Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 5 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 5 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based assessment units of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
The given experiment shows the reaction between the iron nail and copper sulphate. It produces a green colour solution. The products obtained are

(a) Ferric sulphate, copper
(b) Cuprous sulphate, iron
(c) Iron sulphate, cupric sulphate
(d) Ferrous sulphate, copper
Answer:
(d) Ferrous sulphate, copper
Iron and copper sulphate reacts to form ferrous sulphate and copper.
![]()
This reaction is known as displacement reaction.
Question 2.
___________ is a basic salt because it is a salt of weak acid and strong base.
(a) Sodium bicarbonate
(b) Sodium carbonate
(c) Sodium hydroxide
(d) Sodium chloride
Answer:
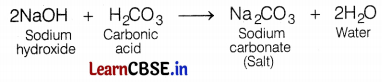
(b) Sodium carbonate
Sodium carbonate (Na2CO3) is the salt of a weak acid (carbonic acid) and a strong base (sodium hydroxide).
Question 3.
Galvanisation is a method of protecting iron from rusting by coating it with a thin layer of
(a) gallium
(b) aluminium
(c) zinc
(d) Silver
Answer:
(c) zinc
Galvanization is a method of protecting iron from rusting by coating it with a thin layer of zinc (Zn) metal.
![]()
Question 4.
Select the acid which contains four hydrogen atoms in it.
(a) Formic acid
(b) Sulphuric acid
(c) Nitric acid
(d) Acetic acid
Answer:
(d) Acetic acid
Acetic acid also known as carboxylic acid has the chemical formula CH3COOH. It has four hydrogen atoms.
Question 5.
Pooja studied the metals. She came to know about its many properties. Which of the following is a characteristic of metals?
(a) They have one to three valence electrons
(b) They have 4 to 8 valence electrons
(c) They are brittle
(d) They are capable of forming anions easily
Answer:
(a) They have one to three valence electrons
Metal can easily give their electrons and form electro-positive ions. They have one to three valence electrons in their valence shell, They are not brittle and do not form anions.
Question 6.
Which of the following is the mineral acid?
(a) Hydrochloric acid
(b) Citric acid
(c) Acetic acid
(d) Lactic acid
Answer:
(a) Hydrochloric acid
The mineral acids are those which are obtained from minerals and do not contain carbon.
e.g. HCl, H2SO4, HNO3, etc.
Question 7.
Which of the following is not a straight-chain hydrocarbon?
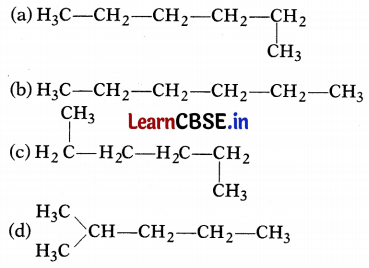
Answer:

It is a branched chain hydrocarbon and not a straight chain hydrocarbon. Rest three are straight-chain hydrocarbons.
Question 8.
In a food chain, the snake predated a rabbit which fed on fresh green bushes. What percentage amount of the energy accumulated by rabbits, would be acquired by snakes?
(a) 90%
(b) 10%
(c) 50%
(d) 25%
Answer:
(b) 10%
According to Lindemann’s 10% energy law, in a food chain, only around 10% of the available energy is passed on to the next trophic level. The rest of the energy is lost to the ecosystem in the form of heat.
Question 9.
Energy in the case of higher plants and animals is obtained by
(a) breathing
(b) tissue respiration
(c) organ respiration
(d) digestion of food
Answer:
(b) tissue respiration
Tissue respiration is the exchange of oxygen and carbon dioxide between different tissues of plants and animals.
Question 10.
In human males, all the chromosomes are paired perfectly, except one. These unpaired chromosomes are
(i) large chromosome
(ii) small chromosome
(iii) Y-chromosome
(iv) X-chromosome
Which of the following options is correct regarding the same?
(a) (i) and (ii)
(b) (iii) and (iv)
(c) (iii) only
(d) (ii) and (iv)
Answer:
(b) (iii) and (iv)
In human males, all chromosomes are paired perfectly, except one. These unpaired chromosomes are the Y-chromosome and X-chromosome. In human males, one pair called the sex chromosomes are unpaired. Here, one is of normal size (X-chromosome) while the other is shorter (Y-chromosome)
![]()
Question 11.
The diagram shows the central nervous system, which has been blocked in three different places by a drug used as an anesthetic.
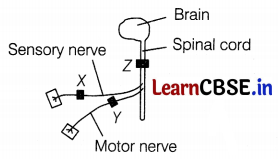
Three men had one anesthetic block at X, Y, and Z respectively. One of the men can move his leg in response to a pinprick but does not feel it. Where is the anesthetic block in this man?
(a) At X
(b) At Y
(c) At Z
(d) No block
Answer:
(c) At Z
Movement of the leg in response to a pinprick is a reflex action. Reflex action is controlled by the spinal cord and does not involve any thinking, hence brain does not play any role.
Question 12.
A Planaria worm is cut horizontally from the middle into two halves P and Q. Another Planaria worm is cut vertically into two halves R and S. Which of the cut pieces of the two Planaria worms could regenerate to form the complete worm?
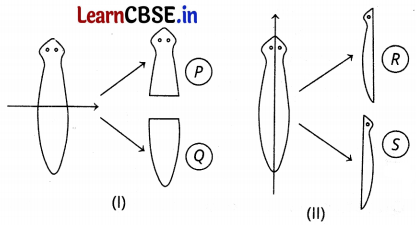
(a) Only P
(b) R and S
(c) P and Q
(d) P, Q, R, and S
Answer:
(d) P, Q, R, and S
The Planaria reproduces through the regeneration method, therefore all the cut parts (i.e. P, Q, R, and S) of the Planaria will regenerate to form complete worms.
Question 13.
The following figures show the path of light rays through three lenses marked L1, L2, and L3 and their focal points F1, F2, and F3, respectively.
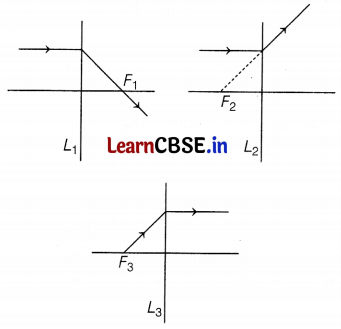
Out of L1, L2, and L3 concave lens/lenses is/are
(a) only L4
(b) only L2
(c) only L3
(d) Both L1 and L3
Answer:
(b) only L2
A concave lens is a diverging lens. It diverges the parallel beam of light rays (as shown in the figure)
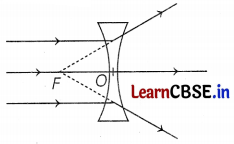
The ray parallel to the principal axis is getting diverged in the case of lens L2 only. So, only lens L2 is a concave lens.
Question 14.
The size of the pupil of the eye is adjusted by
(a) cornea
(b) retina
(c) iris
(d) blind spot
Answer:
(c) iris
Iris is a dark muscular diaphragm that controls the size of the pupil.
Question 15.
Which statement is true for a dominant allele?
(a) It cannot undergo mutation
(b) It gives a greater chance of survival than a recessive allele
(c) It gives the same phenotype in heterozygotes and homozygotes
(d) It is only responsible for male characteristics
Answer:
(c) It gives the same phenotype in heterozygotes and homozygotes
Dominant allele always expresses itself whether present in heterozygous or homozygous conditions. (Excluding exceptions).
Question 16.
Which of the listed tools was used to study the law of inheritance in pea plants by Gregor Johann Mendel?
(a) Family tree
(b) Pedigree tree
(c) Punnett square
(d) Herbarium sheet
Answer:
(c) Punnett square
Punnett square was used by Gregor Johann Mendel to determine the low of inheritance in his experiments with pea plants.
Directions (Q.Nos. 17-20) consist of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true, but R is false.
(d) A is false, but R is true.
Question 17.
Assertion (A): Acetic acid is more acidic than alcohol.
Reason (R): The ion formed after the removal of a proton from acetic acid is less stable.
Answer:
(c) Assertion is true, but Reason is false.
Acetic acid is more acidic than alcohol because of the stability of ions formed after the removal of a proton.
![]()
Question 18.
Assertion (A): Lymph also known as colourless tissue.
Reason (R): It lacks erythrocytes.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Lymph is similar to the plasma of blood but is colorless due to a lack of erythrocytes. Erythrocytes contain hemoglobin, which provides red colour to blood. Due to its absence, lymph is colourless.
Question 19.
Assertion (A): Fertilisation is a unique feature in flowers.
Reason (R): It is followed by pollination.
Answer:
(c) A is true, but R is false.
Fertilization is a unique feature of flowers because the male gamete released by pollen is involved in fertilization. The male gamete unites with the egg and this forms a zygote. It is followed by embryo formation and not pollination.
Question 20.
Assertion (A): A current-carrying solenoid, always comes to rest in a geographical N-S direction, when suspended freely.
Reason (R): One end of the current carrying a straight solenoid behaves as a North pole and the other end as a South pole, just like a bar magnet.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
When a current is flowing through a solenoid, then according to the “clock face rule” one end of the solenoid behaves as the North pole and the other end behaves as the South Pole. Hence, a current-carrying solenoid behaves as a bar magnet. On freely suspending, it comes to rest in the geographical N-S direction. Therefore, both A and R are true and R is the correct explanation of A.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
Discuss the nature of covalent bonds.
Answer:
Carbon has 4 electrons in its valence shell. To complete its octet, it either needs to gain 4 electrons or lose 4 electrons to the other atom. Both these processes are impossible. Therefore, the carbon atom achieves noble gas configuration by sharing 4 electrons with other atoms of itself or atoms of other elements. The bonds that are formed by sharing electrons are known as covalent bonds. In covalent bonding, both atoms share the valence electrons, i.e. the shared electrons belong to the valence shells of both atoms. CH3Cl is called chloromethane, which contains 1 carbon atom, 3 hydrogen atoms, and 1 chlorine atom. (1)
Question 22.
Outline a project, which aims to find the dominant coat colour in dogs.
Answer:
Select a homozygous black (BB) male dog and a homozygous white (bb) female dog and cross-breed them. The resultant offspring (F1-generation) will be
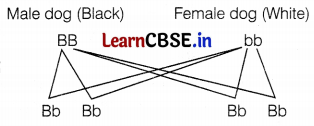
If all the offspring of the F1 generation are black, we can conclude that black colour is dominant over white coat colour in dogs and if all the offsprings are white, the dominant colour will be white. (2)
Question 23.
Difference between exocrine and endocrine glands.
Or
What is a reflex arc? How do muscle cells move?
Answer:
Differences between the exocrine gland and the endocrine gland are as follows.
| Exocrine gland | Endocrine gland |
| It does not pour its secretion into lymph or blood. | It pours its secretion into lymph or venous blood. |
| A duct is often present. | The glands are without ducts or are ductless. |
| The secretion is poured directly over the target tissue. | The secretion is transported to the target tissue through blood. |
| The secretion is enzymatic, lubricant, or excretory. | The secretion contains hormones. |
| Examples: Sweat glands, lacrimal glands, salivary glands, mammary glands, etc. | Examples: Pituitary gland thyroid gland and adrenal gland. |
Or
The pathway taken by nerve impulses in a reflex action is called the reflex arc. They allow rapid response to a stimulus, 0 g. pulling of hand on touching a hot object. (1)
Muscle cells have special proteins that change their shape and arrangement in the cell In response to electrical impulses. This forces the muscle cells to contract and relax, causing their movement. (1)
Question 24.
The refractive indices of the three media are given below.
| Medium | Refractive Index |
| A | 1.6 |
| B | 1.8 |
| C | 1.5 |
A ray of light is traveling from A to B and another ray is traveling from B to C.
(a) In which of the two cases the refracted ray bends towards the normal?
(b) In which case does the speed of light increase in the second medium? Give reasons for your answer.
Answer:
(a) When light travels from an optically rarer medium to an optically denser medium it moves towards the normal. Then, nB > nA. The light ray will bend towards the normal on passing from medium A to B.
(b) The speed of light will increase when the light travels from B to C, since nC < nB and v = (c/n), the speed of light ray will increase in the second medium. (1)
Question 25.
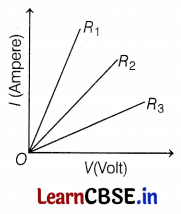
Student experiments and plots the V-I graph of three samples of nichrome wire with resistances R1, R2, and R3, respectively as shown in the figure. Interpret the graph by considering R1, R2, and R3 in proper order.
Or
The diagram shows the lengthwise section of a current-carrying solenoid.
Indicates current entering into the page,
Indicates current emerging out of the page.

Decide which end of the solenoid AorB, will behave as the North pole. Give a reason for your answer. Also, draw field lines inside the solenoid.
Answer:
As we know, slope of V-I graph tells about the resistance and (slope of V and I) ∝ \(\frac{1}{\text { resistance }}\), i.e,
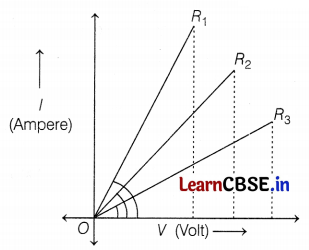
So, R3 > R2 > R1 (2)
From the diagram, we can see that the current is entering from A and emerging from B.
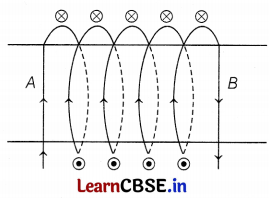
Thus, using the right-hand thumb rule, the direction of magnetic field lines is from B to A. We know that magnetic field lines move from North to South direction. Thus, B represents the North Pole and A represents the South Pole. (1)
Question 26.
(a) Describe the role of Fallopian tubes in the female reproductive system.
(b) Placenta.
Answer:
(a) Fertilisation of ovum by sperm occurs in Fallopian tubes. It acts as a site of fertilization in human females. (1)
(b) The placenta is an organ that develops in the uterus during pregnancy. It provides oxygen and nutrients to the mother. It also removes waste produced by the baby. (1)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
Cheshta, a 10th-class student was asked to identify iron, copper, zinc, and aluminium only by observing the effect of the action of concentrated nitric acid and caustic soda on each metal. How did she put the reactions of these metals with each of the reagents?
Or
Compound ‘X’ and aluminium are used to join railway tracks. Identify the compound ‘X’ and name the reaction. Write the equation for it.
Answer:
| The action of Concentrated Nitric Acid | Action of Caustic Alkali | Inference |
| No characteristic change | No characteristic change | Iron |
| Liberates brown-coloured NO2 gas | No characteristic change | Copper |
| Liberates brown-coloured NO2 gas | Liberates hydrogen gas | Zinc |
| No characteristic change | Liberates hydrogen gas | Aluminium |
Or
X is Fe2O3, i.e. iron (III) oxide.
The reaction involved is thermite reaction or aluminothermy.
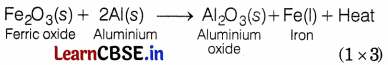
![]()
Question 28.
During the reaction of some metals with dilute hydrochloric acid, the following observations were made.
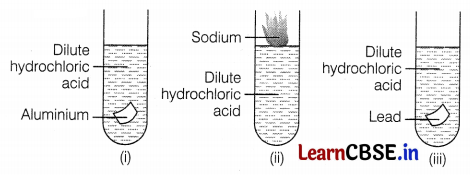
(a) The temperature of the reaction mixture rises when aluminium (Al) is added.
(b) The reaction of sodium metal is found to be highly explosive.
(c) Some bubbles of gas are seen when lead (Pb) is reacted with the acid. Explain these observations with suitable reasons.
Answer:
(a) The temperature of the reaction mixture rises when aluminium is added because it is an exothermic reaction and thus, heat gets liberated in these reactions. (1)
(b) Reaction of sodium metal is found to be highly explosive because it is an exothermic reaction. (1)
(c) When lead is treated with hydrochloric acid, bubbles of hydrogen gas are evolved.

Question 29.
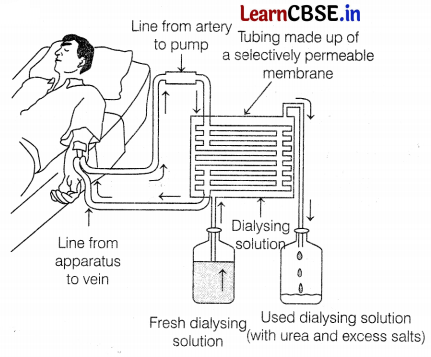
(a) Identify the process taking place in the above diagram. When does this process become essential for an individual?
(b) Describe the above process in detail.
Answer:
(a) The process is called dialysis. This process is essential for an individual who is suffering from complete renal failures, i.e. both kidneys are damaged due to an infection, injury, high BP, etc. (1)
(b) The dialysis machine, also known as an artificial kidney, contains several tubes with a semi-permeable lining suspended in a tank filled with a dialyzing fluid. This fluid has the same osmotic pressure as that of blood except, it is devoid of nitrogenous wastes such as urea. During the procedure of dialysis, the patient’s blood is passed through these tubes.
As the blood passes, the waste products from the blood move into dialyzing fluid by diffusion, and the purified blood is pumped back into the patient’s body. The dialysis unit allows the blood to run along one side of a cellophane membrane and dialysis fluid in opposite directions. This is generally done to maintain the concentration gradient between the patient’s blood and the dialysis fluid. (2)
Question 30.
‘In humans, there is a 50% probability that a boy will be born and 50% probability that a girl will be born’. Justify the statement based on the mechanism of sex determination in human beings.
Answer:
All children will inherit an X chromosome from their mother regardless of whether he is a boy or a girl. Thus, the sex of the child will be determined by what they inherit from their father. A child who inherits an X-chromosome from her father will be a girl and one who inherits a Y-chromosome will be a boy. Humans have 23 pairs of chromosomes. Both males and females carry two sets of sex chromosomes. Male (XY) has one X and one Y sex chromosome. Female (XX) has both X sex chromosomes. (3)
Question 31.
The diagram shown below is the connection of
(a) 1 Ω and 2 Ω resistors in series.
(b) 12 Ω and 2 Ω resistors in parallel.
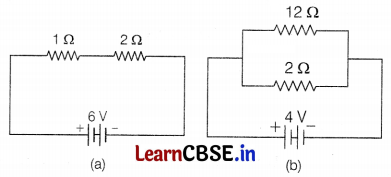
Compare the power used in 2 Ω resistor in each of the following circuits.
(a) A 6V battery in series with 1 Ω and 2 Ω resistors,
(b) A 4V battery in parallel with 12 Ω and 2 Ω resistors.
Answer:
(a) The circuit is shown in lift;
Resistances are connected in series combination.
Current in the circuit, I = \(\frac{V}{R_1+R_2}=\frac{6}{3}\) = 2 A
∴ Power used = I2R
= (2)2 × 2
= 2 × 2 × 2
= 8W (1)
(b) The circuit is shown on right,
In parallel combination, the potential across each resistor is the same and equal to the potential applied to the circuit.
The potential across 2 Ω resistor, V = 4V
Power used = \(\frac{V^2}{R}=\frac{4 \times 4}{2}\) = 8W
The power used in both the cases is same. (2)
![]()
Question 32.
An object of height 5 cm is placed on the principal axis of a concave mirror of focal length 20 cm. If the virtual image is formed by the concave mirror at a distance of 10 cm from its pole, then find
(a) the size of the image.
(b) magnification produced by the mirror.
Answer:
(a) Given, height of an object, h1 = 5 cm, f = -20 cm and v = 10 cm

Alternate solution
Given, height of an object, h1 = 5 cm, f = -20 cm and v = 10 cm
Linear magnification of concave mirror, m = \(\frac{f-v}{f}\)

Question 33.
State whether an a-particle will experience any force in a magnetic field, if (α-particles are positively charged particles)
(a) it is placed in the field at rest.
(b) it moves in the magnetic field parallel to field lines.
(c) it moves in the magnetic field perpendicular to field lines.
Justify your answer in each case.
Answer:
(a) No, it will not experience any force. A magnetic field exerts force on a moving charged particle only. (1)
(b) No, it will not experience any force because the magnetic field exerts a force in a perpendicular direction to the motion of the particle. (1)
(c) Yes, it will experience a force in a direction perpendicular to the direction of its motion and the direction of the magnetic field can be determined by Fleming’s left-hand rule. (1)
Section D
Questions No. 34 to 36 are long answer questions.
Question 34.
Kamini was studying the compound C6H14. She wondered about its different structure with the same formula. Give the name of the compound. Also, write the structural formulae of all the isomers of an alkane with six C-atoms (C6H14).
Or
(a) Calcium reacting with water starts floating. Give the reason behind this and write the balanced chemical equation of the reaction.
(b) Rashmi was studying about CH3Cl compound. Show the bond formation in this compound.
Answer:
The name of the compound is hexane and it has the following five isomers.
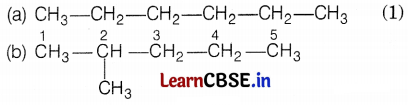
[Here, 5 carbon atoms are arranged in a straight I line, one is branched at 2 C-atoms] (1)
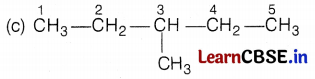
[Here, the branch is at 3C-atom] (1)
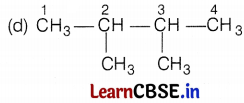
[Here, branches are at C-2 and C-3 atoms] (1)
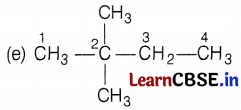
[Here, branches are at C-2 carbon atom only] (1)
Or
(a) Calcium reacts with cold water to form calcium hydroxide and hydrogen gas. (2)

The bubbles of hydrogen gas produced stick to the surface of calcium and hence, it starts floating on the surface of water. (1)
(b) Electronic configuration of carbon, 6 = 2, 4 (K = 2, L = 4)
Electronic configuration of hydrogen, 1 = 1 (K = 1)
Electronic configuration of chlorine, 17 = 2, 8, 7 (K = 2, L = 8, M = 7)
The carbon atom has four outermost electrons, each hydrogen atom has one electron and chlorine has seven outermost electrons. Carbon shares its four outermost electrons with 3 hydrogen atoms and 1 chlorine atom to form CH3Cl as follows.
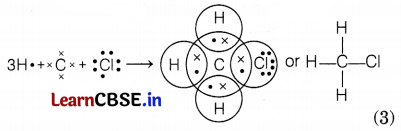
Question 35.
Differentiate between the following.
(a) Pollen tube and style
(b) Fission in Amoeba and Plasmodium
(c) Fragmentation and regeneration
(d) Bud of Hydra and Bryophyllum
(e) Vegetative propagation and spore formation
Or
(a) Hormones are needed by our body in an appropriate amount, slightly more or less secretion causes disorders in our body. Illustrate this by using three examples.
(b) Why do we call the pituitary gland a master gland? Where is it located?
Answer:
(a) Differences between pollen tube and style are
| Pollen Tube | Style |
| A tube growing out of pollen grain when it reaches the stigma. | The middle elongated part of the carpel, i.e. female part of a flower. |
| It transports male gametes from pollen grains to ovules. | The attachment of stigma to the ovary. |
(b) Fission in Amoeba is binary and in Plasmodium is multiple. The difference is
| Binary Fission | Multiple Fission |
| The parental body divides into two identical daughter cells at a time. | The parental body divides into numerous daughter cells simultaneously. |
(c) Differences between fragmentation and regeneration is
| Fragmentation | Regeneration |
| The method in which a multicellular organism breaks up into two or more smaller fragments. | The growth of a whole new organism from any of its body parts, i.e. single segment forming a new individual. |
(d) Differences between the bud of Hydra and Bryophyllum is
| Bud of Hydra | Bud of Bryophyllum |
| It is seen during budding as an outgrowth on the body of Hydra, which gets fully grown and then detaches from the body and becomes a new individual. | It is present on the leaf margins of the leaf of Bryophyllum and develops into a new plant when it comes in contact with soil and other favourable conditions. |
(e) Differences between vegetative propagation and spore formation is
| Vegetative Propagation | Spore Formation |
| New plants are obtained from different parts of the parent body like leaves, stems, etc. | Spores when fall on land, can germinate and produce new fungal colonies under favourable conditions. |
Or
(a) Hypersecretion (more secretion) or hyposecretion (less secretion) of different hormones leads to various disorders In our body. The three common examples are
(i) Goitre: Iodine acts as the necessary component for the synthesis of thyroxine hormone from the thyroid gland. This disorder is caused due to the deficiency of iodine that leads to the hyposecretion of thyroxine.
(ii) Gigantism and dwarfism: Hypersecretion of growth hormone results in gigantism (very tall individual). On the contrary, the hyposecretion or deficiency of growth hormone at an early stage of life makes the person very short, i.e. causes dwarfism.
(iii) Diabetes mellitus: Insulin secreted by the pancreas helps to lower the blood glucose level. When it is secreted in less amount, the body suffers from diabetes.) (3)
(b) The pituitary gland secretes several hormones that regulate various functions of the body. It also controls the functioning of the other endocrine glands. Hence, it is called as master gland. The pituitary gland is located just below the hypothalamus at the base of the brain. (2)
![]()
Question 36.
It is desired to obtain an erect image of an object, using a concave mirror of a focal length of 12 cm.
(a) What should be the range of distance of an object placed in front of the mirror?
(b) Will the image be smaller or larger than the object? Draw a ray diagram to show the formation of the image in this case.
(c) Where will the image of this object be, if it is placed 24 cm in front of the mirror? Draw a ray diagram for this situation to justify your answer. Show the positions of the pole, principal focus, and the center of curvature in the above ray diagrams.
Or
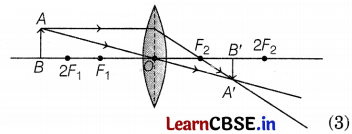
A student wants to project the image of a candle flame on the walls of a school laboratory by using a lens.
(a) Which type of lens should he use and why?
(b) At what distance in terms of focal length F of the lens should he place the candle flame, to get
(i) a magnified and
(ii) a diminished image respectively, on the wall?
(c) Draw ray diagrams to show the formation of the image in each case.
Answer:
(a) f = -12 cm
Thus, range to obtain an erect image
⇒ 0 < u < 12 (1)
(b) the Image will be larger than the object (2)
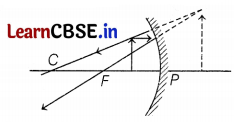
(c) Position of image
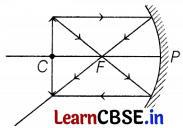
Here, f = -12 cm, u = -24 cm, v = ?
By using the mirror formula,
\(\frac{1}{f}=\frac{1}{v}+\frac{1}{u}\)
⇒ \(\frac{1}{v}=\frac{1}{-12}-\frac{1}{(-24)}\)
⇒ v = -24 cm (2)
Or
(a) He should use a convex lens as real images are formed by it.
(b) (i) For a magnified image, he should place the candle flame between the focus (F) and the center of curvature (2F) of the lens.
(ii) To get the diminished image, he should place the candle flame beyond the center of curvature (2F) of the lens. (2)
(c) (i) For magnified image
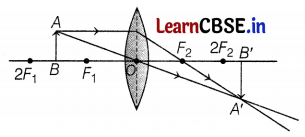
(ii) For diminished image
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
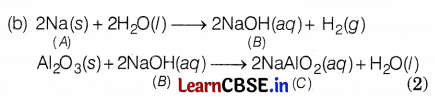
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) ‘A’ is an alkali metal. |
| (ii) ‘A’ gives a compound ‘S’ (molecular mass = 40) on reacting with water. |
| (iii) On treatment with aluminium oxide ‘6’ gives a soluble compound ‘C’. |
Based on the above hints answer the following question.
(a) Identify A and ‘C’.
(b) Write all the chemical reactions (A → B, A → C)
Or
In what forms are metals found in nature? Explain how metals react with oxygen.
Answer:
(a) A = Sodium (Na)
C = Sodium Aluminate (NaAlO2) (2)
Or
Metals are found in both free and combined states.
Reaction of metals with oxygen: All metals combine with oxygen at different rates to form metal oxides. The general equation for this reaction is (2)
metals + oxygen → metal oxide.
Question 38.
To study the ozone layer depletion, Mrs. Sharma, a science teacher drew the given flow chart on the blackboard. After completing this topic she asked some questions from students. Help them by answering the following questions.
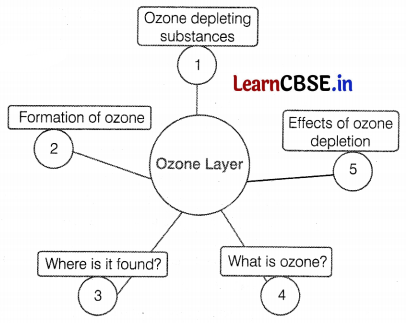
(a) What are ozone-depleting substances?
(b) Ravi wants to draw the ozone layer in a diagram showing different layers of the atmosphere, where does he label it?
(c) How is ozone formed?
Or
What could be the effect of ozone depletion?
Answer:
(a) Substances that are responsible for the depletion of the ozone layer or breakdown of ozone molecules are known as ozone-depleting substances, e.g. CFCs, halogens, nitrous oxide, CCl4, and CH4 are ozone-depleting substances responsible for ozone layer depletion. (1)
(b) The ozone layer is found in the stratosphere around 15-30 km above the earth’s surface. (1)
(c) Atomic oxygen is highly reactive. It combines with molecular oxygen under the action of UV radiations to form ozone.
O2 \(\stackrel{\mathrm{UV}}{\longrightarrow}\) [O] + [O]
2O2 + 2 [O] → 2O3 (Ozone) (2)
Or
Cancers, mutations, effects on eyesight, global warming, weakening of the immune system, etc. are some adverse effects of ozone depletion. (2)
![]()
Question 39.
How does a metal conductor conduct electricity? You would think that a low-energy electron has great difficulty passing through a solid conductor. Inside a solid, the atoms are packed together with very little spacing between them. But it turns out electrons can ‘travel’ through a perfect solid crystal smoothly and easily, almost as if they were in a vacuum.
The motion of electrons in a conductor, however, is very different from that of charges in space. When a steady current flows through a conductor, the electrons in it move with a certain average ‘drift speed’.
One can calculate this drift speed of electrons for a typical copper wire carrying a small current and it is found to be very small of the order of 1 mms-1.
How is it then that an electric bulb lights up as soon as we turn the switch ON? It cannot be that a current starts only when an electron from one terminal of the electric supply physically reaches the other terminal through the bulb because the physical drift of electrons in the conducting wires is a very slow process.
(a) The electrons move with a certain speed inside the conductor when a battery is connected across it, is called ___________
(b) The order of drift speed of electrons in copper wire is ___________
(c) Do the drift speed and thermal speed of electrons inside a metal conductor remain the same?
Or
How does an electric bulb light up as soon as we turn the switch ON?
Answer:
(a) When the battery is connected across the conductor, then a steady current flows through the conductor and the electrons in it move with a certain average speed called drift speed. (1)
(b) The drift speed of electrons for a typical copper wire is found to be in the order of 1 mms-1 = 10-3 m/s. (1)
(c) No, the drift speed of electrons inside the conductor is much smaller than its thermal velocity. (2)
Or
When we turn the switch ON, then free electrons near the electric bulb start drifting due to the electric field inside the conductor, hence electric current starts flowing through the bulb as soon as we turn the switch ON. (2)