Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 3 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 3 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer questions with 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer questions with 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
The graph given below depicts a number of electrons in an atom of different elements A, B, C, and D.
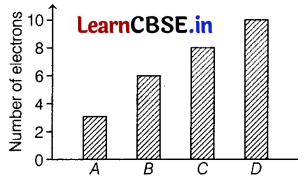
Which of the following element is a metal?
(a) A
(b) B
(c) C
(d) D
Answer:
(a) A
The number of electrons in an atom of element A is 3.
∴ Its electronic configuration is 2, 1, which means it can easily lose its outermost electron and act as a metal. The number of outermost electrons in an atom of elements B, C, and D are 4, 6, and 8 respectively. Therefore, they cannot lose electrons. Hence, they are not considered as metals.
Question 2.
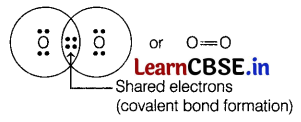
Which of the following will contain a covalent double bond between its atoms?
(a) H2
(b) O2
(c) NaCl
(d) Cl2
Answer:
(b) O2
The oxygen atom has six (6) valence electrons. Thus, to complete its octet, it forms a double bond with another oxygen atom to get an O2 molecule as
Question 3.
Which three allotropes of carbon, do the given figures represent?
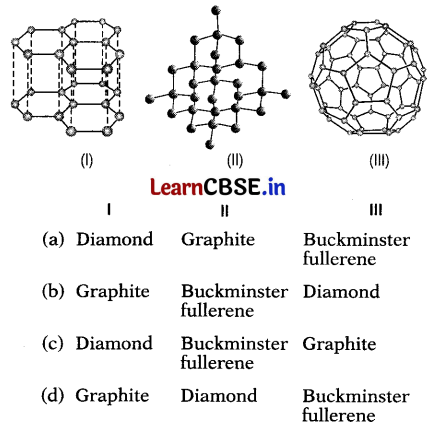
Answer:
(d) I – Graphite, II – Diamond, III – Buckminster fullerene
(I) Is the structure of graphite crystal which consists of layers of carbon atoms or sheets of carbon atoms.
(II) Is the structure of a diamond.
(Ill) The structure of Buckminsterfullerene as their structure resembles geodesic domes.
![]()
Question 4.
Which of the following is the correct representation of the electron dot structure of nitrogen?
![]()
![]()
![]()
![]()
Answer:
![]()
The electronic configuration of N (atomic number 7) is K – 2, L – 5 Therefore, it needs three more electrons to complete its octet. Each nitrogen atom shares three electrons to form a molecule of N2 as

Question 5.
Alkalis are generally soluble in water, based on this which among the following hydroxides is not an alkali?
(a) Ammonium hydroxide
(b) Calcium hydroxide
(c) Copper hydroxide
(d) Sodium hydroxide
Answer:
(c) Copper hydroxide is not an alkali because it is insoluble in water. Hence, it is a base but not an alkali.
Question 6.
__________ is the first member of the alkyne homologous series.
(a) ethene
(b) propyne
(c) ethyne
(d) methane
Answer:
(c) The general formula of alkyne is CnH2n-2. Where n is the number of carbon atoms. The first member of the alkyne homologous series is ethyne (C2H2).
Question 7.
Ajay took calcium oxide in an iron container. He slowly added some water to it. What would he observe?
(a) The container becomes cold
(b) The container becomes hot
(c) Green coloured solution is formed
(d) White precipitate is formed
Answer:
(b) The reaction between calcium oxide and water is highly exothermic, thus during the reaction between calcium oxide and water, the container becomes hot as heat is released.
Question 8.
In the following given food chain, organisms are labelled as A to D. Match the labelling referred to in Column I with their most suitable feature in Column II.
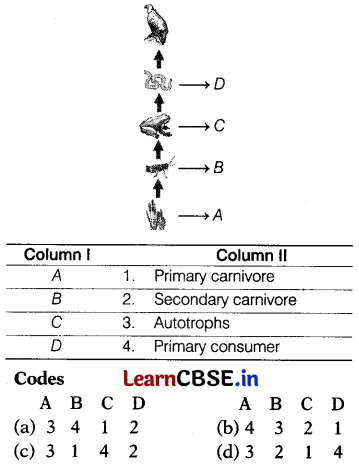
Answer:
(a) A – 3, 6 – 4, C – 1, D – 2
The given food chain is as follows.
Grass (A) → Grasshopper (B) → Frog (C) → Snake (D) → Hawk (E)
A. Grass acts as a producer in the given food chain. These are autotrophic. Thus, makes its food in the presence of sunlight. All living organisms depend upon plants directly or indirectly.
B. Grasshoppers are the primary consumers as they feed on plants (grass) directly.
C. Frogs are the primary carnivores as they feed on plant-eating animals (grasshoppers)
D. Snakes are the secondary carnivores as they eat the flesh of other animals and feed on primary carnivores majorly.
Question 9.
Select the correct option.
(a) Liver – It stores bile
(b) Small intestine – Maximum absorption of water occurs
(c) Stomach – Digestion of proteins begins here
(d) Large intestine – Absorption of digested food takes place
Answer:
(c) Stomach – Digestion of proteins begins here.
All the other options are incorrect, except (c) they can be corrected as
Liver – It synthesizes bile.
Small intestine – Absorption of digested food takes place.
Large intestine – Maximum absorption of water occurs.
![]()
Question 10.
The following results were obtained by a scientist who crossed the F1 generation of pure breeding parents for round and wrinkled seeds.

From these results, it can be concluded that the actual number of round seeds he obtained was
(a) 1881
(b) 22572
(c) 2508
(d) 5643
Answer:
(d) When pure breeding parents for round and wrinkled seeds are crossed, the ratio of phenotype of F2-generation is 3 : 1.
In the given table,
Number of F2 offspring = 7524
Number of round seeds obtained = \(\frac{7524 \times 3}{4}\) = 5643 seeds
Question 11.
Which phytohormone is responsible for the growth of the plant when light is shown from one side?
(a) Gibberellins
(b) Ethylene
(c) ABA
(d) Auxin
Answer:
(d) Auxin
When growing plants detect light, a hormone called auxin is synthesized at the shoot tip. When light shows from one side of the plant, auxin diffuses towards the shady side of the shoot. The concentration of auxin stimulates more growth on one side of the shoot which is away from the light. Thus, the plant appears to bend towards light.
Question 12.
The figure given below shows a female reproductive system in humans with labels A to D.
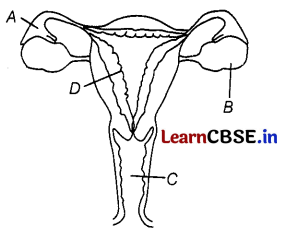
Match the labelling referred to in Column I and correlate with the function in Column II.
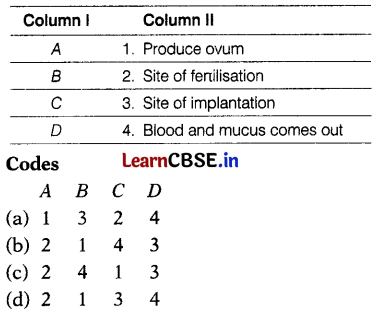
Answer:
(b) A – 2, B – 1, C – 4, D – 3
It can be explained as
A – Oviduct or Fallopian tube is the site ot fertilisation.
B – The ovary produces one mature egg (ovum) every month.
C – In the absence of fertilization, the lining of the uterus slowly breaks and comes out through the vagina as blood and mucus.
D – The lining of the uterus is the site for the implantation of the embryo.
Thus, option (b) is correct.
Question 13.
An object is placed at a distance of 10 cm in front of a plane mirror, then the distance of the image from the mirror will be
(a) 5 cm
(b) 10 cm
(c) 20 cm
(d) 0
Answer:
(b) 10 cm
The distance of the image is equal to the distance of the object from the mirror. Therefore, the distance of the image from the mirror is 10 cm.
Question 14.
If a beam of red light and a beam of violet light are incident at the same angle on the inclined surface of a prism from an air medium and produce angles of refraction r and v respectively, which of the following is correct?
(a) r = v
(b) r > v
(c) r = \(\frac{1}{v}\)
(d) r < v
Answer:
(d) r < v
Since red light deviates the least, therefore refracting angle for the red light will also be minimum. Hence, r < v.
![]()
Question 15.
The arrangement of organisms into a series of groups based on physiological, biochemical, anatomical, and other relationships is
(a) hierarchy
(b) categorization
(c) taxonomy
(d) classification
Answer:
(d) classification
Classification involves the hierarchical arrangement of living organisms into different categories based on common inter-relationships between them.
Question 16.
The maleness of a child is determined by
(a) X-chromosome in the zygote
(b) Y-chromosome in the zygote
(c) the cytoplasm of germ cells which determines the sex
(d) sex is determined by chance
Answer:
(b) Y-chromosome in the zygote
The maleness of a child is determined by the Y chromosome in the zygote inherited from the father.
Direction (Q. Nos. 17-20) These consist of two Statement – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): HCl produces hydronium ions (H3O+) and chloride ions (Cl–) in aqueous solution.
Reason (R): In the presence of water, bases give H+ ions.
Answer:
(c) A is true but R is false.
HCl produces H+ ions in an aqueous solution because, in the presence of water, acids give H+ ions. As H+ ions cannot exist alone, they combine with water molecules and form H3O+.
Question 18.
Assertion (A): In human beings, the respiratory pigment is haemoglobin.
Reason (R): It is a type of protein that has a high affinity for CO2.
Answer:
(c) A is true, but R is false.
R can be corrected as Haemoglobin has a high affinity for carbon monoxide.
Question 19.
Assertion (A): The uterus prepares itself every month to receive a fertilized egg.
Reason (R): The ovary releases one egg every month.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
It can be explained that Since the ovary releases one egg every month, the uterus also prepares itself every month to receive a fertilized egg. Its lining becomes thick and spongy to nourish the developing embryo.
![]()
Question 20.
Assertion (A): The magnetic field produced by a current-carrying solenoid is independent of its length and cross-sectional area.
Reason (R): The magnetic field inside the solenoid has a variable value.
Answer:
(c) A is true but R is false.
The magnetic field is independent of length and the area of the solenoid, it only depends on the number of turns and current flowing through the solenoid. It is uniform inside the solenoid.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
Give a test that can be used to differentiate between butter and cooking oil.
Answer:
Butter contains saturated compounds while cooking oil contains unsaturated compounds. Since unsaturated compounds are oxidized by alk. KMnO4 with the disappearance of its pink colour. Therefore, when cooking oil is treated with a few drops of alk. KMnO4, the pink colour of KMnO4 disappears. With butter, however, the pink colour of KMnO4 does not disappear. (2)
Question 22.
Write a short note on male and female chromosomes.
Or
How is the sex of a child determined in human beings?
Answer:
(i) Autosomes: These chromosomes are responsible for the determination of physical characters. Both males and females contain the same types of autosomes. In both males and females, 22 pairs of autosomes are present. (1)
(ii) Sex chromosomes or Heterosomes: These are responsible for sex determination in human beings. These are also known as heterosomes. These are of different types male and female. This is the 23rd pair of chromosomes in humans. In females, this pair of chromosomes is represented as XX, and in males it is XY (1)
Or
A male gamete carries either one X or one Y chromosome, while a female gamete carries only X chromosomes. Therefore, the sex of the child depends upon what happens during fertilization.
(i) If a sperm carrying an X-chromosome fertilizes the egg, the child born will be a female (XX). (1)
(ii) If a sperm carrying a Y-chromosome fertilizes the egg, the child born will be a male (XY). (1)
Question 23.
Draw a diagram showing the correct positions of the pancreas, thyroid gland, pituitary gland, and adrenal gland in human beings.
Answer:
(a) The pituitary gland (master gland) is present at the base of the brain.
(b) The thyroid gland is present just below the neck.
(c) The Pancreas Is found just below the stomach.
(d) Adrenal glands are present on the top of each kidney. (\(\frac{1}{2}\) × 4)
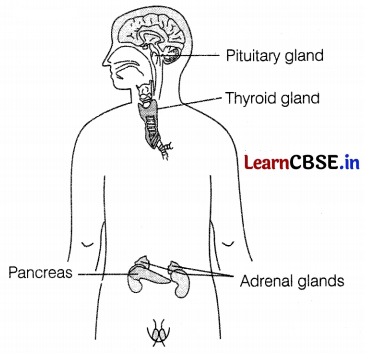
Question 24.
A student-focussed the image of a candle flame on a white screen by placing the flame at various distances from a convex lens. He noted his observation in the following table.
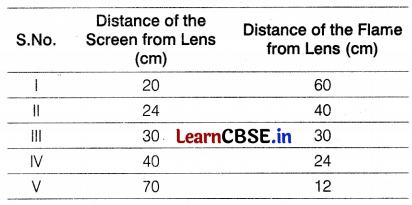
Analyze the above table and give the answers to the following questions.
(i) What is the focal length of a convex lens?
(ii) Which set of observations is incorrect and why?
Answer:
(i) We know that when the object is placed at 2F,
the distance of an object from the lens = distance of the image from the lens.
Therefore, from III observation, the radius of curvature R = 30 cm
Thus, focal length, f = \(\frac{R}{2}=\frac{30}{2}\) = 15 cm (1)
(ii) The last observation is incorrect because when an object is placed at a distance less than 15 cm away from a convex lens, we will have a virtual image, which cannot be taken on screen. (1)
![]()
Question 25.
What is meant by electric current? Write its SI unit. Calculate the amount of charge that flows through a conductor when a current of 5A flows through it for 2 min.
Or
AB is a current-carrying conductor in the plane of the paper as shown in the figure. What are the directions of magnetic fields produced by it at points P and Q?
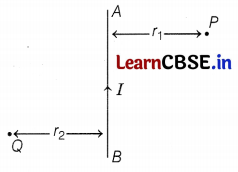
Given r1 > r2, when will the strength of the magnetic field be larger?
Answer:
Electric current is defined as the rate of flow of electric charge through any cross-section of a conductor.
SI unit of electric current is ampere (A).
Given, I = 5 A, t = 2 min = 2 × 60 s = 120 s, q =?
We know that, charge, q = I × t = 5 × 120 = 600 C
Thus, the amount of charge flowing through the conductor is 600 C. (2)
Or
According to the right-hand thumb rule, the magnetic field at P is into the plane of the paper, and at Q, it is out of the plane of the paper.
The strength of the magnetic field at Q will be larger as
strength of the field ∝ \(\frac{1}{r \text { (distance) }}\) (2)
Question 26.
How are water and minerals transported in plants?
Answer:
Water and minerals are transported through the xylem in plants. The cells in roots that are in contact with soil actively take up ions, creating a difference in concentration of ions between the cells sap of roots and soil water. Water moves into the roots to eliminate this difference of concentration forming a steady movement of water in the root xylem. This creates a column of water that is steadily pushed upwards. Loss of water from leaves creates a suction that pulls water from the xylem of the roots to the aerial parts of the body.
Section C
Questions No. 27 to 33 are short answer questions.
Question 27.
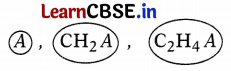
Define the given series. State any two characteristics of this series.
Or
Choose the kind of chemical bonding (ionic bond, covalent bond, both ionic and covalent bonds) present in the following compounds. Potassium chloride, magnesium oxide, sulphuric acid, ammonium hydroxide, zinc sulphide, and phosphorus trichloride (PCl3).
Answer:
A series of similarly constituted compounds in which the members present have the same functional group and similar chemical properties and any two successive members in a particular series differ in their molecular formula by a -CH2 unit, is called a homologous series. (1)
Characteristics of homologous series:
(a) The molecular formulae of any two successive members of a homologous series differ by -CH2– unit.
(b) There is a regular gradation in the physical properties of members of a homologous series. (2)
Or

Question 28.
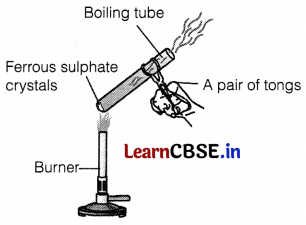
(a) Identify the gases that evolved.
(b) Name the type of reaction shown in the figure.
(c) What is the formula of crystalline ferrous sulphate? Write the equation involved.
Answer:
(a) SO2 and SO3 gases are evolved. (1)
(b) Thermal decomposition reaction occurs here. (1)
(c) The formula of crystalline ferrous sulphate is FeSO4. 7H2O.
Equation involved is
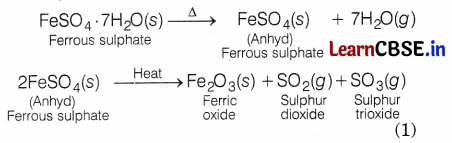
Question 29.
How does the embryo get nourishment inside the mother’s body?
Answer:
The embryo gets nutrition from the mother’s blood with the help of a special tissue called the placenta. This is a disc-like tissue that develops between the uterine wall and the embryo. As the mother eats, the food passes through the digestive system where the food breaks down into small absorbable forms. These nutrients travel through the mother’s bloodstream and get exchanged with the bloodstream of the fetus through the placenta. (3)
![]()
Question 30.
Observe the following cross between tall plants having round seeds and dwarf plants having wrinkled seeds. The individuals obtained in the F1 generation were thereafter self-crossed.
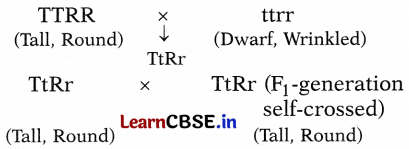
(i) What would be the phenotypes of the individuals obtained in the F2 generation? Give their ratios.
(ii) Why do you think all the individuals of the F1 generation were tall with round seeds?
Answer:
(i) Phenotypic ratio of the individuals in F2-generation.
Parents:
TtRr × TtRr (F1-generation self-crossing)
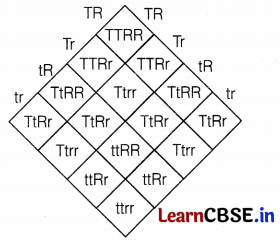
In F2-generation, the phenotypes of the individuals obtained would be
Tall and Round = 9;
Tall and Wrinkled = 3;
Dwarf and Round = 3;
Dwarf and Wrinkled = 1
Thus, the ratio is 9 : 3 : 3 : 1. (2)
(ii) The appearance of all tall plants with round seeds in the Frgeneration shows that the tallness and round-shaped seeds are dominant traits over the dwarfness and wrinkled shape of the seeds. (1)
Question 31.
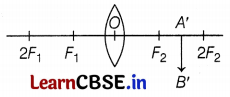
The above figure shows an incomplete ray diagram of an object, where the image A’ B’ is formed after refraction. All the rays parallel to the principal axis pass through the principal focus of the lens. O is the optical center of the lens.
(a) Based on the text and the data given in the above paragraph, what is the position of the object AB in front of the lens?
(b) What will be the size of image A B’ concerning the size of the object AB?
(c) What is the sign of linear magnification produced by the lens?
Answer:
(a)
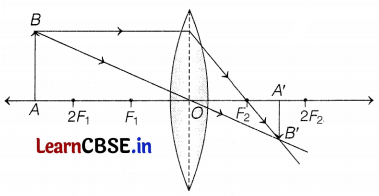
The position of object AB would have been beyond 2F1. (1)
(b) The size of the image would have been smaller than the size of the object. (1)
(c) Magnification (M) = \(=\frac{\text { Height of image }\left(h_1\right)}{\text { Height of object }\left(h_0\right)}\)
∴ Sign = \(\frac{-v e}{+v e}\) = -ve
(∵ Height of image below the principal axis = -ve and height of object above the principal axis = +ve)
Hence, the magnification produced by the above lens is negative. (1)
Question 32.
An electric lamp of 100 Ω, a toaster of 50 Ω, and a water filter of resistance 500 Ω are connected in parallel to a 220 V source.
(a) What is the resistance of an electric iron connected to the same source that takes as much current as all three appliances?
(b) What is the current through it?
Answer:
Let resistance of lamp, R1 = 100 Ω
Resistance of toaster, R2 = 50 Ω
Resistance of filter, R3 = 500 Ω
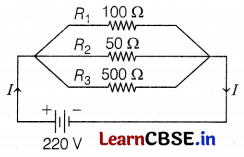
(a) Net resistance, \(\frac{1}{R}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\)
[∵ R1, R2 and R3 are connected in parallel]
\(\frac{1}{R}=\frac{1}{100}+\frac{1}{50}+\frac{1}{500}=\frac{16}{500}\)
or R = \(\frac{500}{16}\) = 31.25 Ω
(b) The resistance of iron to take the same current as much current drawn by all the appliances should be 31.25 Ω.
Current through circuit, I = \(\frac{V}{R}=\frac{220}{31.25}\) = 7.04 A
Thus, the current through iron is 7.04 A. (3)
![]()
Question 33.
Justify.
(a) Two circular coils A and B are placed close to each other. If the current in coil A is changed, will some current be induced in the coil B? Give a reason.
(b) When magnetic field lines are drawn around a current-carrying circular loop, it has been observed that they are close to its axis. But these lines keep on diverging as we move away from the center. Explain this observation.
Answer:
(a) When current in coil A is changed, a changing magnetic field is set up around it. This changing magnetic field also links with coil 6 and hence, some current will be induced in coil 6. (1\(\frac{1}{2}\))
(b) We know that the magnetic field is stronger near the current-carrying conductor and weaker as we move away from the conductor. In the case of a current-carrying circular loop, the magnetic field is stronger near the periphery but weaker near the center of the loop. Due to this, the magnetic field lines appear as straight lines near the center. As we move towards the periphery of the circular loop, the magnetic field lines appear to be diverging, so that they can be circular around the wire of the loop. (1\(\frac{1}{2}\))
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
Arshdeep took two iron salts. First iron salt A reacts with NaOH to form a green precipitate. Another iron salt B reacts with NaOH to form a brown precipitate. Identify the iron salts, A and B alongwith their colours, and write the reactions involved.
Or
(a) A student mixes sodium sulphate powder in barium chloride. What change would the student observe in mixing the two powders? Justify your answer and explain how he can obtain the desired change. (2)
(b) List two observations you would record in your 30 minutes after adding iron filings to copper sulphate solution.
Answer:
(a) Iron salts A and B are ferrous sulphate and ferric sulphate respectively. (1)
(b) The ferrous salt is generally green coloured, whereas ferric salts are brown. (1)
(c) When a ferrous salt solution is treated with a solution of sodium hydroxide, a greenish precipitate of ferrous hydroxide is obtained. Hence, salt A is FeSO4.
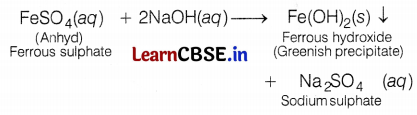
When a ferric salt solution is treated with a 1 solution of sodium hydroxide, a brown precipitate of ferric hydroxide is produced. Hence, salt B is Fe2(SO4)3.
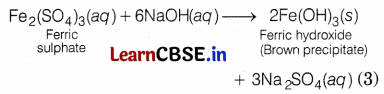
Or
(a) When the student mixes sodium sulphate powder in barium chloride in a dry state, no change will be observed. But when he dissolved them in water, barium sulphate precipitated out and sodium chloride remained in the solution. This is called a double displacement reaction. (2\(\frac{1}{2}\))
(b) The colour of copper sulphate solution changes when an iron filling is dipped in it because iron being more reactive than copper, displaces copper metal from aqueous copper sulphate solution. Thus, the blue colour of copper sulphate fades away to give a green colour solution of ferrous sulphate.
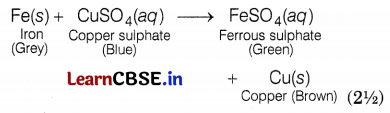
Question 35.
(a) Write the equations and steps involved in photosynthesis. In which way are the steps of photosynthesis different in desert plants?
(b) What is ‘translocation’? Why is it essential for plants? Where in plants are the following synthesized?
(i) Sugar
(ii) Hormones
Or
Give reasons:
(a) The placenta is essential for fetal development.
(b) Blocking of vas deferens prevents pregnancy.
(c) Wind acts as a pollinating agent.
(d) Use of condoms prevents pregnancy.
(e) Blocking of Fallopian tubes prevents pregnancy.
Answer:

Three events that occur during photosynthesis are as follows.
(i) Absorption of light energy by chlorophyll.
(ii) Conversion of light energy into chemical energy and splitting of water into hydrogen and oxygen.
(iii) Reduction of CO2 into carbohydrates.
Desert plants take up CO2 at night and prepare intermediate molecule, which is absorbed by the chlorophyll during the day to make carbohydrates. (2\(\frac{1}{2}\))
(b) Translocation: The transport of food prepared in the leaves to other parts of the plants is known as translocation. The plants need to supply food to all parts of the plants. Food is needed for producing energy, which in turn is required by all parts of the plants to perform their activities.
(i) Sugars are synthesized in leaves and then transported to storage organs like roots, fruits, and seeds.
(ii) Plant hormones are synthesized at the tips of the stems and roots. (2\(\frac{1}{2}\))
Or
(a) Placenta is essential for foetal development because it helps in the nutrition, respiration, excretion, etc., of the fetus through the maternal supply.
(b) Blocking of vas deferens prevents the passage of sperm, hence, there is no fertilization so it prevents pregnancy.
(c) Wind acts as a pollinating agent because it helps in the transfer of light-weighted pollen grains from the anther to the stigma of a flower.
(d) Condoms prevent the entry of sperm into the vagina, hence preventing pregnancy.
(e) If the Fallopian tube is blocked, sperm and egg do not meet and fuse and fertilization does not take place. (5)
![]()
Question 36.
(a) Define the following terms in the context of spherical mirrors.
(i) Pole
(ii) Centre of curvature
(iii) Principal axis
(iv) Principal focus
(b) Draw a ray diagram to show the principal focus of a
(i) concave mirror and
(ii) convex mirror
(c) Consider the following diagram in which M is a mirror and P is an object 1 and Q is its magnified image formed by the mirror.
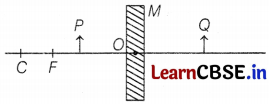
State the type of the mirror M and one characteristic property of the image Q.
Or
A 6 cm tall object is placed perpendicular to the principal axis of a convex lens of focal length 25 cm. The distance of the object from the lens is 40 cm. By calculation determine
(a) the position and
(b) the size of the image formed.
(i) The pole of the spherical mirror is the mid-point of its reflecting surface.
(ii) Centre of Curvature of a spherical mirror is the center of the imaginary sphere of which, the mirror is a part,
(iii) Principal Axis of a spherical mirror is the line joining the pole and center of curvature.
(iv) The Principal Focus of a concave mirror is a point on the principal axis of the mirror at which the light rays coming parallel to the principal axis, after reflecting meet. (2)
(b)
(c) The given diagram in the question can be redrawn as
So, M is a concave mirror and the image Q formed is enlarged. (1)
Or
Given, the height of the object, h0 = 6 cm
The focal length of the lens, f = 25 cm
Distance of object, u = -40 cm

Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
A student decided to observe the conductive nature of ionic compounds in, different physical states. He took two samples of compounds. In the first case, solid common salt was taken to make a circuit in which the bulb did not glow. Secondly, he dissolved the same salt in water and completed the circuit as given in the figure. In this case, the bulb glows.
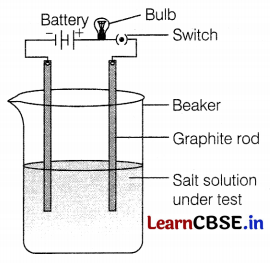
(a) What conclusion can you draw from this activity?
(b) Why does salt conduct electricity in an aqueous solution but not in the solid state?
Or
If we take the sugar solution in water and test the conductivity, will the bulb glow?
Answer:
(a) We can conclude that ionic compounds such as common salt conducts electricity in aqueous solution only. (2)
(b) Common salt (sodium chloride) conducts electricity in a molten state because of the presence of free ions in the solution while in the solid state, there are no free ions. (2)
Or
No, the bulbs will not glow. Sugar solution is not an ionic compound because it does not give free ions in the solution. Hence, there will be no electrical conductivity. (2)
Question 38.
Sheenu took three different types of solid wastes R, S, and T. She buried them under the soil in a pit, as she wanted to study their rate of decomposition. Her findings are shown in the given graph.
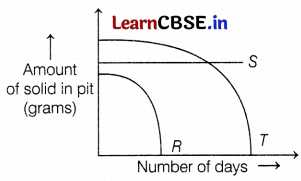
(a) Among R, S, and T, which of the following wastes will not decompose at all?
(b) What are the safe methods of disposal of the non-biodegradable waste?
(c) What type of solid waste comes under the category of R?
Or
What are the other artificial methods of waste disposal?
Answer:
(a) S is the solid waste that will not decompose at all. According to the graph, there is no change in the amount of S waste. So, no decomposition takes place in any number of days. Hence, it is a non-biodegradable waste. (1)
(b) Non-biodegradable waste can be decomposed by recycling or by dumping it underground into landfills. (1)
(c) Solid waste R completely decomposes in very few days which means it is an easily decomposable biodegradable waste.
e.g. Cow dung, fruit pulp, etc., comes under this category. (2)
Or
Incineration is a method of waste disposal in which the burning of substances takes place at high temperatures to form ash. It is used to dispose of hospital or harmful wastes of biomedical industries. (2)
![]()
Question 39.
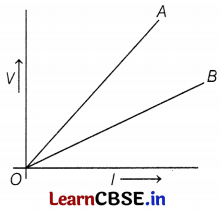
The V-I graphs for two wires A and B are shown in the figure above. This graph is based on Ohm’s law. The slope of the V-I graph gives resistance to the conductor.
(a) If wires A and B are of the same materials having equal length, then which wire is thicker?
(b) If wires A and B are of the same materials and have the same diameter, then which wire is shorter?
(c) If both wires are of the same length and the same diameter, then which wire has maximum resistivity?
Or
If the temperature of wire A is more than the temperature of wire B, then which one has the least resistance?
Answer:
(a) As wires A and B are of the same material, then ρA = ρB
Also, IA = IB
Now, the slope of the V-I graph = R = ρ.\(\frac{1}{A}\)
⇒ A = ρ.\(\frac{1}{R}\)
⇒ A ∝ \(\frac{1}{R}\)
⇒ d ∝ \(\frac{1}{\sqrt{R}}\)
As, the slope of A > slope of B
∴ RA > RB ⇒ dA < dB Hence, wire B is thicker wire. (1)
(b) Given, wires A and B are of the same material,
∴ ρA = ρB
Also, dA = dB
As, I = \(A \cdot \frac{R}{\rho}\)
⇒ I ∝ R
∵ RA > RBB
⇒ IA > IB
Hence, wire B is a shorter wire. (1)
(c) The greater the slope of the V-I graph, the greater the resistance of a given metallic wire. In the given graph, wire A has a greater slope than B. Hence, wire A has greater resistance.
For the wires of same length and thickness, resistance depends on the nature of the material of the wire,
i.e. RA = ρA \(\frac{l}{A}\) and RB = ρB \(\frac{l}{A}\)
⇒ \(\frac{R_A}{R_B}=\frac{\rho_A}{\rho_B}\)
⇒ R ∝ ρ
Hence, the wire A is made of a material of high resistivity. (2)
Or
Given, TA > TB
As, the resistance of a wire is directly proportional to the temperature, i.e. R ∝ T
Hence, wire B has the least resistance. (2)