Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 11 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 11 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Identify the colour of the gas that evolved in the following experiment.
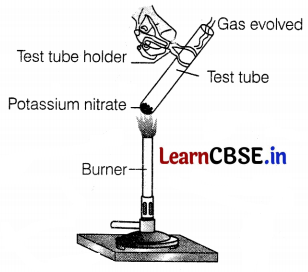
(a) Colourless
(b) Yellow
(c) Whitish grey
(d) Black
Answer:
(a) Colourless
When potassium nitrate is heated, then colourless gas is evolved, i.e. oxygen.

Question 2.
The ability of metals to be drawn into thin wire is known as
(a) ductility
(b) malleability
(c) sonorousity
(d) conductivity
Answer:
(a) ductility
These all are physical properties of metals, The ability of metals to be drawn into thin wire is known as ductility.
![]()
Question 3.
The graph given below represents the neutralization reaction. The pH of the solution changes as we add an excess base to an acid.
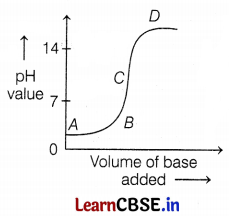
Which letter denotes the area of the graph where both base and salt are present?
(a) A
(b) B
(c) C
(d) D
Answer:
(d) D
The pH value of the base is above 7. So, in addition to the base, an acid neutralization reaction takes place.
Acid + Base → Salt + Water
In addition to the excess base, the pH of the solution rises above 7 due to the presence of base alongwith salt. Hence, D is the correct letter which represents both base and salt.
Question 4.
Which of the following is an example of an amorphous form of carbon?
(a) Wood
(b) Oil
(c) Chalk
(d) Coke
Answer:
(d) Coke
Coke is an example of an amorphous form of carbon that is obtained as a residue in the destructive distillation of coal.
Question 5.
Raj observed that after the reaction between hydrochloric acid and sodium hydroxide, sodium chloride and water are formed. Then, he searched for sodium chloride. Some of his statements about NaCl salt are given below.
I. It is also prepared by the chlor-alkali process.
II. It is a neutral salt, having a pH value = 7.
III. It is a white crystalline solid.
IV. It is also called rock salt.
Select the correct statement(s).
(a) I, II and IV
(b) II, III and IV
(c) II and III
(d) III and IV
Answer:
(b) II, III and IV
Among the given statements related to NaCI, only the (I) statement is wrong, because sodium hydroxide (NaOH) is prepared by the chlor-alkali process, but not NaCl salt.
Question 6.
A jewelry-making unit tested a few elements based on their physical properties.
| Properties | A | B | C | D |
| Malleable | Yes | No | No | Yes |
| Ductile | Yes | No | No | Yes |
| Lustre | Yes | No | Yes | No |
| Melting Point | High | Low | Low | High |
Which of the above elements were discarded for usage by the company?
(a) A, B, and D
(b) B, C, and D
(c) A and C
(d) B and C
Answer:
(b) B, C, and D
The elements B, C, and D were discarded for usage by the company because for making jewellery, the element should be malleable, ductile, and lustrous, and should have a high melting point. So, these properties were only seen in element A. So, the rest of the others were discarded.
![]()
Question 7.
The bleaching powder gives the smell of chlorine due to
(a) formation of chlorine on exposure to the atmosphere
(b) instability of bleaching powder
(c) formation of excess of chlorine
(d) displacement reaction
Answer:
(a) formation of chlorine on exposure to the atmosphere
Bleaching powder (CaOCl2) reacts with carbon dioxide present in the atmosphere and produces calcium carbonate and chlorine gas, that’s why it gives the smell of chlorine.
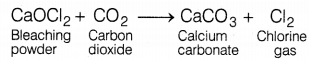
Question 8.
The diagram given below shows the inheritance of flower colour in pure-breeding roses.
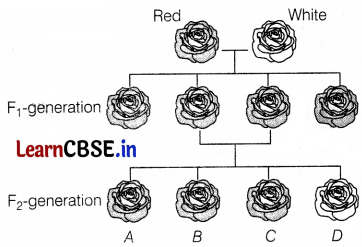
Which of the following flowers are heterozygous for colour in the F2 generation?
(a) A and C
(b) A and B
(c) C only
(d) B and C
Answer:
(d) B and C
When a pure-breed red colour flower crosses with a pure-breed white colour flower, then F1 progeny results in all heterozygous red flowers. When the F1 generation is allowed to cross only B and C will be heterozygotes.
Question 9.
Adrenaline hormone is secreted in the body during emergencies. What would be the effects of increased concentration of adrenaline on the body?
| Concentration of Glycogen in the Liver | Concentration of Glucose in the Blood | |
| (a) | Decrease | Increase |
| (b) | Increase | Increase |
| (c) | No Effect | Decrease |
| (d) | Increase | No Effect |
Answer:
(a) Concentration of Glycogen in the Liver – Decrease, Concentration of Glucose in the Blood – Increase
The concentration of glycogen in the liver will decrease. While the concentration of glucose in the blood will increase.
Question 10.
Name the type of movement that helps the food to move through the digestive tract.
(a) Amoeboid movement
(b) Peristaltic movement
(c) Ciliated movement
(d) Pseudopodial movement
Answer:
(b) Peristaltic movement
Peristaltic is a series of wave-like muscle contractions that move food through the digestive tract.
Question 11.
What is the role of microvilli present in the small intestine?
(a) They help to move the food along
(b) They make a large surface area for absorption
(c) They protect against bacteria
(d) They move mucus over the surface
Answer:
(b) They make a large surface area for absorption
Microvilli increase the surface area for the absorption of products of digestion, i.e. amino acid, glucose, etc,
![]()
Question 12.
Which of the following correctly defines autosomes?
(a) Chromosomes that do not play any role in sex determination
(b) Chromosomes which play a role in determining the sex of the newborn
(c) Both (a) and (b)
(d) Neither (a) nor (b)
Answer:
(a) Chromosomes that do not play any role in sex determination
Autosomes are the chromosomes that do not play a role in sex determination.
Question 13.
An electric fan runs from the 220V mains. The current flowing through it is 0.5A. How much energy is transformed in 2 min?
(a) 13200 J
(b) 110 J
(c) 1000 J
(d) 1500 J
Answer:
(a) 13200 J
Given, the potential difference, V = 220V
Current, I = 0.5 A
Time, t = 2 min = 120 s
Power, P =?
We know that,
Power (P) = VI
= 220 × 0.5
= 110 W
and E = Pt
= 110 × 120
= 13200 J
Question 14.
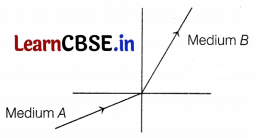
A light ray enters from medium A to medium B as shown in the figure. The refractive index of medium B relative to A will be
(a) greater than unity
(b) less than unity
(c) equal to unity
(d) zero
Answer:
(a) greater than unity
Since the light rays in medium 6 go towards normal, So, it has a greater refractive index and lesser velocity of light w.r.t. medium A. Hence, the refractive index of medium B w.r.t. medium A is greater than unity.
Question 15.
According to the law of dominance, which character expresses itself in the F1 generation?
(a) Dominant character
(b) Recessive character
(c) Hybrid character
(d) None of these
Answer:
(a) Dominant character
The dominant character expresses itself in the F1 generation.
![]()
Question 16.
What do you understand by phenotype?
(a) External appearance of an organism
(b) Genetic makeup of an organism
(c) Both (a) and (b)
(d) None of the above
Answer:
(a) External appearance of an organism
Directions (Q.Nos. 17-20) consist of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true, but R is not the correct explanation of A.
(c) A is true, but R is false.
(d) A is false, but R is true.
Question 17.
Assertion (A): Carbon reacts with oxygen to form carbon dioxide which is an acidic oxide.
Reason (R): Non-metals form acidic oxides.
Answer:
(a) Both (A) and (R) are true, but (R) is the correct explanation of (A).
Carbon is a non-metal form of acidic oxides, i.e. their aqueous solution turns blue litmus solution red.
Question 18.
Assertion (A): Urethra in the human male acts as a urinogenital canal.
Reason (R): The Urethra carries only urine, while sperms are carried by the vas deferens only.
Answer:
(c) A is true, but R is false.
R can be corrected as The urethra is the tube that allows urine to pass out of the body. In human males, it is a long tube that runs through the penis. It also carries semen in human males and is the common passage for both urine and sperm.
Question 19.
Assertion (A): Mendel self-crossed F1 progeny to obtain F2 generation.
Reason (R): The F1 progeny of a tall plant with round seeds and a dwarf plant with wrinkled seeds are all dwarf plants having wrinkled seeds.
Answer:
(c) A is true, but R is false.
The R can be corrected as The F1 progeny of a tall plant with round seeds and a dwarf plant with wrinkled seeds are all tall plants with round seeds. This is because tallness and round shape are both dominant traits, while dwarfness and wrinkled shape of seeds are recessive traits.
Question 20.
Assertion (A): The blue colour of the sky appears due to the scattering of the blue colour.
Reason (R): Blue colour has the shortest wavelength in the visible spectrum.
Answer:
(a) Both (A) and (R) are true, but (R) is the correct explanation of (A).
During the daytime, the sky appears blue. This is because the size of the particles in the atmosphere is smaller than the wavelengths of visible light. So, they scatter the light of shorter wavelengths. The scattered blue light enters our eye, according to Rayleigh’s scattering.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
An alkali metal A gives a compound B (molecular mass = 40) on reacting with water. The compound B gives a soluble compound C on treatment with aluminium oxide. Identify A, B, and C and give the reactions involved.
Answer:
Let, the atomic weight of alkali metal A be x.
When A reacts with water, it forms a compound B having molecular mass 40.
Let the reaction be
2A + 2H2O → 2AOH (B) + H2 ↑
According to the question,
x + 16 + 1 = 40 (Given)
∴ x = 40 – 17 = 23
23 is the atomic weight of Na (sodium). (1)
Therefore, the alkali metal (A) is Na and the reaction is
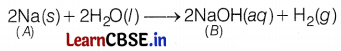
So, compound B is sodium hydroxide (NaOH).
Sodium hydroxide reacts with aluminium oxide (Al2O3) to give sodium aluminate (NaAlO2). Thus, C is sodium aluminate (NaAlO2). The reaction involved is
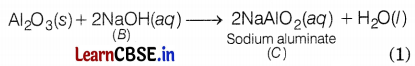
Question 22.
State any two practices, which can help in the protection of our environment.
Answer:
Two practices which can help in the protection of our environment are
(a) Disposable of the waste after its separation as biodegradable and non-biodegradable. (1)
(b) Judicious use of unleaded petrol and alternate sources of energy, e.g. solar energy, wind energy, hydro energy. (1)
![]()
Question 23.
Why do all gametes formed in human females have X chromosomes?
Or
Suppose, normal maize has starchy seeds which remain smooth when dry. A mutant form has sugary seeds which go wrinkled when dry. When the mutant was crossed with a normal plant, an F1 was produced which has smooth seeds. What would be the relative ratio of the different seed types, if the F1 was allowed to self-pollination?
Answer:
Human females have two ‘X’-chromosomes called sex-chromosomes. During meiosis, at the time of gamete formation, one X-chromosome enters each gamete. Hence, all the gametes possess an X chromosome. (2)
Or
Let the genotype of a normal plant with smooth seeds be SS and that of wrinkled seeds be ss. For a cross between SS and Ss, all the offspring produced will have genotypes (smooth seeds).
If the F1 with smooth seeds Ss is allowed to self-crossed then maize plants with smooth and maize plants with wrinkled seeds will be formed in the ratio of 3 : 1. (1)

As it is evident from Punnett square, 3 plants with smooth seeds (SS, Ss) and 1 (ss) with wrinkled seeds are formed. (1)
Question 24.
Six wires labelled as A, B, C, D, E, and F have been designed as per the following parameters.
| Wire | Length | Diameter | Material | Resistance |
| A | L | 2d | Aluminium | R1 |
| B | 2l | d/2 | Constantan | R2 |
| C | 3l | d/2 | Constantan | R3 |
| D | l/2 | 3d | Copper | R4 |
| E | 2l | 2d | Aluminium | R5 |
| F | l/2 | 4d | Copper | R6 |
Answer the following questions using the above data.
(a) Which of the wires has maximum resistance and why?
(b) Which of the wires has minimum resistance and why?
Answer:
(a) The wire C has maximum resistance because it has maximum length, least thickness, and highest resistivity. (1)
(b) The wire F has the minimum resistance since it has the least length, maximum thickness, and least resistivity. [using, R = ρ\(\frac{l}{A}\)] (1)
Question 25.
Draw a ray diagram to show what happens after reflection to the incident ray when it is parallel to the principal axis and falling on a convex mirror.
Or
In an electrical circuit, two resistors of 2 Ω and 4 Ω are connected in series to a 6V battery. Find the heat dissipated by the 4 Ω resistor in 5 s.
Answer:
Appear to come from focus of mirror after reflection.
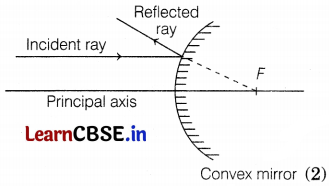
Or
Given, R1 = 2 Ω, R2 = 4 Ω, t = 5s, V = 6V
∴ Net resistance, R = R1 + R2 = 2 Ω + 4Ω = 6 Ω
∴ Current, I = \(\frac{\mathrm{V}}{R}=\frac{6 \mathrm{~V}}{6 \Omega}\) = 1 A
In series, the same 1 A current passes through both resistors.
∴ Heat dissipated, H = I2R1 × t
= (1)2 × 4 × 5
= 20 J
![]()
Question 26.
(a) State the role of ATP in cellular respiration.
(b) What ensures sufficient exchange of gases in plants?
Answer:
(a) ATP is considered as energy of a cell. It is used as a fuel in various cellular activities such as the contraction of muscles, contractions of nerve impulses, protein synthesis, etc.
(b) Stomata and large intercellular spaces present throughout the plant body ensure the sufficient exchange of gases in the plant. (2)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
An organic compound A is a constituent of many medicines and is used as an antifreeze and has the molecular formula C2H6O. Upon reaction with alk. KMnO4, the compound A is oxidized to another compound B with the formula C2H4O2. Identify the compounds A and B. Write the chemical equation for the reaction that leads to the formation of B.
Or
Name the compound formed when ethanol is heated more than concentrated sulphuric acid at 443K. Also, write the chemical equation of the reaction starting with the role of concentrated sulphuric acid in it. What would happen, if hydrogen is added to the product of this reaction in the presence of catalysts such as palladium or nickel?
Answer:
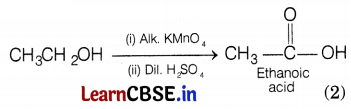
The organic compound A which is a constituent of many medicines and acts as antifreeze with the molecular formula C2H6O is ethanol (CH3CH2OH). Ethanol is oxidized to ethanoic acid (B) upon reaction with alk. KMnO4.

Or
When ethanol is heated more than concentrated sulphuric acid at 443K, ethene is formed. The reaction involved is
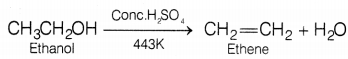
Conc. H2SO4 acts as a dehydrating agent, it removes water molecules from a compound. If hydrogen is added to the product, i.e. ethene then ethane is formed.
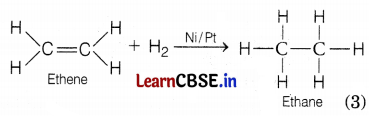
Question 28.
The diagram shown below is of the electrolytic refining of copper.
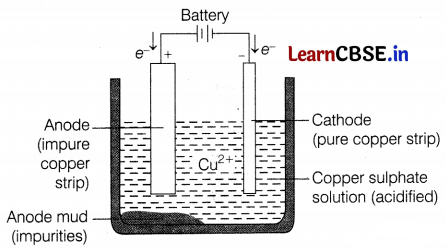
In this process, a thick block of impure metal is used as an anode, and a thin strip of pure metal is used as a cathode. A solution of metal is used as an electrolyte.
(a) What happens when an electric current is passed in the given system?
(b) Draw your conclusion from the given figure.
(c) Illustrate the reaction of the process with the help of a chemical equation.
Answer:
(a) When an electric current is passed, copper ions from the electrolyte are reduced as copper which gets deposited on the cathode. (1)
(b) An equivalent amount of pure copper from the anode gets oxidized to copper ion and goes into the solution and from there, it goes to the cathode and gets deposited. (1)
(c) The reactions that take place during the refining of copper are as follows: (1)
At cathode, Cu2+ (aq) + 2e– → Cu(s)
At anode, Cu(s) → Cu2+ (aq) + 2e–
This cycle is repeated until the whole of the copper ion from the impure block is dissolved and deposited on the cathode.
Question 29.
Give three characteristics of a food chain.
Answer:
The characteristics of the food chain are as follows:
- It is always straight and proceeds in a progressive straight line. (1)
- It helps in understanding the food relationship and interactions among various organisms in an ecosystem. (1)
- It also helps us to understand the movement of toxic substances in an ecosystem and the problem of their biological magnification. (1)
Question 30.
Ram wants to know about a device that works based on a current-carrying conductor placed in a magnetic field.
(a) What conclusion do you get from the observation that a current-carrying wire deflects a compass needle placed near it? Name the device that works on that principle.
(b) Write uses of galvanometer.
(c) When the force is experienced by a current-carrying conductor placed in a magnetic field largest?
Answer:
(a) The conclusion that can be drawn a magnetic field exists around a current-carrying conductor. Based on this principle, if a current-carrying conductor is placed in a uniform magnetic field, then a force is exerted on that conductor. Galvanometer works on this principle. (1)
(b) A galvanometer is used to detect very small currents or deflection of charges. Special purpose galvanometer can measure charges upto a few nano coulombs. It can also be used to measure current and potential differences by biasing with a resistor. (1)
(c) The force experienced by a current-carrying conductor placed in a magnetic field is the largest when the conductor is kept perpendicular to the direction of the magnetic field. (1)
![]()
Question 31.
(a) The following figure shows a ray of light travelling from medium A to medium B.
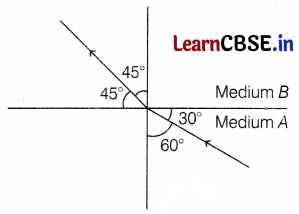
Determine the refractive index of medium B relative to medium A. From the given media, which one will be a denser medium?
(b) The speed of light in a transparent medium is 0.6 times that of its speed in a vacuum. What is the refractive index of the medium?
Answer:
(a) Given, the angle of incidence, i = 60°
The angle of refraction, r = 45°
The refractive index of the medium B relative to medium A,

Hence, the medium B is the denser medium. (2)
(b) As, refractive index (n)

Question 32.
A student wants to get the image of a candle flame on the walls of a school laboratory by using a lens.
(a) Which type of lens should he use and why?
(b) At what distance in terms of focal length F of the lens should he place the candle flame, to get a magnified and diminished image respectively, on the wall?
Answer:
(a) He should use a convex lens as real images are formed by it. (1)
(b) For a magnified image, he should place the candle flame between the focus (F) and centre of curvature (2F) of the lens. To get a diminished image, he should place the candle flame beyond centre of curvature (2F) of the lens. (2)
Question 33.
Name the hormone secreted by the thyroid gland and describe its main functions.
Answer:
The thyroid gland is a bilobed structure situated at the posterior surface of the larynx. Its structure resembles ‘H’. Its weight in humans is about 25-30 g.
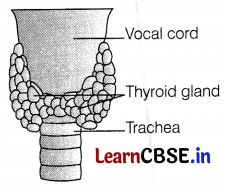
Functions of Thyroid Gland: (2)
- It mainly releases thyroxine or thyroid hormone which is a product of iodine. This hormone regulates metabolic activities in the body. (\(\frac{1}{2}\) + \(\frac{1}{2}\))
- It controls BMR rate, protein synthesis, increased heartbeat rate, and uses of glucose. (\(\frac{1}{2}\) + \(\frac{1}{2}\))
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
A compound X is formed by the reaction of a carboxylic acid C2H4O2 and an alcohol in the presence of a few drops of H2SO4. The alcohol on oxidation with alk.KMnO4 followed by acidification gives the same carboxylic acid as used in this reaction. Give the names and structures of carboxylic acid, alcohol, and the compound X. Also write the reaction.
Or
(a) A teacher of a school instructed the students to carry out the following reactions and also told them to write the reactions involved when ethanol is heated with alkaline potassium permanganate, ethanoic acid with sulphuric acid, and sodium respectively.
(b) What happens when barium chloride solution is mixed with copper sulphate solution?
Answer:
Carboxylic acid having molecular formula C2H4O2 is acetic acid or ethanoic acid. Its structure is (1)
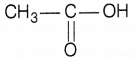
Since, an alcohol which on oxidation with alk. KMnO4 followed by acidification gives ethanoic acid. Therefore, it must be ethanol. Its structure is CH3-CH2-OH.
Since compound X is formed by the reaction of ethanoic acid with ethanol in the presence of a few drops of cone, H2SO4. Therefore, compound X must be an ester, i.e. ethyl ethanoate. Its structure is
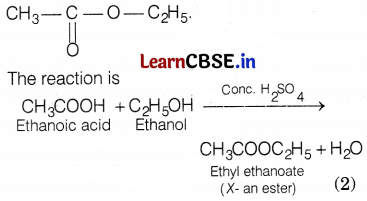
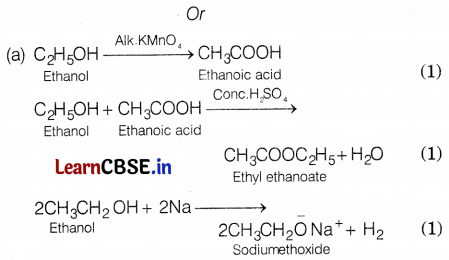
(b) When barium chloride solution is mixed with copper sulphate solution a white precipitate is obtained. The reaction involved is as follows:
BaCl2 (aq) + CuSO4 (aq) → BaSO4 (s) + CuCl2 (aq) (2)
![]()
Question 35.
(a) Mention secondary sexual characters in human males and females.
(b) Why are traits acquired during the lifetime of an individual not inherited?
Or
(a) Some glands produce chemical substances that change the activity of target organs. Name these substances. Also, gives the effect of the chemical produced by the adrenal gland pancreas.
(b) What is a reflex arc? How do muscle cells more?
Answer:
(a) Secondary sexual characters in males and females are as follows:
In Male,
- Hair growth on the face, chest, armpit (axillae), and over the pubis.
- The voice becomes deep and hoarse due to the growth of the larynx called Adam’s apple.
- The body becomes muscular.
In Female,
- Growth of breasts and hair in axillae and over pubis.
- The Pelvis region becomes broad and the buttocks become heavy due to the deposition of fat.
- Development of accessory sex organs. (3)
(b) Acquired characters are structural, functional, and behavioral changes that an individual develops during their lifetime due to a particular environment, disease, trauma, use, and disuse, conditioned and unconditioned learning, etc. These traits are not passed to the DNA of germ cells and are lost with the death of an individual. Therefore, acquired traits like structural changes, and experience, cannot be transmitted to the progeny, i.e. these are not inherited. (2)
Or
(a) These substances are called hormones. The adrenal gland secretes adrenaline and prepares the body for a fight-or-flight response. e.g.lincreased heart rate and breathing. Dilation of the eyes. The pancreas secretes hormones that control the conversion of excess glucose to glycogen in the liver. Thus, maintains the concentration of glucose in the blood. (2)
(b) The pathway taken by nerve impulses in a reflex action is called the reflex arc. They allow rapid response, e.g. pulling of hand on touching a hot object. Muscle cells have special proteins that change their shape and arrangement in the cells in response to an electrical impulse. This forces the muscle cells to contract and relax, causing their movement. (3)
Question 36.
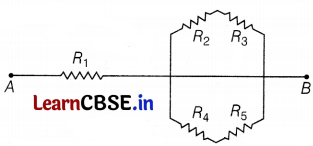
The diagram above shows an electric circuit in which resistances R1, R2, R3, R4, and R5 are connected. There is some air gap at the joint of wires.
(a) What is the resistance of an air gap?
(b) Derive an expression for equivalent resistance for the circuit.
(c) If the wire of resistance R1 and resistivity ρ is stretched to double its length, how will its resistance and resistivity be affected?
Or
Study the following circuit and answer the questions that follow:
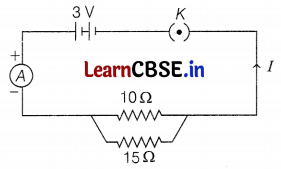
(a) State the type of combination of two resistors in the circuit.
(b) How much current is flowing through 10 Ω and 15 Ω resistors?
(c) What is the ammeter reading?
Answer:
(a) The resistance of an air gap is very large almost infinite. Since there is no contact between the conducting wires. (1)
(b) R2 and R3 are in series. Thus, for this combination, R’ = R2 + R3
Similarly, R4 and R5 are in series, so R” = R4 + R5.
R’ and R” are in parallel, then
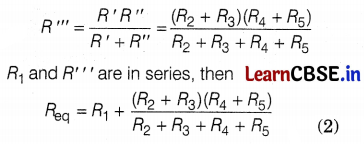
(c) Let the initial length of wire be l and the area of cross-section is A.
When the wire is stretched to double its length, the final length is l2 = 2l1
But the volume of the wire remains the same.

∴ Resistance is increased by 4 times.
Resistivity remains constant, as it is the property of the material and does not depend on the dimensions of the material. (2)
Or
(a) Two resistors are in parallel combination. (1)
(b) Current through
10 Ω resistor, I1 = \(\frac{V}{R}=\frac{3}{10}\) = 0.3A
15 Ω resistor, I2 = \(\frac{V}{R}=\frac{3}{15}\) = 0.2A (2)
(c) Ammeter reading = Total current flowing through the circuit = 0.3 + 0.2 = 0.5A (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
Two students take test tubes separately with different solutions, copper sulphate, and iron sulphate solution. They immersed an iron nail in the first test tube and copper wire in the latter as shown in the figure. Observe the two test tubes A and B and answer the following questions.
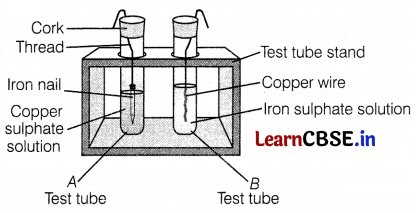
(a) Which type of reaction is involved in test tube A? Give its chemical equation.
(b) Draw your conclusion for test tube B.
Or
What will be the change in colour in test tube A?
Answer:
(a) In test tube A,
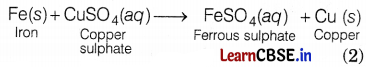
The reaction involved is a displacement reaction as copper is displaced by iron from its sulphate solution.
(b) As iron is more reactive than copper, copper will not displace iron from its sulphate solution; hence, there will be no reaction. (2)
Or
The copper sulphate solution is blue, but after the displacement reaction takes place, the ferrous sulphate solution forms which are light green. (2)
![]()
Question 38.
Mr. Sharma was suffering from health issues. He went for thorough health check-ups and was diagnosed to be HIV positive. Soon, this news spread in his neighbourhood, and on account of this, he faced social isolation.
Answer the following questions based on the information given above.
(a) Do you think people’s indifference towards HIV-positive people is justifiable? What kind of approach should we have towards the person suffering?
(b) How can one protect oneself from this disease?
(c) What values are not promoted by Mr. Sharma’s neighbours?
Or
Write down the modes of transmission of HIV.
Answer:
(a) not, we should exhibit human approach/behaviour and should not treat them as isolated. (1)
(b) One should be aware of the mode of transmission of disease, danger of sharing needles, transfusion of contaminated blood, etc. (1)
(c) Sympathy for fellow human beings ‘care’ and humane nature values are not promoted by Mr. Sharma’s neighbours. (2)
Or
Modes of transmission of HIV are discussed below:
(a) Unprotected sexual interaction with a previously infected person.
(b) Reusing needles used by infected individuals. (2)
Question 39.
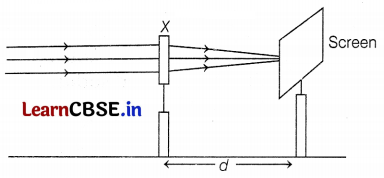
Prachi determined the focal length or device X by focusing a distant object on the screen as shown in the above diagram. The following table gives different positions of images and their sizes for different positions of objects in the case of image formation by lens.
| Position of Object | Position of Image | Relative Size of Image |
| At infinity | At focus F2 | Highly diminished and point-sized |
| Beyond 2F1 | Between F2 and 2F2 | Diminished |
| At 2F1 | At 2F2 | Same Size |
| Between F1 and 2F1 | Beyond 2F2 | Enlarged |
| At focus F1 | At infinity | Infinitely large or highly enlarged |
| Between focus F1 and optical centre O | On the same size of the lens as the object | Enlarged |
Based on the diagram and table, answer the following questions.
(a) In the diagram, what is the device X and the distance d?
(b) What will be the nature of the image, if an object is placed at infinity?
(c) If the magnification of the converging lens is +1, then write the nature of the image.
Or
Calculate the focal length of a lens for an object placed 50 cm from the lens producing a virtual image at a distance of 10 cm in front of the lens.
Answer:
(a) When the parallel rays coming from a distant object (at infinity), pass through a convex lens, after refraction through the lens, the rays converge at focus on the other side of the lens. Hence, device X is the convex lens and distance d is its focal length. (1)
(b) When the object is at infinity, the nature of the image will be real and inverted. (1)
(c) As the magnification is positive, so the image formed will be virtual and erect.
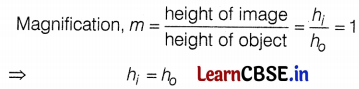
The image formed is of the same size as that of the object. (2)
Or
Given, object distance, u = -50 cm
Image distance, v = -10 cm
Focal length, f =?
