Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 6 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 6 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based assessment units of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Identify the colour of wet blue litmus paper brought near the mouth of the delivery tube in the following experiment.
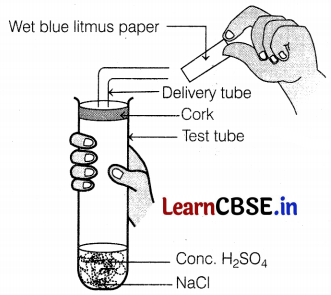
(a) The litmus paper will turn red
(b) The litmus paper will remain blue
(c) The litmus paper will turn green
(d) The litmus paper will dry
Answer:
(a) The litmus paper will turn red
The given experiment is carried out between concentrated sulphuric acid and sodium chloride, which react with each other to form HCl gas. HCl being acidic turns blue litmus red.
Question 2.
The equation given below depicts which type of chemical reaction?
A + BC → AC + B
(a) Combustion
(b) Combination
(c) Decomposition
(d) Displacement
Answer:
(d) Displacement
In a displacement reaction, one element takes the place of another element in a compound. A more reactive element displaces a less reactive element from its compounds.
A + BC → AC + B
Here, A is more reactive than 6; therefore displaces it from its compound.
![]()
Question 3.
Which of the following does not belong to the same homologous series?
(a) CH4
(b) C2H6
(c) C3H8
(d) C4H8
Answer:
(d) C4H8
C4H8 does not belong to the same homologous series because successive members of a homologous series differ by -CH2 unit.
Question 4.
Which of the following is a characteristic of metals?
(a) They have one to three valence electrons
(b) They have 4 to 8 valence electrons
(c) They are brittle
(d) They are capable of forming anions easily
Answer:
(a) They have one to three valence electrons
Metal can easily give up its electrons and form electropositive ions. They have one to three valence electrons. They are not brittle and do not form anions.
Question 5.
A girl met with an accident and her leg got fractured. She went to an orthopedics for treatment. The doctor mixed a white powder in water and applied it to her leg. What could be the white powder?
(a) Talcum powder
(b) Gypsum
(c) Plaster of Paris
(d) Copper phosphate
Answer:
(c) Plaster of Paris
Plaster of Paris reacts with water and turns into a solid hard mass known as gypsum which is used to fix fractures,
Question 6.
Aman added a few mL of cone, sulphuric acid to zinc granules. Which of the following reactions takes place when metal zinc reacts with it?
(a) Zn + 2H2SO4 → ZnSO3 + 2H2O + 2SO2
(b) Zn + 2H2SO4 → Na2ZnO2 + H2
(c) Zn + 2H2SO4 → ZnSO4 + SO2 + 2H2O
(d) Zn + H2SO4 → ZnSO4 + H2
Answer:
(d) Zn + H2SO4 → ZnSO4 + H2
Zn reacts with a strong acid (H2SO4) to form ZnSO4 (zinc sulphate) and hydrogen gas is evolved.
Question 7.
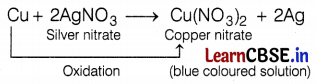
On adding some copper turnings to silver nitrate solution what will you observe?
(a) The solution turned blue
(b) Yellow precipitate was formed
(c) White precipitate was formed
(d) The solution turned red
Answer:
(a) The solution turned blue
When copper turnings are added to silver nitrate solution, a blue-colored solution is formed after some time because copper is oxidized to Cu2+ ions and forms copper nitrate. It displaces silver from its solution and forms a blue-colored solution of Cu(NO3)2.
Question 8.
In the human digestive system, different glands secrete different enzymes. The enzymes pepsin and trypsin are secreted respectively by
(a) pancreas and liver
(b) stomach and gall bladder
(c) stomach and pancreas
(d) pancreas and salivary gland
Answer:
(c) stomach and pancreas
The stomach and pancreas secrete pepsin and trypsin enzymes respectively to breakdown proteins into simpler substances.
![]()
Question 9.
Which of the following is embedded in the uterine wall?
(a) Zygote
(b) Embryo’s head
(c) Placenta
(d) Eggs
Answer:
(c) Placenta
The placenta is embedded in the uterine wall.
Question 10.
Traits are passed on from parents to their offspring. The process through which this occurs is called
(a) inheritance
(b) heredity
(c) variation
(d) evolution
Answer:
(a) inheritance
The process through which traits are passed on from parents to offspring is called inheritance.
Question 11.
Blood glucose levels are regulated in the human body by the secretion of two hormones. Which hormone is secreted when glucose concentration increases in blood?
(a) Insulin
(b) Glucagon
(c) ADH
(d) Thyroxine
Answer:
(a) Insulin
Insulin is responsible for lowering the blood glucose level whereas glucagon is opposite in function. Hence, deficiency of insulin in the body leads to diabetes mellitus.
Question 12.
Which barrier method of contraception provides the prevention of STDs along with birth control?
(a) Diaphragm
(b) Condom
(c) Intra-Uterine Contraception Device
(d) Both (a) and (c)
Answer:
(b) Condom
Condoms are the only way to protect against Sexually Transmitted Diseases (STDs) such as HIV and herpes.
Question 13.
The radius of curvature of a concave mirror is 12 cm. Then, the focal length will be
(a) 12 cm
(b) 6 cm
(c) -24 cm
(d) -6 cm
Answer:
(d) -6 cm
Given, the radius of curvature, R = 12 cm
We know that the focal length of the concave mirror has a negative value.
Hence, focal length, f = \(\frac{-R}{2}=\frac{-12}{2}\) = -6 cm
Question 14.
To an astronaut, the sky on the moon appears dark because
(a) there is no light on the moon.
(b) there is no atmosphere on the surface of the moon.
(c) moon is a non-luminous object.
(d) the surface of the moon absorbs all the sunlight.
Answer:
(b) there is no atmosphere on the surface of the moon.
There is no atmosphere on the moon. Thus, light rays are not scattered and hence sky appears dark.
Question 15.
Surgical methods of birth control include
(a) vasectomy and spermicide
(b) intra-uterine device and tubectomy
(c) vasectomy and tubectomy
(d) tubectomy and spermicide
Answer:
(c) vasectomy and tubectomy
In vasectomy (males) a small portion of vas deferens is removed or tied. In tubectomy, (females) a small portion of the fallopian tube is removed or tied.
![]()
Question 16.
The development of a seedling from an embryo under appropriate conditions is called
(a) regeneration
(b) germination
(c) vegetative propagation
(d) pollination
Answer:
(b) germination
Germination is a process occurring in plants in which the embryo develops into a seedling under appropriate conditions.
Directions (Q.Nos. 17-20) consist of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): Carbon and its compounds are used as fuel for most applications.
Reason (R): On combustion of carbon, a large amount of heat and light is released.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Carbon burns in oxygen (air) to form carbon dioxide and water. During the reaction, a large amount of heat and light is released. Hence, they are used as fuels. Carbon and its compounds keep burning without the requirement of additional energy.
![]()
Question 18.
Assertion (A): A dominant allele is an allele whose phenotype expresses even in the presence of another allele of that gene.
Reason (R): It is represented by a capital letter, e.g. T.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
The dominant allele is an allele whose phenotype will be expressed even in the presence of another allele of that gene. It is represented by a capital letter, e.g. T
Question 19.
Assertion (A): Blood clotting prevents the leakage of blood from the site of an injury.
Reason (R): Platelets circulate the body and plug these leaks.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
Platelets clump together and form a plug around the site of injury, preventing the leakage of blood from the site of an injury.
Question 20.
Assertion (A): If an electron, moving vertically from outer space, enters the earth’s magnetic field, it gets deflected towards the West.
Reason (R): Electron has a negative charge.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
The earth’s magnetic field is towards the North and the velocity of electron is downwards. By applying, Fleming’s left hand rule, the direction of force is towards West. Also, electron has a negative charge.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
When zinc metal is treated with a dilute solution of a strong acid, a gas is evolved, which is utilised in the hydrogenation of oil. Name the gas evolved. Write the chemical equation of the reaction involved and also write a test to detect the gas formed.
Answer:
When zinc reacts with dilute solution of strong acid (like hydrochloric acid HCl), it forms salt and hydrogen gas is evolved which is used in hydrogenation of oil.
Zn + 2HCl → ZnCl2 + H2 ↑
To test the presence of H2 gas, a burning splinter is brought near the mouth of the test tube, the gas burns with a pop sound. (2)
![]()
Question 22.
How do auxins promote the growth of a tendril around a support? Give examples of another plant hormones.
Answer:
Auxins promote cell elongation and are present at the shoot tip. When tendril comes in contact with a support, auxin stimulates faster growth of the cells on the opposite sides. Thus, the tendril coils around the support. The other plant hormones are cytokinins, gibberellins and abscisic acid. (2)
Question 23.
In mammals separation of oxygenated and deoxygenated blood is necessary. Why?
Or
Which blood corpuscle has haemoglobin pigment? What is its function?
Answer:
In higher animals, the energy needed is very high to maintain homeostasis because of which the requirement of O2 is very high. Thus, they need fully saturated blood with oxygen, not mixed with deoxygenated blood, so their separation is necessary. (2)
Or
A red coloured pigment called haemoglobin is present in the cytoplasm of RBC. It combines with oxygen for its transportation. (1)
The main function of RBC (Red Blood Corpuscle) is to transport oxygen from the lungs to different body parts and carbon dioxide from the tissues to the lungs. (1)
Question 24.
The values of current I flowing in a given resistor for the corresponding values of potential difference V across the resistor are as given below.
| I (Amperes) | 0.5 | 1.0 | 2.0 | 3.0 | 4.0 |
| V (Volts) | 1.6 | 3.4 | 6.7 | 10.2 | 13.2 |
Plot a graph between V and I and also calculate the resistance of that resistor.
Answer:
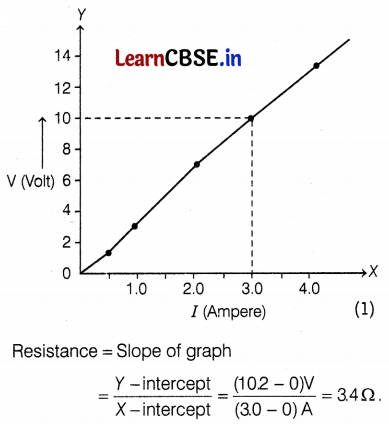
Thus, the resistance of the resistor is 3.4 Ω. (1)
Question 25.
Find the distance at which an object should be placed in front of a convex lens of focal length 10 cm to obtain an image of double its size.
Or
(a) Why does the sky appear dark instead of blue to an astronaut?
(b) Why does the sun appear reddish early in the morning?
Answer:
Given, focal length of lens, f = 10 cm
Distance of an object, u = ?
In convex lens, the image is real and virtual.
∴ Magnification, m = \(\frac{v}{u}\) = ±2
⇒ v = ±2u
Using lens formula, \(\frac{1}{f}=\frac{1}{v}-\frac{1}{u}\)
Case I: If v = +2u
\(\frac{1}{10}=\frac{1}{2 u}-\frac{1}{u}\)
⇒ \(\frac{1}{10}=\frac{1-2}{2 u}=\frac{-1}{2 u}\)
⇒ u = -5 cm
Case II: If v = -2u
\(\frac{1}{10}=\frac{1}{-2 u}-\frac{1}{u}=\frac{-1-2}{2 u}=\frac{-3}{2 u}\)
⇒ u = -15 cm (2)
Or
(a) Sky appears dark to the astronauts because there is no atmosphere and hence, no scattering of light takes place in the space. (1)
(b) At sunrise and sunset, the sun and the sky appears red. Light from the sun near the horizon passes through thicker layers of air and covers larger distance in the atmosphere before reaching our eyes. Near the horizon, most of the blue light and shorter wavelengths are scattered away by the particles. Therefore, the light that reaches our eyes is of longer wavelengths. This gives rise to the reddish appearance of the sun and the sky. (1)
![]()
Question 26.
The diagram shows the flow of energy through an ecosystem.
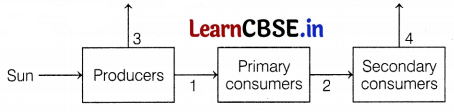
(a) Which law is depicted from this diagram.
(b) The smallest amount of energy transferred between organisms and the largest amount of energy lost to the ecosystem is represented by which arrows?
Answer:
(a) Ten Percent Law, It states that only 10% of the energy entering in a particular trophic level of organism is available for transfer to the next higher trophic level. (1)
(b) The smallest amount of energy transferred is represented by arrow 2 because as we go above in food chain, the amount of energy transformed is reduced by 10%. The largest amount of energy lost is represented by arrow 3 because plant have maximum amount of energy. Hence, loss of energy during energy transfer will also be maximum. (1)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
Study the reactions given below:
Which of the following chemical reactions will occur. Give suitable reason for each.
(a) Zn (s) + CuSO4 (aq) → ZnSO4 (aq) + Cu (s)
(b) Fe (s) + ZnSO4 (aq) → FeSO4 (aq) + Zn (s)
(c) Zn (s) + FeSO4 (aq) → ZnSO4 (aq) + Fe (s)
Answer:
(a) Zinc is more reactive than copper. So, it will displace copper from CuSO4. Hence, this reaction will take place. (1)
(b) Iron is less reactive than zinc, so it cannot displace zinc from zinc sulphate solution. The reaction will not take place. (1)
(c) Zinc is more reactive than iron, it will displace iron from FeSO4 solution. Hence, this reaction will take place. (1)
Question 28.
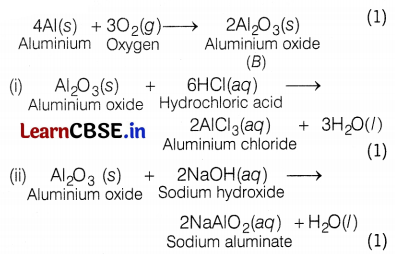
A metal X, which is used in the thermite process, when heated with oxygen gives an oxide Y which is amphoteric. Identify X and Y. Write balanced chemical equations of the reactions of oxide Y with hydrochloric acid and sodium hydroxide.
Or
What is meant by electrolytic reduction? How is sodium obtained from its molten chloride? Explain.
Answer:
28. Since, the metal ‘X’ is used in the thermite process, therefore,
‘X’ is aluminium and Y is aluminium oxide, Al2O3 (amphoteric).
A balanced chemical equation is as follows:
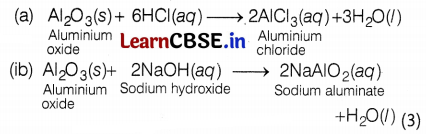
Or
In electrolytic reduction, the metals are extracted by the electrolysis of their salts. Sodium is obtained by the electrolysis of molten chlorides. Sodium metal is deposited at the cathode (the negatively charged electrode), whereas chlorine is liberated at the anode (the positively charged electrode).
At cathode, Na+ + e– → Na
At anode, 2Cl– → Cl2 + 2e–
Overall reaction, 2NaCl → 2Na+ + Cl2 (3)
![]()
Question 29.
‘A trait may be inherited, but may not be expressed’. Justify this statement with the help of a suitable example.
Answer:
Let us take the following example to justify the given statement. Mendel crossed tall pea plants with dwarf pea plants.
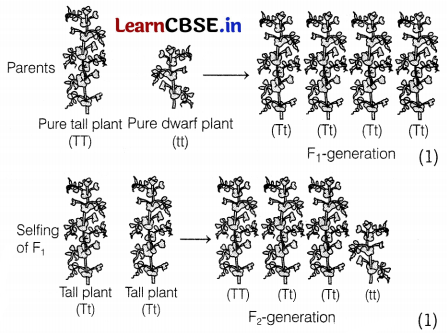
Mendel’s observation F1 generation contained all tall plants with genotype Tt, where T represents a dominant trait and ‘t’ represents a recessive trait. When Frgeneration under went selfing, the trait that was unexpressed in F1 (dwarf) was observed in some F2-progeny. Thus, both traits, tall and dwarf, were expressed in F2-generation in the ratio of 3 : 1. (1)
Question 30.
Rohan wants to have an emergent ray parallel to an incident ray on refraction through a glass slab.
(a) Draw a ray diagram to show the refraction of light through a glass slab and mark an angle of refraction and the lateral shift suffered by the ray of light while passing through the slab.
(b) In refraction of light through a rectangular glass slab, the emergent ray is parallel to the direction of the incident ray. Why?
(c) A ray of light normally on the surface of a transparent glass slab. Draw a ray diagram to show its path.
Answer:
(a) Ray diagram of refraction through a glass slab.
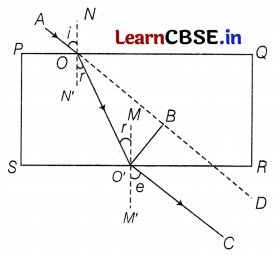
In the above figure,
∠N’OO’ = ∠r = angle of refraction
∠MO’O = ∠r = angle of refraction
Distance, O’B = lateral displacement or shift (1)
(b) Since the extent of bending of the ray of light after refraction at the opposite faces of the rectangular glass slab is equal and opposite, so emergent ray is parallel to the incident ray. (1)
(c) If the ray of light falls normally on the surface of the glass slab, it will not refract or deviate, and thus, ∠i = ∠e = 0°
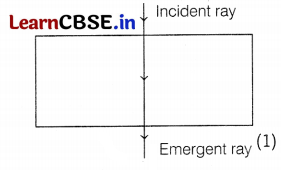
Question 31.
Three 2 Ω resistors, A, B, and C are connected as shown in the figure. Each of them dissipates energy and can withstand a maximum power of 18 W without melting.
(a) Find the maximum current that can flow through the resistor A.
(b) Find the maximum current that can flow through resistors B and C.
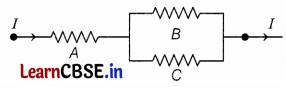
Answer:
(a) Given, resistance, R = 2 Ω
Maximum power, Pmax = 18W
Maximum current, Imax =?
As we know, P = I2R
⇒ I = \(\sqrt{\frac{P}{R}}=\sqrt{\frac{18}{2}}\) = 3A = Imax
The maximum current that can flow through resistor A is 3A. This current divides along B and C because they are in parallel combinations.
(b) Voltage across B and C remain the same and hence I ∝ \(\frac{1}{R}\)
Since B and C have the same resistance same current flows through them.
i.e. \(\frac{3}{2}\) = 1.5A flows through B and C.
Question 32.
A magnetic compass shows a deflection when placed near a current-carrying wire.
(a) How will the deflection of the compass needle be affected, if the current in the wire is increased?
(b) How will the direction of deflection of the compass needle be affected if the direction of current through the wire is reversed?
Answer:
(a) If the current in the wire is increased, then the deflection increases. The strength of the magnetic field is directly proportional to the magnitude of current passing through the straight conductor. (1\(\frac{1}{2}\))
(b) If the direction of current through the circular wire is reversed, then the direction of deflection of the compass needle will be also reversed in the opposite direction. (1\(\frac{1}{2}\))
![]()
Question 33.
Why should biodegradable and non-biodegradable wastes be discarded in two separate dustbins? Also, mention about 3R’s.
Answer:
3R is an initiative that can be used to minimize and manage waste generation, usage, and disposal. Biodegradable materials are broken down by microorganisms in nature into simple harmless substances. Non-biodegradable materials need a different treatments like heat and temperature for disposal and hence, both should be discarded in two different dustbins. (1)
The 3 R’s are as follows.
- Reduce: This means to cut back on the amount of trash we generate.
- Reuse: It means to find new ways to use things that otherwise would have been thrown out.
- Recycle: This means to turn something old and useless like plastic into something new and useful (like picnic benches, to make roads, etc.) (2)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
(a) Combustion of coke is a combination reaction. CO2 is not a pollutant. Then why is the combustion of coke harmful?
(b) Sneha observed that a metal A, which is used in the thermite process, when heated with oxygen gives an oxide B, which is amphoteric. Identify A and B. Write down the reactions of oxide B with HCl and NaOH.
Or
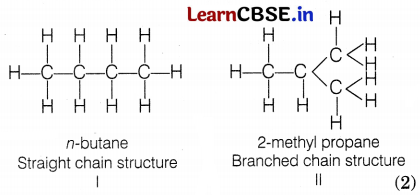
What is a structural isomer? Also, draw the isomeric structures of butane. Compare the structure of benzene and cyclohexane by drawing them.
Answer:
(a) CO2 is not a pollutant as it is present in the atmosphere upto a certain percent. However, it helps to maintain the temperature of the Earth. Combustion of coke is harmful as it increases the concentration of CO2 in the atmosphere which causes global warming (greenhouse effect). (2)
(b) Metal A is aluminium (Al) which is used in a thermite reaction. Al reacts with oxygen to form aluminium oxide, Al2O3 (B), which is amphoteric. (1)
Or
Carbon compounds having the same molecular formula but different structural formulas are called structural isomers. (1)
e.g. Butane (C4H10) shows the following two structural isomers.
Structures of benzene and cyclohexane are as follows:
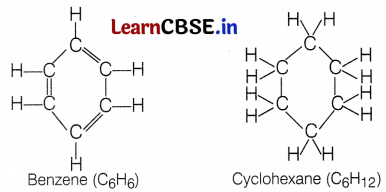
Benzene (C6H6) has six C-atoms and six H-atoms, it contains three double bonds alternately between two C-atoms. Cyclohexane (C6H12) has six C-atoms each possessing two H-atoms, thus twelve H-atoms in total. It does not consist of any double bond. (2)
Question 35.
(a) Why does menstruation occur?
(b) Sexual act always has the potential to result in pregnancy. What are the various ways to avoid pregnancy? Elaborate on any one method.
Or
Mention the functions of the following.
(a) Medulla oblongata
(b) Olfactory lobes
(c) Cerebellum
(d) Hypothalamus
(e) Cerebrum
Answer:
(a) Menstruation occurs when the egg is not fertilized. Every month, the uterus prepares itself to receive a fertilized egg. In case, the egg is not fertilized, this lining breaks and discharges out from the body through the vagina in the form of blood. This is called menstruation. (2)
(b) Ways to avoid pregnancy are called contraceptive methods. It includes
- Mechanical barrier
- Drugs (pills)
- IUCD, e.g. copper-T
- Surgical method for permanent contraception. (2)
Condoms: It is a fine rubber balloon-like structure worn over the penis during sexual intercourse. Semen is collected in it and not discharged into the vagina. This method also prevents the spread of STDs such as AIDS. (1)
Or
The functions of different parts of the brain are
- Medulla oblongata: It controls involuntary actions and regulates reflex responses. It also controls blood pressure, salivation, and vomiting.
- Cerebellum: It controls and coordinates different muscular actions. It is responsible for voluntary actions and maintains the equilibrium of the body during walking, drinking, catching, etc.
- Cerebrum: performs thinking, reasoning, speech, intelligence, and usage of information.
- Olfactory lobes are responsible for detecting smell from different receptors.
- The hypothalamus controls body temperature, urge to eat, drink, etc.
![]()
Question 36.
Neha decorates the window of her room using three identical bulbs B1, B2, and B3 as shown in the figure. When all three bulbs glow, the current of 3A is recorded by ammeter A.
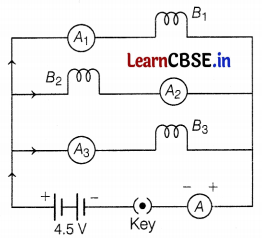
(a) If the resistance of each bulb is 4.5 Ω, then find the equivalent resistance of the combination of bulbs.
(b) Draw the above diagram with a voltmeter connected to the circuit.
(c) If bulb B2 gets fused, then what will be the total current in the circuit?
Or
(a) Define power and state its SI unit.
(b) A torch bulb is rated 5V and 500 mA. Calculate its power and resistances.
(c) Find the energy consumed when a torch of power 2.5 W is lighted for 2\(\frac{1}{2}\) hours.
Answer:
(a) Since all the bulbs are connected in parallel combination
∴ \(\frac{1}{R_{\text {eq }}}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\)
= \(\frac{1}{4.5}+\frac{1}{4.5}+\frac{1}{4.5}\)
= \(\frac{3}{4.5}\)
∴ Req = \(\frac{4.5}{3}\) = 1.5 Ω (1)
(b) As voltmeter reads potential difference across the resistor. So, it is connected in parallel with all the resistors. The ammeter is always connected in series. The circuit diagram connected with the voltmeter is shown in the figure below.
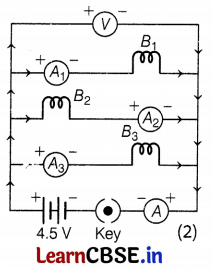
(c) When bulb B2 gets fused, then the current in bulb B2 will become zero and the current in bulb B1 and B2 will remain the same.
∴ Total current, I = I1 + I2 + I3
= 1A + 0 + 1A
= 2A (2)
Or
(a) The rate of doing work. It is equivalent to the amount of energy consumed per unit time. The unit of power is the watt (W). When a body does work at the rate of 1 joule per second, its power is 1 watts. (1)
(b) Given, voltage rating, V = 5V and current rating I = 500 mA
As we know power of a bulb
P = VI
= 5 × 500 × 10-3 [∵ A = 10 mA-3]
= 2.5 W
= 2.5 × 10-3 kW
Resistance of bulb,
R = \(\frac{V}{I}\) (ohm’s law)
⇒ R = \(\frac{5}{500 \times 10^{-3}}\) = 10 Ω (2)
(c) Energy consumed in 2\(\frac{1}{2}\) hour
E = P.t
= \(\frac{2.5 \times 2.5}{1000}\) [2\(\frac{1}{2}\) h = 2.5 h]
= \(\frac{6.25}{1000}\)
= 0.00625 kWh (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) ‘A’ is an organic compound, containing 2 carbon atoms. |
| (ii) Compound ‘A’ is used as a solvent. |
| (iii) ‘A’ on heating with conc. H2SO4 forms a compound ‘B’. |
| (iv) ‘B’ on the addition of one mole of hydrogen in the presence of ‘Ni’ forms a compound ‘C’. |
| (v) One mole of ‘C’ on heating forms one mole of CO2 and three moles of H2O. |
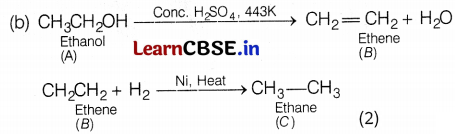
Based on the above hints answer the following questions.
(a) Give the IUPAC name of A and B.
(b) Write the chemical equation of the reactions involved. (A → B and B → C).
Or
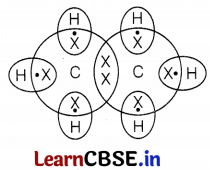
Name the chemical reaction which occurs in steps 3 and 5. Draw the electron dot structure of compound C.
Answer:
(a) A = Ethanol, B = Ethene (2)
Or
The chemical reaction that occurs in step (iii) is called dehydration and in step (v) is the combustion reaction.
Dehydration, Combustion.
Electron dot structure of ethane (C2H6)
Question 38.
Sarita experimented on two plant varieties, one with red petals and the other with white petals. When these two varieties are cross-pollinated, all the offspring have red petals.
(a) Which gene will be the dominant?
(b) Choose a suitable letter to represent the two genes.
(c) If the F1-generation self-pollinated, a total of 2400 plants were produced.
How many of these would be having red and white petals? Give the genotype of F2-generation.
Or
When F1-generation was cross-pollinated with plants having rr genes a total of 2400 plants were produced. How many of these would be having red and white petals? Give the phenotype of F2-generation.
Answer:
(a)
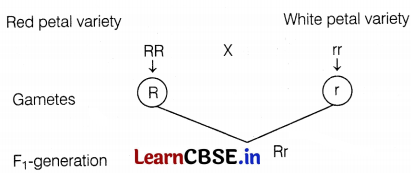
The red colour gene expresses itself in the presence of a white petal variety. Therefore, a red colour gene will be dominant. (1)
(b) R for red and r for white petals. (1)
(c)
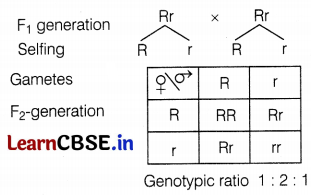
If F1-generation is self-pollinated, then the genotypic ratio of F2-generation will be 1 : 2 : 1 (RR : Rr : rr). So, out of 2400 plants, 600 (rr) will have white petals and 1800 (RR, Rr) will have red petals. (2)
Or
If F1-generation is cross-pollinated with rr, then the Phenotypic ratio of F2-generation will be Rr : rr, 1 : 1. So, out of 2400 plants, 1200 (Rr) will have white petals and 1200 (rr) will have red petals. (2)
![]()
Question 39.
The refractive index of a medium concerning vacuum is called the absolute refractive index of the medium. It is given by, µ = \(\frac{\sin i}{\sin r}\)
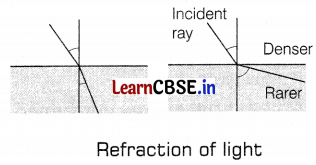
Absolute refractive indices of some of the materials A, B, C, and D are given in the following table.
| Medium | Refractive Index |
| A | 1.54 |
| B | 1.33 |
| C | 2.42 |
| D | 1.65 |
(a) How is the absolute refractive index related to the speed of light?
(b) In which of the material given in the above table, light travels fastest?
(c) The speed of light in air is 3 × 108 ms-1 and that in medium X is 2.5 × 108 ms-1. Then, find the refractive index of medium X.
Or
If the refractive index of P concerning Q is 2. Then, find the refractive index of Q concerning P.
Answer:
(a) Absolute refractive index (µ)

(b) Since B has the least refractive index, it indicates that B is much optically rarer than all other mediums, hence light travels fastest in medium B. (1)
(c) Refractive index of X

Or
Given, QµP = 2
∴ The refractive index of Q w.r.t, P is,
\(P \mu_Q=\frac{1}{Q \mu_P}=\frac{1}{2}=0.5\) (2)