Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 12 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 12 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
When NaOH and HCl are mixed in equal molar quantities, the result is
(a) the formation of salt + H2O
(b) the formation of salt + H2 (g)
(c) the formation of salt + O2 (g)
(d) the formation of salt + N2
Answer:
(a) the formation of salt + H2O
When NaOH and HCl are mixed in equal molar quantities, the acid-base reaction takes place and we get salt (NaCl) and water.
NaOH + HCl → NaCl (Salt) + H2O
Question 2.
Mineral acids are stronger acids than carboxylic acids because
(i) Mineral acids are completely ionized.
(ii) Carboxylic acids are completely ionized.
(iii) Mineral acids are partially ionized.
(iv) Carboxylic acids are partially ionized.
(a) (i) and (iv)
(b) (ii) and (iii)
(c) (i) and (ii)
(d) (iii) and (iv)
Answer:
(a) (i) and (iv)
Mineral acids like HCl are completely ionized in solvents like water whereas carboxylic acids such as CH3COOH are only partially ionized.
![]()
Question 3.
Four metals Zn, Fe, Cu, and Al are taken and added to the following solutions one by one. The results obtained are tabulated as given below. Based on the data given which of the following elements is most reactive?
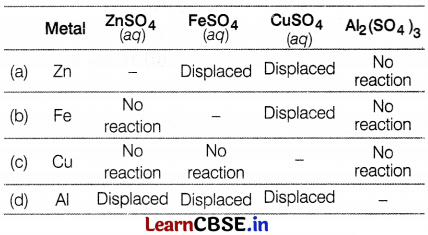
Answer:

From the results given in the table, Al is the most reactive among others because it shows displacement reactions in three solutions, Zn shows, displacement reactions in two solutions and iron shows displacement in one solution. The least reactive metal is copper as it does not react with any solution.
Question 4.
A coating of zinc will be observed in
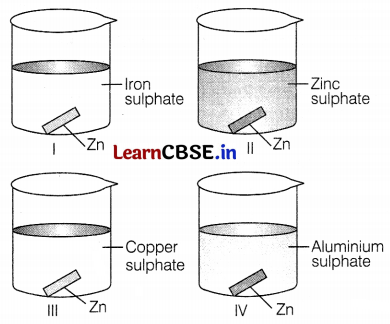
(a) I and III
(b) Only I
(c) Only II
(d) III and IV
Answer:
(a) I and III
Zn, being more reactive than iron and copper, therefore displaces the iron and copper from their solutions. Hence, Fe and Cu will form a coating on zinc due to displacement reaction.
Question 5.
Which of the following is not an heteroatom CH2—O—CH2—CH2(Br) are
(a) oxygen
(b) carbon
(c) hydrogen
(d) bromine
Answer:
(b) carbon
Oxygen (O). chlorine (Cl) and bromine (Br) are heteroatoms. Please remember that apart from C and H atoms, all other atoms present in an organic compound are hetero atoms.
Question 6.
Amit checked the odour of onion juice and then he added a few drops of onion juice in dil. HCl and in dil. NaOH solution in separate test tubes. Which of the following observations he would not observe?
(a) Odour of onion juice in sodium hydroxide vanished.
(b) Odour of onion juice in dilute hydrochloric acid remained the same.
(c) Onion acts as an olfactory indicator.
(d) Odour of onion juice in dilute HCl vanished.
Answer:
(d) Odour of onion juice in dilute HCl vanished.
(a), (b) and (c) observations will be observed by him. Onion is an olfactory indicator whose odour changes or vanishes in a basic medium and remains unchanged in an acidic medium.
Question 7.
Carbon forms four covalent bonds by sharing its four valence electrons with four univalent atoms, e.g. hydrogen. After the formation of four bonds, carbon attains the electronic configuration of
(a) helium
(b) neon
(c) argon
(d) krypton
Answer:
(b) neon
The compound formed is methane (CH4). In this, the carbon atom has a complete octet and configuration of neon which is a noble gas element.
![]()
Question 8.
The sequence of anaerobic respiration in our muscle cells during heavy exercise is

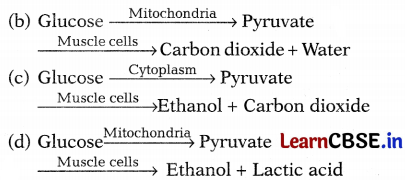
Answer:
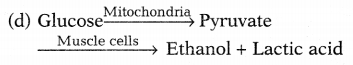
Conversion of glucose into pyruvate takes place in mitochondria. This pyruvate converts to ethanol and lactic acid in human muscle cells. This causes muscle cramps after prolonged exercise.
Question 9.
Consider the following diagram that represents the reproductive system in the human male.
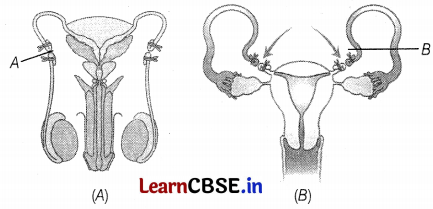
The blockages shown at A and B would most likely interfere with the ability to
(a) transport gametes
(b) produce mature gametes
(c) eliminate waste products through the urethra
(d) express secondary sex characters
Answer:
(a) transport gametes
The blockages shown at A and B would likely interfere with the ability to transport gametes. Figure A shows the vasectomy in which vas deferens are removed or tied up through a small incision on the scrotum. Figure (B) shows tubectomy, in which small part of the Fallopian tube is removed or tied up through a small incision in the abdomen or through the vagina.
Question 10.
Which of the following substances breaks down starch?
(a) Lipase
(b) Amylase
(c) Complex sugars
(d) Trypsin
Answer:
(b) Amylase
Amylase is a carbohydrate-digesting enzyme, which is responsible for the breakdown of starch into smaller units.
Question 11.
Which one of the following sets of options correctly depicts reproduction in Amoeba and yeast, respectively?
(a) Budding and binary fission
(b) Binary fission and budding
(c) Multiple fission and binary fission
(d) Fragmentation and grafting
Answer:
(b) Binary fission and budding
Binary fission in Amoeba and budding in yeast.
Question 12.
Which of the following compounds do not cause the thinning of the ozone layer?
(a) Chlorofluorocarbon
(b) Carbon dioxide
(c) Nitrogen oxide
(d) Methane
Answer:
(d) Methane
Methane is not responsible for the depletion of the ozone layer. The primary cause of ozone depletion is the presence of chlorine-containing source gases (primarily CFCs and related halocarbons), CO2, and oxides of nitrogen.
Question 13.
In circuit
(a) ammeter and voltmeter both are connected in series.
(b) ammeter is connected in parallel and the voltmeter in series.
(c) ammeter is connected in series and voltmeter in parallel.
(d) ammeter and voltmeter both are connected in parallel.
Answer:
(c) ammeter is connected in series and voltmeter in parallel.
In any circuit, the ammeter should be connected in series and the voltmeter should be connected in parallel.
Question 14.
The splitting of white light into seven colours on passing through a glass prism is due to
(a) dispersion
(b) refraction
(c) scattering
(d) reflection
Answer:
(a) dispersion
The splitting of white light into its component colours on passing through glass is called dispersion.
![]()
Question 15.
At the time of puberty, both boys and girls show lots of changes in appearance. Select the hormone responsible for these changes in boys.
(a) Oestrogen
(b) Adrenaline
(c) Testosterone
(d) Thyroxine
Answer:
(c) Testosterone
Testosterone is responsible for changes in boys during puberty.
Question 16.
Which structure among the following options forms villi towards the embryo, creating a large surface area for the exchange of nutrients, gases, etc. between the mother and the embryo?
(a) Placenta
(b) Lacteals
(c) Oviduct
(d) Cervix
Answer:
(a) Placenta
The placenta forms villi towards the embryo for the exchange of nutrients, gases, and waste materials between the mother and embryo.
Directions (Q.Nos. 17-20) These consist of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): Tungsten metal is selected for making filaments of incandescent lamps.
Reason (R): Tungsten has a high melting point.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
Tungsten has a high melting point. Therefore, it is used for making filament of bulbs.
Question 18.
Assertion (A): A network of food chains existing together in an ecosystem is known as a food web.
Reason (R): An animal like a kite cannot be a part of a food web.
Answer:
(c) A is true, but R is false.
In the food web, different food chains are interconnected. Each food chain consists of different trophic levels, i.e. producers, consumers, and detrivores. So, kites can also be a part of the food web.
Question 19.
Assertion (A): In woody plants, gaseous exchange occurs through lenticels.
Reason (R): Lenticels are specialized cells found along with stomata on the stem of woody plants.
Answer:
(c) (A) is true, but (R) is false.
In woody plants, gaseous exchange occurs through the small pores found on stems called lenticels. Stomata on the stem aid in gaseous exchange, in herbaceous plants.
![]()
Question 20.
Assertion (A): A fuse is connected in series with the circuit.
Reason (R): Fans, lamps, etc, are connected in parallel in household wiring.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
A fuse is connected in series with the circuit to protect the circuit. Fans, lamps, etc. are connected in parallel in household wiring so that if any of them fuses, then it does not affect the working of other appliances.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
Plaster of Paris is used to make sculptures and metal casting is used as decorative material in buildings. It should be generally stored in moisture-proof containers. Explain, why support your response with the help of a chemical equation.
Answer:
Plaster of Paris (POP) is chemically calcium sulphate hemihydrate (CaSO4 . \(\frac{1}{2}\)H2O). when it comes in contact with water it sets into a hard solid mass, called gypsum.

To prevent this, POP must be stored in moisture-proof containers. (2)
Question 22.
What could be the reasons for adopting contraceptive methods?
Answer:
The reasons for adopting contraceptive methods are as follows:
- Protection from STDs.
- Restricting the number of children (population control).
- Maintaining a sufficient gap between successive births.
- Enjoying a good reproductive health. (\(\frac{1}{2}\) × 4)
Question 23.
In the food chain given below, which organisms will be the least in numbers?
Algae – Protozoan – Small fish – Large fish
Or
Why are crop fields known as artificial ecosystems?
Answer:
In the given food chain, i.e,
Algae → Protozoans → Small fish → Large fish
The large fish (tertiary consumer/top carnivore) will have the least number of organisms. In any food chain, there are generally a greater number of organisms at the lower trophic levels. Thus, the producers have the greatest numbers and top carnivores have the least numbers. (2)
Or
Artificial ecosystems are those ecosystems that are modified and managed by human beings. Crop fields are man-made. Here plants do not grow naturally rather most of the plants are grown by humans according to the season, type of soil, etc. Crop fields are not like wild forest area, which is left to the care of nature and can sustain itself. In crop fields, the land is managed, the soil is prepared for sowing seeds, then irrigated, and further progress is also kept under observation to get a good yield. This is why crop fields are known as artificial ecosystems. (2)
Question 24.
The refractive indices of the three media are given below.
| Medium | Refractive Index |
| A | 1.6 |
| B | 1.8 |
| C | 1.5 |
A ray of light is travelling from A and B and another ray is travelling from B to C.
(a) In which of the two cases the refracted ray bends towards the normal?
(b) In which case does the speed of light increase in the second medium?
Give a reason for your answer.
Answer:
(a) When light travels from an optically rarer medium to an optically denser medium it moves towards the normal. Since nB > nA, the light ray will bend towards the normal on passing from medium A to B. (1)
(b) The speed of the light will increase when the light travels from B to C, since nC < nB and v = (c/n), the speed of the light ray will increase in the second medium. (1)
Question 25.
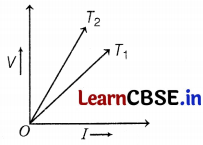
The voltage-current (V-I) graph of a metallic circuit at two different temperatures T1 and T2 is shown in the figure. Which of the two temperatures is higher and why?
Or
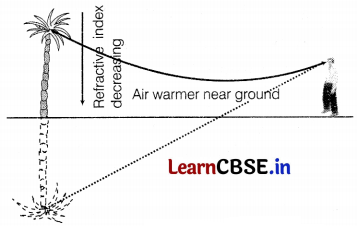
Explain the phenomenon and its cause in the above diagram. What will happen when light travels from hot air to cold air?
Answer:
Resistance is equal to the slope of the V-I graph. Here, slope of graph for temperature T2 is higher, so resistance for temperature T2 is higher, As R ∝ T, T2 > T1 (2)
Or
The phenomenon observed in the given figure is a mirage. A mirage is formed due to the bending of light because of the temperature difference between different layers of air. Due to this the refractive index changes with height. When light travels from hot air (rarer) to cold air (denser), then it bends towards the normal. (2)
![]()
Question 26.
If you have to go to a village during an AIDS prevention program, what points will you explain to the villagers?
Answer:
A few points for the prevention of AIDS are as follows.
- Use condoms while having sex.
- Pregnant women should be checked for pre-existing HIV.
- Use a disposable syringe.
- Blood transfusion should be done carefully. (\(\frac{1}{2}\) × 4)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
What is a homologous series of substances? Explain, why carbon forms compounds mainly by covalent bonds.
Or
Name the type of carbon compounds that can be hydrogenated. With the help of a suitable example explain the process of hydrogenation.
Answer:
A series of similarly constituted compounds in which the members present have the same functional group and similar chemical properties and any two successive members in a particular series differ in their molecular formula by (—CH2) unit is called a homologous series, e.g. CH4, C2H8, C3H8 are the members of alkane family. (2)
Carbon has 4 electrons in its outermost shell and needs to gain or lose 4 electrons to attain a noble gas configuration. Losing or gaining 4 electrons is not possible due to energy considerations, hence it shares electrons to form covalent bonds. (1)
Or
Only unsaturated hydrocarbons, i.e. alkenes and alkynes can be hydrogenated.
e.g. In the presence of a catalyst Ni/Pd, ethyne is hydrogenated into ethane.
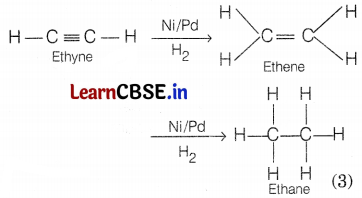
Question 28.
Study the given diagram and answer the following questions.
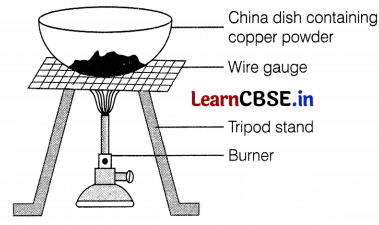
(a) Write the chemical reaction involved in the process.
(b) Mention the colour of copper powder and the substance formed after heating it.
(c) How can we reverse the above reaction? Write the equation for the reverse reaction and state the substance that undergoes oxidation and the substance that undergoes reduction.
Answer:
(a) Copper powder upon heating with oxygen (present in air) to form cupric oxide or copper (II) oxide.
2Cu (s) + O2 (g) → 2CuO (s) (Cupric oxide) (1)
(b) (i) Copper powder is brownish.
(ii) The substance formed after heating is cupric oxide. It has black colour. (1)
(c) The reaction can be reversed by heating cupric oxide in the presence of hydrogen.
CuO (s) + H2 (g) → Cu (s) + H2O (g)
In this reaction, H2 undergoes oxidation to H2O, and CuO is reduced to Cu. (1)
Question 29.
Mention the pathway of urine starting from the organ of its formation. Name four substances that are reabsorbed from the initial filtrate in the tubular part of the nephron.
Answer:
The pathway of urine starting from the organ of its formation is as follows:
Kidneys → Ureters → Urinary bladder → Urethra
- Kidney: It contains millions of complex nephrons and filters about 170 to 200 liters of blood. It produces 1-1.8 liters of urine daily.
- Ureters: These carry urine from the kidneys to the urinary bladder.
- Urinary bladder: This is a muscular sac-like structure where urine is stored until released.
- Urethra: This is a short muscular tube that carries urine from the urinary bladder to the outside of the body. (2)
The four substances reabsorbed from the initial filtrate are
- Amino acid
- Glucose
- Salts
- Major amount of water (1)
![]()
Question 30.
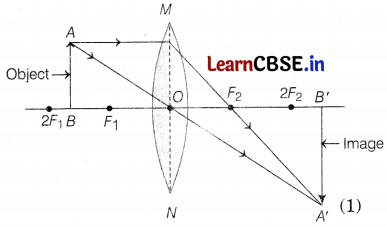
Manoj has taken a thin converging lens to form a real magnified image of an object in front of it.
(a) Write the position of the object.
(b) Draw a ray diagram to show the image formation.
(c) If the power of the lens is +2D, then what will be the focal length of the lens?
Answer:
(a) Object is placed between F and 2F.
(b) The ray diagram is as follows:
(c) Given, P = +2D [P = Power of lens]
Since power is positive, the lens is a convex lens.
∴ Focal length, f = \(\frac{1}{P}=\frac{1}{2}\) = 0.5 m = 50 cm (1)
Question 31.
A solenoid is taken. It is allowed by the flow of an electric current. Raju observes the pattern of the magnetic field due to the current-carrying solenoid. Answer the following questions based on his observations.
(a) State the factors on which the strength of an electromagnet depends.
(b) Why did the television get a dark patch when the magnet was brought near its screen?
Answer:
(a) Factors affecting the strength of an electromagnet:
- The number of turns in the coil If the number of turns in the coil is increased, the strength of the electromagnet increases.
- The current flowing in the coil If the current in the coil is increased, the strength of the electromagnet increases.
- The length of the air gap between the poles If the length of the air gap between the poles of an electromagnet, decreases then its strength increases. (2)
(b) The television has an electromagnet installed in it. When a magnet is brought closer to the screen, the two magnetic field lines interfere and spoil its functioning. (1)
Question 32.
A spherical mirror produces an image of magnification -1 on a screen placed at a distance of 50 cm from the mirror.
(a) Write the type of mirror.
(b) Find the distance of the image from the object.
Answer:
(a) The mirror is a concave.
(b) Given, m = -1, u = -50 cm
∴ Magnification, m = \(\frac{-v}{u}\)
⇒ -1 = \(\frac{-v}{-50}\)
⇒ v = -50 cm (1)
Therefore, the image is real and inverted and of the same size as that of the object.
Here, the object is placed at the center of curvature, so the object distance is equal to the image distance, since, the screen is placed at a distance of 50 cm from the mirror, therefore object distance = image distance = 50 cm.
The distance of the image from an object is zero, i.e. the object is formed at 2f. (2)
Question 33.
A motorcycle rider without a helmet met with an accident and suffered a spinal cord injury. In this case, which signals will get disrupted and why?
Answer:
In case of a spinal cord injury, signals for reflex action and involuntary action will get disturbed. Reflex action is monitored and controlled through the spinal cord of the nervous system and not by the brain. Nerves from all over the body meet in a bundle in the spinal cord on their way to the brain. In case of any injury to the spinal cord, the signals coming from the nerves as well as signals coming to the receptors will be disturbed. (3)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
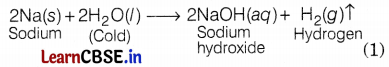
Manish took different metals and observed that all metals reacted differently with hot water, cold water, and steam. Explain the reactions of sodium, magnesium, and iron metals with cold water, hot water, and steam.
Or
(a) An element P reacts with water to form a compound Q, which is used in whitewashing—the compound Q on heating forms an oxide R which on treatment with water gives back B. Identify P, Q, and R and give the reactions involved.
(b) How photochemical reactions have played an important role in photography?
Answer:
Reactions of Na, Mg, and Fe metals with different types of water are shown below.
With cold water: Sodium reacts violently with water. The reaction is
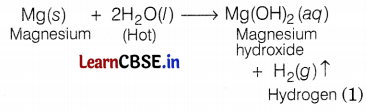
With hot water: Magnesium reacts with hot water to form magnesium hydroxide and hydrogen.
With steam: Iron does not react either with cold or hot water. It reacts with steam. The reactions are as follows:
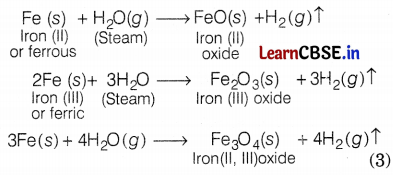
Or
(a) Element P is calcium (Ca), and when it reacts with water, it forms calcium hydroxide, Thus, compound Q is calcium hydroxide Ca(OH)2, which is used in whitewashing. (1)
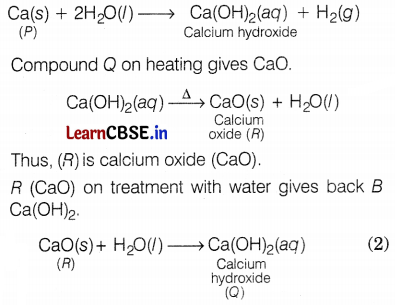
(b) A photographic film used in black and white photography is a celluloid film coated with silver chloride. Its work is based on the decomposition of silver chloride in the presence of sunlight.

Question 35.
(a) ‘Plants also perform chemical coordination’. Elaborate.
(b) Name various plant hormones. Also give their physiological effects on plant growth and development.
Or
(a) Explain how traits are controlled by genes only.
(b) Give an example in which both genes exist independently of each other in humans.
Answer:
(a) Plants also perform chemical coordination for various activities with the help of hormones. These are the chemical compounds released by stimulated cells that diffuse to various locations in plants performing different functions. These hormones produced by plants are also called phytohormones. (2)
(b) Different types of hormones produced by plants are Auxin, Gibberellins, cytokines, Abscisic acid, and Ethylene. (3)
| Plant Hormone | Physiological Effect |
| Auxin | Synthesized in the young tip of roots and shoots. It diffuses towards the shady side of the plant, which stimulates the cells to grow longer, resulting in the bending of the shoot towards the light. |
| Promotes cell elongation and division. | |
| Plays an important role in the formation of roots and seedless fruits. | |
| Gibberellins | Helps in the growth of the stem and flower. |
| Helps in the germination of seeds. | |
| Cytokinins | Promote cell division and delay leaf aging. |
| Also, stimulates leaf expansion. | |
| Abscisic acid | Growth inhibitor |
| Reverses the growth-promoting effects of auxins and gibberellins. | |
| Promotes transverse growth. | |
| Ethylene | Essential for fruit ripening, promotes senescence and abscission of leaves. |
Or
(a) Plants have hormones that can trigger growth. If the hormone works efficiently, a lot of hormones will be made (i.e., tall plant).
If the gene for the enzyme has alteration, the enzyme will not be efficient. Hence, the amount of hormone produced will be less (i.e., small plant). This proves that the traits (characters) are controlled by genes only. (2)
(b) Both genes exist independently of each other in humans can be explained by the codominance phenomenon of blood groups. There are four types of blood groups A, B, AB, or 0, and controlled by genes IA, IB, and IO. The genes IA and IB show codominance because both express themselves independently as shown.
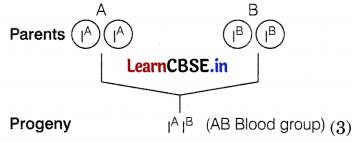
![]()
Question 36.
The diagram below shows a circuit in which 4Ω, 10Ω, and 20Ω resistors are connected in parallel combination. A voltmeter, an ammeter, and a 20V battery are also connected to the circuit. Calculate
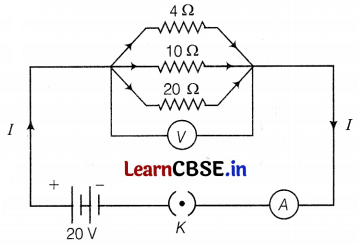
(a) The total effective resistance of the circuit.
(b) The current through each resistor.
(c) Write the Joule’s law of heating.
Or
Study the following circuit and answer the questions that follow.
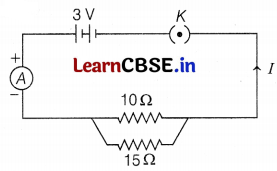
(a) State the type of combination of two resistors in the circuit.
(b) How much current is flowing through (i) 10Ω and (ii) 15Ω resistor?
(c) What is the ammeter reading?
Answer:
Given, R1 = 4Ω, R2 = 10Ω, and R3 = 20Ω, V = 20V
(a) Total effective resistance of the circuit,
\(\frac{1}{R_{\text {eff }}}=\frac{1}{R_1}+\frac{1}{R_2}+\frac{1}{R_3}\)
(Here, resistances are connected in parallel combination)
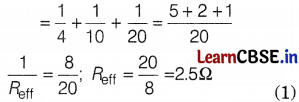
(b) Current through each resistor,

(c) According to Joule’s law of heating, the amount of heat produced in a resistor is
(i) directly proportional to the square of the current flowing through the resistor, i.e. H ∝ I2
(ii) directly proportional to the resistance of the resistor, i.e. H ∝ R
(iii) directly proportional to the time for which the current flows through the resistor.
i.e. H ∝ t ⇒ H = I2Rt (2)
Or
(a) Two resistors are in parallel combination. (1)
(b) Current through
(i) 10Ω resistor, I1 = \(\frac{V}{R}=\frac{3}{10}\) = 0.3A
(ii) 15Ω resistor, I2 = \(\frac{V}{R}=\frac{3}{15}\) = 0.2A (2)
(c) Ammeter reading = Total current flowing through the circuit
= 0.3 + 0.2
= 0.5A (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
The table given below shows the hints given by the quiz master in a quiz.
| Hints |
| (i) Compound ‘X’ is used in cough syrups and many tonics. |
| (ii) ‘Y’ is formed on heating ‘A’ in the presence of alk. KMnO4 |
| (iii) ‘A’ is also soluble in water in all proportions. |
Based on the above hints answer the following questions.
(a) Name the compound X. Write its chemical formula.
(b) Which gas is evolved when the compound X reacts with sodium? Write the chemical equation involved in the reaction of X with sodium.
Or
Complete the following equation for X and identify Y.
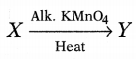
Answer:
(a) The compound X is ethanol as it is soluble in water in all proportions and used in cough syrups. The chemical formula is CH3CH2OH.
(b) Hydrogen gas is evolved when ethanol reacts with sodium.
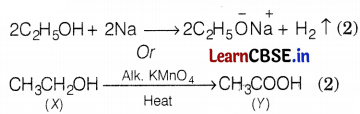
The compound ‘Y’ is ethanoic acid.
![]()
Question 38.
Priyanka has green eyes. On the other hand, her parents and brother have black eyes. Deepak (Priyanka’s husband) has black eyes. Deepak’s mother has green eyes and their father has black eyes.
(a) based on the above-given information, which eye colour is recessive and why?
(b) What is the genotype of the eye colour of Priyanka’s brother?
(c) What is the probability of Priyanka’s and Deepak’s child having green eyes?
Or
With the help of a cross, Priyanka’s brother can have 50% of the offspring with green eyes.
Answer:
(a) Green eye color is recessive based on the above-given information. As it is expressed only in homozygous conditions. (1)
(b) The genotype of Priyanka’s brother’s eye colour is CaBB or Bb. (1)
(c) Since Deepak’s mother has green eyes (bb) and father has black eyes (BB), so genotype of Deepak’s eyes (black) will be Bb. The Genotype of Priyanka’s eyes will be bb.
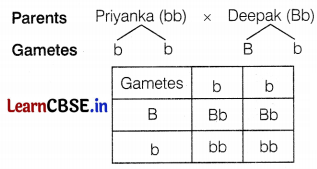
The probability that the offspring of Priyanka and Deepak will have green eyes is 50%. (2)
Or
Let the person marrying Priyanka’s brother be
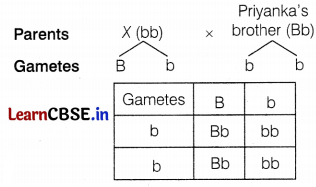
When Priyanka’s brother marries a green eye coloured girl, there will be a 50% chance of the offspring having green eyes. (2)
Question 39.
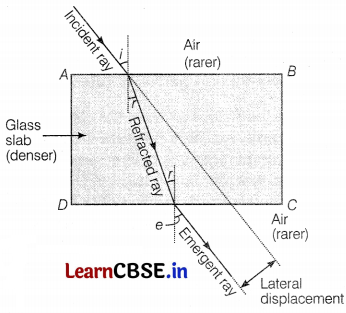
The above figure shows a glass slab at which an incident ray falls at an angle of incidence i. The emergent ray is coming from the glass slab at an angle of emergence e. The refractive index of the glass slab is 1.5. The speed of light in air is 3 × 108 m/s.
(a) Based on the text and data given in the above paragraph, what is the angle between incident ray and emergent ray?
(b) Which quantity remains constant when a light travels from air to a glass slab?
(c) What is the speed of light in the glass slab?
Or
What will happen when incident light falls normally to the surface of the glass slab?
Answer:
(a) As the incident ray and the emergent ray are parallel to each other. So, the angle between them is zero. (1)
(b) When the light travels from one medium to another medium, then the frequency of the light remains constant. (1)
(c) The speed of light is air, c = 3 × 108 m/s
The speed of light in the glass, v =?
Refractive index of glass, µg = 1.5
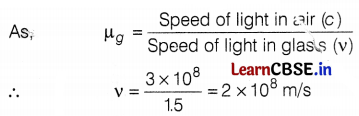
Or
When the incident ray falls normally to the surface of the glass slab, there is no bending of the ray of light, it goes straight without any deviation. (2)