Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 4 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 4 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based assessment units of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Pratyush took sulphur powder on a spatula and heated it. He collected the gas evolved by inverting a test tube over it, as shown in the figure below.
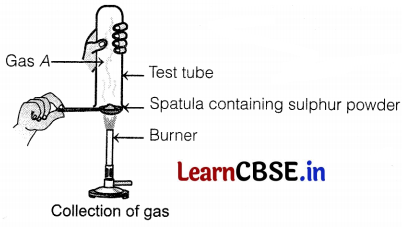
Identify gas A in the given experiment.
(a) SO2
(b) SO3
(c) H2S
(d) N2
Answer:
(a) SO2
The gas that evolved in the given experiment is sulphur dioxide (SO2).
Question 2.
Carbon can use four hydrogen atoms to form methane (CH4), because
(a) valency of carbon is four
(b) valency of hydrogen is one
(c) Both (a) and (b)
(d) carbon gets noble gas configuration by making four covalent bonds with hydrogen
Answer:
(c) Both (a) and (b)
Carbon has 4 electrons in its valence shell, while hydrogen has one electron in its valence shell. To complete their octet and duplet respectively, they form covalent bonds. Carbon utilizes its 4 valence electrons and forms 4 covalent bonds with 4 hydrogen atoms, using one valence electron with each hydrogen atom.
![]()
Question 3.
A molecule of ammonia (NH3) has
(a) only single bonds
(b) only double bonds
(c) only triple bonds
(d) two double bonds and one single bond
Answer:
(a) only single bonds
A molecule of ammonia (NH3) has only single bonds and these are covalent.
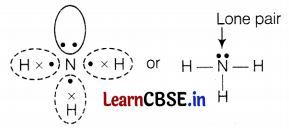
Question 4.
With the reference to four given oxides, which one of the options in the table is correct?
| Oxide | Nature of Oxide |
| (a) SO2 | Basic |
| (b) H2O | Acidic |
| (c) Al2O3 | Amphoteric |
| (d) CaO | Neutral |
Answer:
(c) Al2O3 – Amphoteric
(a) SO2 is acidic oxide because S is non-metal and non-metals form acidic oxide.
(b) H2O is a neutral oxide.
(c) Al2O3 is an amphoteric oxide that behaves as an acid as well as a base.
(d) CaO is basic oxide because Ca is a metal and metals form basic oxide.
Question 5.
Gold is used for making jewellery. What are the properties of gold that make it a suitable metal for making jewellery?
(a) Ductility
(b) Malleability
(c) Lustrous
(d) All of these
Answer:
(d) All of these
Gold has all given properties that make it suitable for making jewellery. Gold is ductile, malleable, and lustrous. It can be drawn into thin sheets and wires and has a shiny appearance.
Question 6.
Due to the formation of ________, silver articles become black on prolonged exposure to air.
(a) Ag3N
(b) Ag3O
(c) Ag2S
(d) Ag2S and Ag3N
Answer:
(c) Ag2S
Silver articles become black because silver reacts with the gas present in the air to form a black coating of Ag2S. The reaction is as follows.
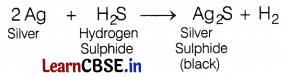
Question 7.
Which among the following are unsaturated hydrocarbons?
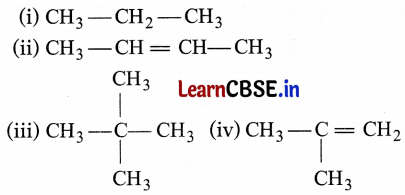
(a) (i) and (iii)
(b) (ii) and (iii)
(c) (ii) and (iv)
(d) (ii), (iii) and (iv)
Answer:
(c) Unsaturated hydrocarbons have double or triple bonds in their structure. Both (ii) and (iv) have double carbon-carbon bonds in their structures.
![]()
Question 8.
Which of the following statement(s) is (are) true about the heart?
(i) The Left atrium receives oxygenated blood from different parts of the body while the right atrium receives deoxygenated blood from the lungs.
(ii) The left ventricle pumps oxygenated blood to different body parts while the right ventricle pumps deoxygenated blood to the lungs.
(iii) The left atrium transfers oxygenated blood to the right ventricle which sends it to different body parts.
(a) Only (i)
(b) Only (ii)
(c) Only (ii)
(d) (i) and (iii)
Answer:
(c) Only (ii)
Statement (ii) is correct about the heart.
The route of blood circulation in the heart is as follows.
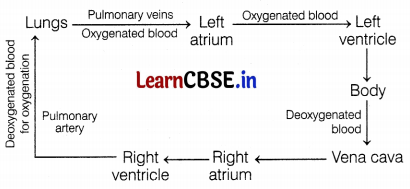
Question 9.
A plant is kept in the dark for two days. A leaf from this plant is used in an experiment to investigate the effect of two factors on photosynthesis as shown in the diagram.
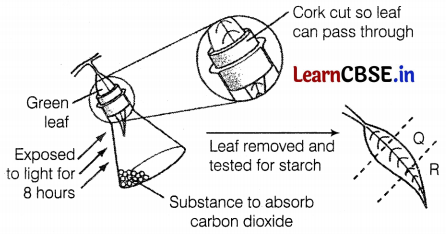
What are the colours of Q and R, when the leaf is tested for starch, using an iodine solution?
| Q | R |
| (a) Blue/Black | Brown |
| (b) Brown | Brown |
| (c) Blue/Black | Blue/Black |
| (d) Brown | Blue/Black |
Answer:
(b) Q – Brown, R – Brown
The colours of both Q and R regions when tested with iodine solution appeared brown. This indicates that there was no starch in both regions. In both regions Q and R photosynthesis did not occur, since part Q of the leaf was covered by the cut part of the cork, while part R was exposed to KOH kept in the flask to absorb CO2. In the absence of light (for Q) and CO2 (for R), photosynthesis did not occur, thus starch was not formed and regions Q and R tested negative with iodine (A positive test for starch converts the leaf blue-black with iodine). (1)
Question 10.
A cross between a tall pea plant (TT) and a short pea plant (tt) resulted in progeny that were all tall plants because
(a) tallness is the dominant trait
(b) shortness is the dominant trait
(c) tallness is the recessive trait
(d) height of the pea plant is not governed by gene ‘T’ or ‘t’.
Answer:
(a) tallness is the dominant trait
In the F1 generation, the cross between TT and tt will result in all tall plants as shown in the figure given below. Thus, tallness is the dominant trait that expresses itself regardless of its presence in the homozygous or heterozygous state.
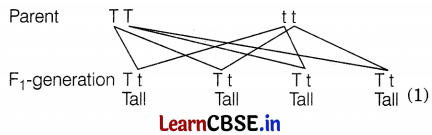
Question 11.
The correct sequence of organs in the male reproductive system for the transport of sperm is
(a) testis → vas deferens → urethra
(b) testis → ureter → urethra
(c) testis → urethra → ureter
(d) testis → vas deferens → ureter
Answer:
(a) testis → vas deferens → urethra
Sperms formed in testis are delivered through the vas deferens which join with another tube called the urethra coming from the urinary bladder.
![]()
Question 12.
Consider the following figure concerning the brain.
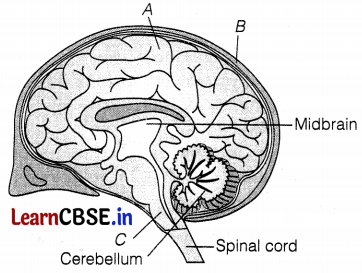
Identify the unlabelled parts (A, B, and C) by choosing the correct option below.
| A | B | C | |
| (a) | Medulla | Cerebrum | Skull |
| (b) | Skull | Medulla | Cerebrum |
| (c) | Cranium | Cerebrum | Medulla |
| (d) | Cerebrum | Cranium | Medulla Oblongata |
Answer:
(d) A – Cerebrum, B – Cranium, C – Medulla Oblongata
A is the cerebrum, which is responsible for reasoning, speech, hearing, intelligence, and usage of information.
B is the cranium, which protects the brain, while C is the medulla oblongata which controls involuntary actions, such as breathing, blood pressure (BP), etc.
Question 13.
An object is placed in front of a convex mirror. Its image is formed
(a) at a distance equal to the object distance in front of the mirror.
(b) at twice the distance of the object from the mirror.
(c) half the distance of the object in front of the mirror.
(d) behind the mirror and its position varies according to the object’s distance.
Answer:
(d) behind the mirror and its position varies according to the object’s distance.
An object is placed in front of a convex mirror, its image is formed behind the mirror and its position varies according to the object’s distance.
Question 14.
The bluish colour of water in the deep sea is due to
(a) the presence of algae and other plants found in water
(b) reflection of the sky in the water
(c) scattering of light
(d) absorption of light by the sea
Answer:
(c) scattering of light
The bluish colour of water in the deep sea is due to the scattering of light as the very fine particles of H2 and O2 molecules present in water are scattered mainly blue light.
Question 15.
Which statement about the genotypes of 1 organism is correct?
(a) Dominant alleles are only found in homozygous
(b) One recessive allele always causes a recessive phenotype
(c) Recessive phenotypes must be homozygous
(d) The dominant phenotype must be heterozygous
Answer:
(c) Recessive phenotypes must be homozygous
A recessive trait requires a homozygous condition to express itself.
Question 16.
The development of a seedling from an embryo under appropriate conditions is called
(a) regeneration
(b) germination
(c) vegetative propagation
(d) pollination
Answer:
(b) germination
Germination is a process occurring in plants in which the embryo develops into a seedling under appropriate conditions. (1)
Direction (Q.Nos. 17-20) consists of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): Graphite is slippery to the touch.
Reason (R): The various layers of carbon atoms in graphite are held together by weak Vander Waals’ forces.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
A graphite crystal consists of various layers of carbon atoms in which each carbon atom is joined to three other atoms by strong covalent bonds. The various layers of carbon atoms in graphite are held together by weak van der Waals forces making it slippery to touch.
![]()
Question 18.
Assertion (A): Plants lack the nervous system, but they do coordinate.
Reason (R): It is so because of hormones.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Plants lack the nervous system but coordinate via hormones like auxin, gibberellins, cytokinins, etc.
Question 19.
Assertion (A): Blood pressure is much greater in arteries than veins.
Reason (R): The force exerted by blood against the wall of the vessel is called blood pressure.
Answer:
(b) Both A and R are true, but R is not the correct explanation of A.
Blood pressure is much greater in arteries because of the faster pumping action of the heart which sends blood rapidly in arteries. (1)
Question 20.
Assertion (A): The magnetic field produced by a current-carrying solenoid is independent of its length and cross-sectional area.
Reason (R): The magnetic field inside the solenoid has a variable value.
Answer:
(c) A is true but R is false.
The magnetic field produced by a current-carrying solenoid is independent of its length and area of cross-section. It only depends on the number of turns and current flowing through the solenoid. So, Assertion is true.
The magnetic field produced inside a current-carrying solenoid is uniform. So, the Reason is false. (1)
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
A solid limestone is burnt to form quicklime and a gas. Define the reaction involved and also write its chemical equation.
Answer:
A reaction in which a single reactant breaks down to form two or more products is known as a decomposition reaction. The chemical equation can be represented as
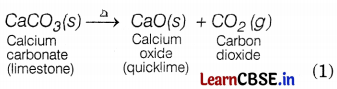
Question 22.
Explain how the movement of leaves of a sensitive plant is different from the movements of shoots towards light.
Answer:
Movements in Mimosa pudica (sensitive plant) occur in response to touch. In such movements, plant cells change shape by changing the amount of water in them resulting in folding up and drooping of leaves. This movement is independent of growth. (1)
Plants respond to a stimulus by growing in a particular direction and the movement is due to growth. This growth is directional. Movement of shoots towards light indicates phototropism, i.e. movement occurs in response to light. (1)
![]()
Question 23.
An acid is secreted in our stomach. Why?
Or
Differentiate between respiration in plants and respiration in animals.
Answer:
HCl (Hydrochloric Acid) is the acid secreted inside the stomach. It is important for the body because
(i) It makes the medium inside the stomach acidic, which is necessary for the activation of an enzyme called pepsin. (1)
(ii) It also kills any bacteria, entering the stomach along with food. (1)
Or
| Respiration in Plants | Respiration in Animals |
| (i) Plants do not possess a respiratory system. | (i) Animals have respiratory systems. |
| (ii) Diffusion of respiratory gases directly takes place into the cells. | (ii) The respiratory gases are transported upto the tissue cells. |
Question 24.
Observe the following incomplete ray diagram of an object where the image A’B’ is formed after refraction from a convex lens.
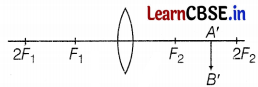
Based on the above information fill in the blanks.
(a) The position of object AB would have been ________
(b) The size of the object would have been ________ than the size of the image.
Answer:
(a) The position of object AB would have been beyond 2F1.
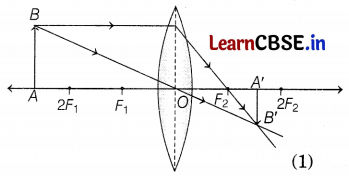
(b) The size of the object would have been bigger than the size of the image. (1)
Question 25.
The given figure shows three resistors.
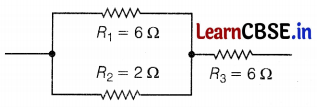
Find the combined resistance.
Or
For the current carrying solenoid as shown below, draw magnetic field lines and give a reason to explain that out of the three points A, B, and C at which point the field strength is maximum and at which point it is minimum.
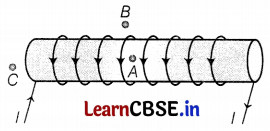
Answer:
Here, R1 is parallel to R2
For parallel combinations,
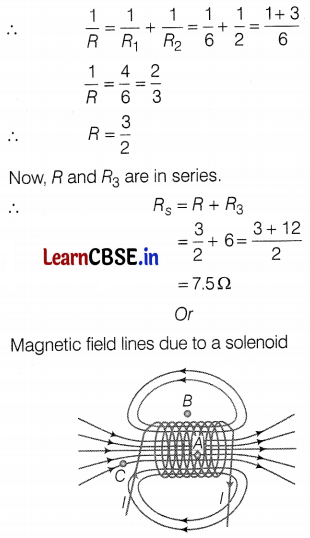
In the case of an ideal solenoid, the magnetic field strength is maximum at point A and is minimum or zero at point B. This is because the magnetic field is strong, where magnetic field lines are crowded, and is weak, where magnetic field lines are far apart. At point C, the density of the field lines is less than that of point A but greater than that of point B. So, the order of the magnetic field at points A, B, and C is BB < BC < BA. (2)
Question 26.
Vegetarian food habits can sustain a larger number of people. Justify the statement in terms of the food chain.
Answer:
In a food chain, about 90% of the energy gets lost at each trophic level. This means that in long food chains, very little energy from the producer is available to the top carnivores. For example,

But in shorter food chains, more energy will be available to man.
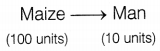
Hence, vegetarian food habits, where nutrition is derived directly from producers, can sustain a large number of people. (2)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
State which of the following chemical reactions will take place or which will not, giving the suitable reason for each.
(a) Zn (s) + CuSO4 (aq) → ZnSO4 (aq) + Cu (s)
(b) Fe (s) + ZnSO4 (aq) → FeSO4 (aq) + Zn (s)
(c) Zn (s) + FeSO4 (aq) → ZnSO4 (aq) + Fe (s)
Answer:
(a) Zinc displaces copper from copper sulphate because zinc is more reactive than copper as it is placed above copper in the reactivity series of metals. (1)
(b) This reaction will not occur because iron is less reactive than zinc as it is placed below zinc in the reactivity series of metals. (1)
(c) Zinc displaces iron because zinc is more reactive than iron as it is placed above iron in the reactivity series of metals. (1)
![]()
Question 28.
What are soaps chemically? How do they differ from synthetic detergents? Also, mention their uses.
Or
An organic compound A is a constituent of many medicines is used as an antifreeze and has the molecular formula C2H6O. Upon reaction with alk.KMnO4, the compound A is oxidized to another compound B with the formula C2H4O2. Identify the compounds A and B. Write the chemical equation for the reaction that leads to the formation of B.
Answer:
Soaps are chemically sodium or potassium salts of long-chain carboxylic acids. In contrast, detergents are ammonium or sulphonate salts of long-chain carboxylic acids. Both soaps and detergents are used as cleansing agents. Soaps are used for washing clothes only in soft water, i.e. water which does not contain Mg2+ and Ca2+ ions, while detergents are used to make shampoos and products for cleaning clothes even in hard water. (3)
Or
The organic compound A which is a constituent of many medicines and act as antifreeze with the molecular formula C2H6O is ethanol (CH3CH2OH). Ethanol is oxidized to ethanoic acid (B) upon reaction with alk. KMnO4.

Question 29.
A green stemed rose plant denoted by GG and a brown stemed rose plant denoted by gg are allowed to undergo a cross with each other. List your observations regarding
(a) the colour of the stem in their F1 progeny
(b) the percentage of brown stemmed plants in F2 progeny if F1 plants are self-pollinated.
(c) the ratio of GG and Gg in the F2 progeny.
![]()
Answer:
(a)
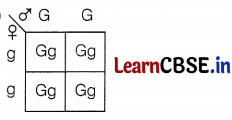
All the plants will be with green stems in F1 progeny.
(b)
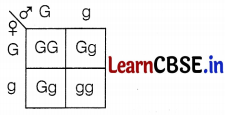
25% of plants will be brown-stemmed. (1)
(c) 25% GG
50% Gg
Question 30.
Explain, how pesticides accumulate in the environment.
Answer:
Pesticides are poisonous chemical substances, which are sprayed over crop plants to protect them from pests and diseases. These pesticides mix up with soil and water. From here they are absorbed by the growing plants along with water. These chemicals enter the environment through the food chains, in the following manner. (1)
When herbivorous animals eat plants, these poisonous chemical pesticides enter into their bodies through the food chain. When the carnivorous animals eat herbivores, then the pesticides get transferred to their bodies. In this process of transfer of food through food chains, these harmful chemicals get concentrated at each subsequent trophic level. This is known as biomagnification or bio-concentration. (2)
![]()
Question 31.
Rohit wants to have an inverted image of an object using a concave mirror. If he kept the object at a distance of 50 cm from the mirror and the image is formed at a distance of 30 cm from the mirror, then
(a) specify the range of focal length of the concave mirror.
(b) draw the ray diagram to show image formation in this case.
(c) state the magnification produced by a concave mirror.
Answer:
(a) Given, object distance, u = -50 cm, image distance, v = -30 cm
From the mirror formula,
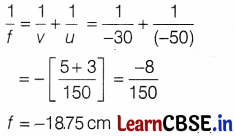
(b) The ray diagram for image formation by concave mirror is shown below.
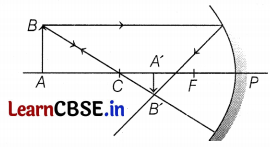
(c) Magnification of mirror,
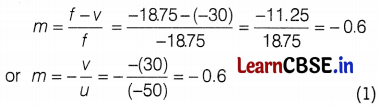
Question 32.
What is meant by the “electrical resistance” of a conductor? State how the resistance of a conductor is affected when
(a) a low current passes through it for a short duration.
(b) a heavy current passes through it for about 30 seconds.
Answer:
The electrical resistance of a conductor may be defined as the property of any substance to oppose the flow of current through it.
(a) The resistance of the conductor will increase when a low current passes through it for a short duration. (2)
(b) The resistance of the conductor will decrease when a heavy current passes through it. (1)
Question 33.
A current is allowed to flow through a solenoid. State your observations for the following cases and give reasons for the same in each case.
(a) The solenoid behaves like a magnet.
(b) The solenoid is a temporary magnet.
Answer:
(a) A solenoid is a long coil of circular loops of insulated copper wire. Magnetic field lines are produced around the solenoid when a current is allowed to flow through it. The field lines produced by it are similar to the field lines produced by a bar magnet which can be seen in the figure.
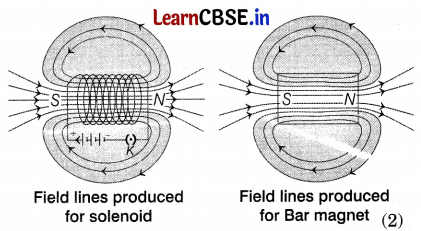
(b) The solenoid behaves like a temporary magnet which is formed by placing a soft iron core inside it. The magnetism of this magnet lasts so long as there is current flowing through it and loses its magnetic property when the current in the solenoid is stopped. It is also known as an electromagnet. (1)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
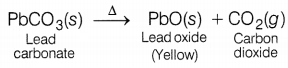
(a) The carbonate of metal X is a white solid. It decomposes when heated to form carbon dioxide and yellow solid oxide. What is metal X?
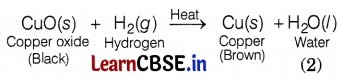
(b) What happens when Rakesh passes hydrogen gas over the heated copper oxide? Write the chemical equation involved in this reaction.
Or
Write three physical properties of each of the acids and bases. How will you show with an example that metal oxides are basic? Give chemical equation also.
Answer:
(a) Metal X is lead (Pb). Metal carbonate, i.e. Lead carbonate is a white solid amorphous powder.
When lead carbonate is heated CO2 gas is evolved along with the formation of lead oxide. (3)
(b) When hydrogen gas is passed over heated copper oxide (CuO) the black coating on the surface turns brown and copper is obtained.
Or
(i) Properties of Acids
(a) Acids are sour.
(b) Acids turn blue litmus to red.
(c) Acids are corrosive.
Properties of Bases
(a) Bases have a bitter taste.
(b) Bases feel soapy to touch.
(c) Bases turn red litmus to blue. (2)
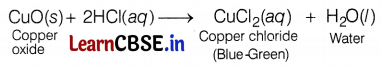
(ii) The reaction between copper oxide and dil. hydrochloric acid:
The colour of the solution becomes blue-green due to the formation of copper (II) chloride. Metallic oxide, i.e. CuO behaves as a base and forms salt and water when it reacts with an acid like HCl. Hence, metallic oxides are basic. (3)
![]()
Question 35.
Given below are certain situations. Analyse and describe its possible impact on a person.
(a) The fallopian tube of a female is plugged.
(b) Ovaries are not functional.
(c) The urethra is not functioning properly.
(d) Testosterone is not produced by the testis.
(e) Oestrogen and progesterone are not secreted by the ovaries.
Or
(a) What are animal hormones? List their two characteristics.
(b) Name the hormone,
(i) which brings change in male humans during the beginning of adolescence.
(ii) which coordinates the level of sugar in blood?
Answer:
(a) If the fallopian tube of a female is plugged then the egg will not be able to reach the uterus. Therefore, fertilization will not take place.
(b) If ovaries are not functional in the females then it will not produce ova or egg cells, which are necessary for the process of fertilization.
(c) As urethra is a common passage for both sperm and urine. So, if the urethra is not functioning properly, it will affect the transport of sperm.
(d) The testosterone also known as male sex hormone is produced by the testis. So if the testis does not produce testosterone, then it will not bring sexual maturation at puberty.
(e) Oestrogen and progesterone are the female sex hormones. These bring about sexual character during puberty and also play a major role in menstruation. (1 × 5)
Or
(a) Hormones are chemical substances, which control and coordinate the activities performed by organisms.
Characteristics of Hormones:
(i) They are poured directly into the bloodstream and are carried throughout the body by the circulatory system.
(ii) They act only on the specific target organs.
(b) (i) Testosterone produced by the testes regulates the changes in males during the adolescence period.
(ii) Insulin (decreases blood sugar) and glucagon (increases blood sugar), secreted by the pancreas coordinate the sugar level in blood.
Question 36.
It is desired to obtain an erect image of an object, using a concave mirror of a focal length of 12 cm.
(a) What should be the range of distance of an object placed in front of the mirror?
(b) Will the image be smaller or larger than the object? Draw a ray diagram to show the formation of the image in this case.
(c) Where will the image of this object be, if it is placed 24 cm in front of the mirror? Draw a ray diagram for this situation and also justify your answer.
Show the positions of the pole, principal focus, and the center of curvature in the above ray diagrams.
Or
A thin converging lens forms a real magnified image and a virtual magnified image of an object in front of it.
(a) Write the positions of the objects in each case.
(b) Draw ray diagrams to show the image formation in each part.
(c) How will the following be affected by cutting this lens into two halves along the principal axis?
(i) Focal length
(ii) Intensity of the image formed by the half lens.
Answer:
(a) Given, focal length, f = -12 cm, Thus, range to obtained erect image
⇒ 0 < u < 12 (1)
(b) The image will be larger than the object
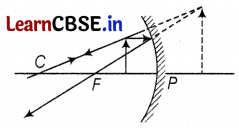
(c) Position of image
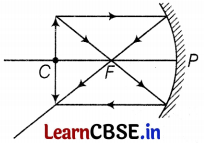
Here, f = -12 cm, u = -24 cm, v = ?
By using the mirror formula,
\(\frac{1}{f}=\frac{1}{v}+\frac{1}{u}\)
⇒ \(\frac{1}{v}=\frac{1}{-12}-\frac{1}{(-24)}\)
⇒ v = -24 cm (3)
Or
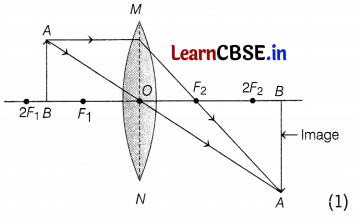
(a) (i) Object is placed between F and 2F.
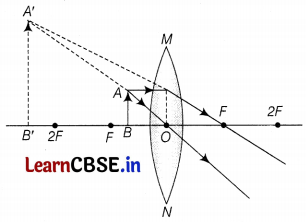
(ii) The object is placed between the optical centre and F.
(b) The ray diagrams are as follows.
Part (a)
Part (b)
(c) (i) There will be no change in focal length.
(ii) Intensity will become one-fourth. (1)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) ‘X’ metal acts as a good reducing agent. |
| (ii) ‘X’ reduces ‘Y’ and MnO2. |
| (iii) The reaction with ‘Y’ is used for joining broken railway tracks. |
Based on the above hints answer the following questions.
(a) Identify ‘X’ and ‘Y’.
(b) Write all the chemical reactions.
Or
Name the reaction which occurs in step 3. Define it.
Answer:
(a) X = Aluminium (Al), Y = Iron (III) oxide (Fe2O3) (2)
(b) 3MnO2 (s) + 4Al (s) → 3Mn (l) + 2Al2O3 (s) + Heat
Fe2O3 (s) + 2Al (s) → 2Fe (l) + Al2O3 (s) + Heat (2)
Or
The reaction involved is a thermite reaction. The reaction of metal oxide to form metal by using aluminium powder as a reducing agent is known as a thermite reaction. (2)
![]()
Question 38.
If we cross pure-breed tall (dominant) pea plants with pure-breed dwarf (recessive) pea plants, we get pea plants of F1 generation. If we now self-cross the pea plant of the F1 generation, then we obtain the pea plant of the F2 generation.
(i) What do the plants of the F1 generation look like?
(ii) What is the ratio of tall plants to dwarf plants in F2-generation?
(iii) Statement type of plants not found in F1-generation but appeared in F2-generation mentioning the reason for the same.
Or
Write the phenotypic and genotypic ratio of plants in F2-generation.
Answer:
(i) The plants produced in the F1 generation are all tall.
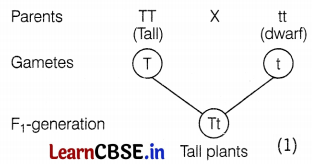
(ii) The ratio of tall plants to dwarf plants in F2-generation
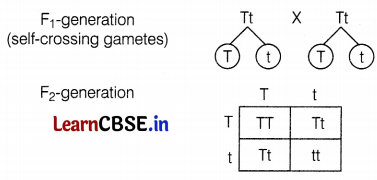
TT and Tt are phenotypically tall plants and tt is a dwarf plant ratio of tall plants in F2 generation = 3 : 1.
(iii) Dwarf pea plants are not found in F1-generation but appeared in F2-generation. This observation indicates that both the traits of shortness and tallness are inherited in the F1 generation. But only the tallness tract is expressed in the F1 generation. Because for a plant to be tall, a single copy of ‘T’ is enough. But dwarfness in pea plants is a successive trait and it requires a homozygous condition (tt) to express itself. (2)
Or
The phenotypic ratio of pea plants in F2-generation is 3 : 1. (1)
The genotypic ratio of pea plants in F2-generation is 1 : 2 : 1. (1TT : 2Tt : 1tt). (2)
Question 39.
Vinita and Roshan demonstrated a circuit that operates the two headlights and the two sidelights of a car, in their school exhibition. Based on their demonstrated circuit, answer the following questions.
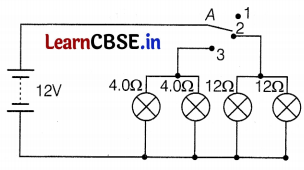
(a) State what happens when switch A is connected to
(i) Position 2
(ii) Position 3
(b) Find the potential difference across each lamp when lit.
(c) Calculate the current
(i) in each 12 Ω lamp when lit.
(ii) in each 4 Ω lamp when lit.
Or
Show, with calculations, which type of lamp, 4.0 Ω or 12 Ω, has the higher power.
Answer:
(a) (i) 12 Ω lamp (only) on
(ii) 4 Ω lamps (only) on (1)
(b) 12V for both sets of lamps and all of them are in parallel. (1)
(c) (i) 12 Ω lamps are on when the wire is connected to position 2.
The voltage across both 12 Ω lamps = 12 V
V = IR
I = \(\frac{V}{R}\)
= \(\frac{12}{12}\)
= 1A
(ii) lamps are on when the wire is connected to position 3.
The voltage across both 4 Ω lamps = 12 V
V = IR
I = \(\frac{V}{R}\)
= \(\frac{12}{4}\)
= 3A (2)
Or
Power, P = V2/R
All lamps are in parallel and hence same V for all lamps.
For 4 Ω lamps, P = \(\frac{12 \times 12}{4}\) = 36W
For 12 Ω lamps, P = \(\frac{12 \times 12}{12}\) = 12W
Hence, 4 Ω lamps will have higher power. (2)