4} P+\frac{9}{4} P\) + 0
= \(\frac{9}{2}\)P
= \(\frac{3}{2}\)P1 (1\(\frac{1}{2}\))
Question 32.
A student wants to study the electromagnetic induction. He has rotated a conductive coil with a machine. Then, based on his observations, answer the following questions.
(a) What kind of energy conversion takes place in the machine?
(b) In what way, the magnitude of induced current can be increased?
Answer:
(a) In this device, mechanical energy is converted into electrical energy. (1)
(b) The magnitude of induced current can be increased in the following ways.
- By increasing the speed of rotation.
- Increasing the number of turns of the conducting wire of the current carrying coil. (2)
Question 33.
How do Mendel’s experiments show that traits may be dominant or recessive?
Answer:
Mendel crossed a pure tall pea plant (TT) with a pure dwarf pea plant (tt) and observed that all the progeny were hybrid tall (Tt), i.e. only one of the traits was able to express itself in the F1 generation, which is the dominant trait. The other trait is called the recessive trait which remains suppressed.
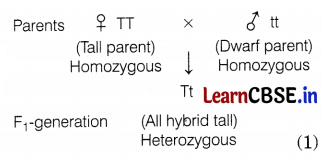
However, when he self-crossed the plants of F1 generation, he observed that one-fourth of the plants were dwarf and three-fourths were tall.
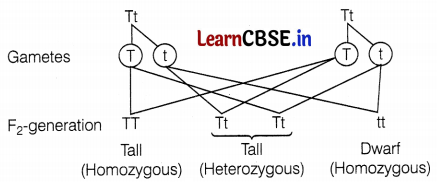
The expressed trait T for tallness is dominant, while the trait ‘t’ of dwarfness is recessive. Thus, Mendel’s experiments show that traits may be dominant or recessive. (2)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
An organic compound A of molecular formula C2H4 on reduction gives another compound B of molecular formula C26. B on reaction with chlorine in the presence of sunlight gives C of molecular formula C2H5Cl. Name the compounds A, B, and C. Also, write a chemical equation for the conversion of A to B.
Or
(a) In an experiment, vidit determines the melting point of different compounds of distinct nature. He found that the compound with an ionic nature has a maximum boiling point. State the reason why ionic compounds have high melting points.
(b) The number of carbon compounds is more than those formed by all other elements put together. Justify the statement by giving two reasons.
Answer:
The compound A of molecular formula C2H4 is an alkene. Upon reduction with hydrogen, it gives compound B of molecular formula C2H6. The compound B upon chlorination gives compound C of molecular formula C2H5Cl.
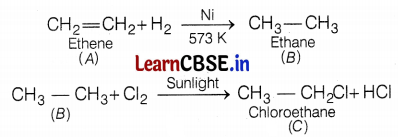
The chemical equation involved in the conversion of A to B is as follows.

Or
(a) In ionic compounds, strong electrostatic forces of attraction are present between the oppositely charged ions. When these compounds are heated, a lot of heat energy is consumed to break these strong electrostatic forces of attraction. Therefore, ionic compounds have high melting and boiling points. (2)
(b) The number of carbon compounds is more than those formed by all other elements put together because (i) carbon shows catenation and (ii) it has a valency of four. Carbon shares its four valence electrons with other atoms of carbon, this is called catenation. It has a valency of four. So, it can form bonds with four other atoms of carbon or atoms of some other monovalent element (which shows the valency of one).
![]()
Question 35.
(a) Explain the difference between androecium and gynoecium.
(b) How does reproduction help in providing stability to the population of a species?
Or
(a) What is cellular respiration? How many ATP molecules are obtained by the oxidation of one glucose atom?
(b) With the help of an experiment, show that chlorophyll is necessary for photosynthesis.
Answer:
(a) Differences between androecium and gynoecium are as follows:
| Androecium | Gynoecium |
| This is the male reproductive organ of a plant. | This is the female reproductive organ of a plant. |
| Each unit of this is called a stamen. | Each unit of this is called a carpel/pistil. |
| The terminal bloated part of the stamen is called the anther, in which male gametes or pollen grains are produced. | The lower bloated part of the carpel/pistil is called an ovary, in which the ovule is present, A Female gamete or egg is produced in the ovule. |
(b) A species occupies a well-defined niche in an ecosystem, using its ability to reproduce. During reproduction, copies of DNA pass from one generation to the next. This copying of DNA takes place with consistency in reproducing organisms and This is important for the maintenance of body design features (physiological as well as structural) which allows the organism to use that particular niche. Reproduction is, therefore, linked to the stability of the population of a species. (2\(\frac{1}{2}\))
Or
(a) In cells, the biochemical process in which glucose is oxidized in the presence of oxygen is known as aerobic or cellular respiration. During the oxidation of food, energy gets released which is stored in the form of ATP through the electron transport system. On complete oxidation, one molecule of glucose forms 38 ATP molecules. (2)
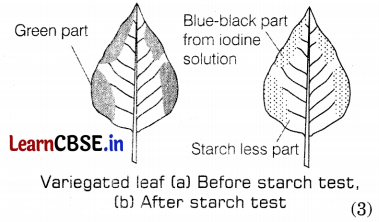
(b) Experiment to show that chlorophyll is necessary for photosynthesis: In this experiment plants having variegated leaves are selected. These plants are kept in the dark for 48 hours to make them starch-free. Then, the paint is researched by keeping it in the sun. After a few hours, the green and non-green areas are marked on a leaf. Following this, the leaf is tested for starch in which only green areas i.e. chlorophyll containing part turn blue-black due to the presence of starch. This shows that chlorophyll is essential for photosynthesis.
Question 36.
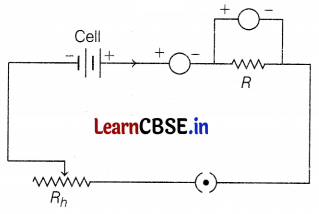
A circuit diagram is given above, answer the following questions based on the diagram.
(a) Name the device that is connected in series in the circuit and the component that controls the amount of current in the circuit.
(b) State and explain Ohm’s law.
(c) If resistance R is an electric bulb is labelled at 100W-220V. The bulb is connected to a supply of a battery 120V. Find the heat produced by the bulb in 2 min.
Or
Explain the following questions.
(a) Why is tungsten used almost exclusively for filament of electric lamps?
(b) Why are the conductors of electric heating devices such as bread toasters and electric irons made of alloys rather than pure metals?
(c) Why is the series arrangement not for used domestic circuits?
Answer:
(a) The name of the device that is connected in series in the circuit is the ammeter. It measures the current, so it is connected in series in the circuit. The rheostat is the component of the circuit that controls the amount of current in the circuit. (1)
(b) According to Ohm’s law, the electric current flowing through a conductor is directly proportional to the potential difference applied across its ends, providing the physical conditions (such as temperature) remain unchanged.
If V is the potential difference applied across the ends of a conductor through which current I flows, then according to Ohm’s law,
V ∝ I (at constant temperature)
or V = IR
or I = \(\frac{V}{R}\)
where R is the constant of proportionality called resistance of the conductor at a given temperature. (2)
(c) Given, P = 100 W and V = 220 V
∴ Resistance of bulb, R = \(\frac{V^2}{P}=\frac{220 \times 220}{100}\) = 484 Ω (1)
When the bulb is connected to a 120V supply, the heat is produced in 2 min, i.e., t = 120 s
H = \(\frac{V^2}{R} \times t=\frac{120 \times 120}{484} \times 120\) = 35702 J = 3.57 kJ (1)
![]()
Alternate Solution:
Given, power of bulb, P = 100 W and V = 220 V
Resistance of the bulb, R = \(\frac{V^2}{P}=\frac{220 \times 220}{100}\) = 484 Ω
When the bulb is connected to a 120 V supply, the current flowing through the bulb is given by
I = \(\frac{V^{\prime}}{R}=\frac{120}{484}\)
⇒ I = \(\frac{30}{121}\)A
The heat produced by the bulb in 2 min (120 s),
H = I2Rt
= \(\left(\frac{30}{121}\right)^2\) × 484 × 120
= 3570.2 J
= 3.57 kJ
Or
(a) Tungsten has a high melting point (3380°C). It does not melt at high temperatures. It retains as much of the heat generated so that it becomes very hot and emits light. That is the reason why tungsten is used as the filament of electric lamps. (1)
(b) Conductors of electric heating devices are made of alloys because alloys do not oxidize (burn) readily at high temperatures, unlike metals. Also, alloys have a greater resistivity (generally) as compared to their constituent pure metals. (2)
(c) There are 2 reasons for not using series connections for domestic circuits.
- Devices of different current ratings cannot be connected as the current is constant in a series circuit.
- If one device fails, the circuit is broken and all devices stop working. (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
Four groups of students were assigned separately to the experiment on the interaction of iron nails with a solution of copper sulphate. Each group recorded the observations as given below in the table.
| Group of Students | Initial Colour of Solution | Final Colour of Solution | Change in the Iron Nail |
| A | Blue | Colourless | Brown Coat |
| B | Green | Green | Brown Coat |
| C | Blue | Blue | Brown Coat |
| D | Blue | Light Green | Brown Coat |
(a) Which type of reaction is observed in the given experiment?
(b) Which group of students recorded all the observations correctly? Also, write the chemical equation involved.
Or
Discuss the reason behind the brown coating on iron nails.
Answer:
(a) The change in colour of the initial and final solution and the deposition of a layer on an element indicates the occurrence of displacement reaction. (2)
(b) The blue colour of copper sulphate changes into a light green colour solution due to the formation of ferrous sulphate by a displacement reaction.
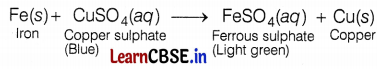
The displaced copper gets deposited on the iron nail and forms a brown coating on it. Hence, students of the D group recorded all the correct observations. (2)
Or
The brown coating on iron nails is due to copper deposition. According to the reactivity series, iron is more reactive than copper, so, it will displace copper from its sulphate solution, and then solid copper will get deposited on the iron nail. (2)
Question 38.
During an online biology lecture, Ms. Kavita demonstrated the topic food web on a PowerPoint presentation as given below. A food web is the interconnection of different food chains, which correlate at various trophic levels operating in an ecosystem.
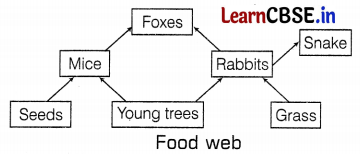
She further asked the following questions from the students
(a) How much percentage of energy is lost at each trophic level?
(b) Name the primary consumer in the given food web.
(c) How many food chains are present in the given food web?
Or
If all the foxes are killed due to a disease, what will be its impact on the food web?
Answer:
(a) About 90% of energy is lost at each trophic level. (1)
(b) The primary consumers are the organisms who directly feed on the producers. In the given food web, rabbits and mice are the primary consumers, whereas foxes and snakes are the secondary consumers. (1)
(c) 6 food chains constitute to form the given food web. These are as follows.
- Seeds → Mice → Foxes
- Young trees → Mice → Foxes
- Young trees → Rabbits → Foxes
- Grass → Rabbits → Foxes
- Grass → Rabbits → Snake
- Young trees → Rabbits → Snake (2)
Or
The foxes feed on the rabbits and mice. If all the foxes are killed then there will be no direct predator of rabbits and mice, hence the number of rabbits and mice (i.e. both are primary consumers) will increase in the given ecosystem, which will disturb the ecological balance. (2)
![]()
Question 39.
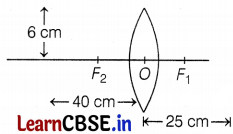
The above images show the position and height of an object in front of a convex lens. A 6 cm tall object is placed perpendicular to the principal axis of a convex lens of focal length 25 cm. The distance of the object from the lens is 40 cm.
(a) Based on the text and data in the above paragraph, determine the position of the image.
(b) The magnification produced by the convex lens.
(c) Size and nature of the image formed.
Or
Find the power of the lens.
Answer:
(a) Given, the height of the object, h0 = 6 cm
Focal length, f = 25 cm
Distance of object, u = -40 cm
Using lens formula, \(\frac{1}{f}=\frac{1}{v}-\frac{1}{u}\)
