Students must start practicing the questions from CBSE Sample Papers for Class 10 Science with Solutions Set 10 are designed as per the revised syllabus.
CBSE Sample Papers for Class 10 Science Set 10 with Solutions
Time: 3 Hours
Maximum Marks: 80
Instructions
- This question paper consists of 39 questions in 5 sections.
- All questions are compulsory. However, an internal choice is provided in some questions. A student is expected to attempt only one of these questions.
- Section A consists of 20 objective-type questions carrying 1 mark each.
- Section B consists of 6 Very Short questions carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
- Section C consists of 7 Short Answer type questions carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
- Section D consists of 3 Long Answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
- Section E consists of 3 source-based/case-based units of assessment of 4 marks each with sub-parts.
Section A
Select and write the most appropriate option out of the four options given for each of the questions 1-20.
Question 1.
Consider the given flowchart.
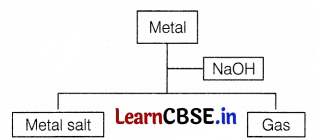
Which of the following two combinations are correct?
(a) (i) and (iii)
(b) (ii) and (iii)
(c) (ii) and (iv)
(d) (i) and (iv)
Answer:
(c) (ii) and (iv)
The correct combination is (ii) and (iv) as [1]
(i) When Al reacts with NaOH, then the salt of aluminium is formed with the evolution of hydrogen gas.
2Al (s) + 6NaOH (aq) → 2Na3AlO3 + 3H2 ↑
(ii) When silver and sodium hydroxide react together, then no reaction takes place. Hence no evolution of gas occurs.
(iii) Magnesium and sodium hydroxide do not react with each other. Hence, no evolution of gas.
(iv) When zinc is reacted with sodium hydroxide, then zinc salt is formed with the evolution of hydrogen gas.
Zn (s) + 2NaOH (aq) → Na2ZnO2 + H2 ↑
Question 2.
Metals are refined by using different methods. Which of the following metals are refined by electrolytic refining?
(a) Au
(b) Ca
(c) Na
(d) K
Answer:
(a) Au
Electrolytic refining is used for metals like Cu, Zn, Ag, Au, etc. The method to be used for refining an impure metal depends on the nature of the metal as well as on the nature of impurities present in it.
![]()
Question 3.
__________ can be used as an acid-base indicator by a visually impaired student.
(a) Litmus
(b) Vanilla essence
(c) Turmeric
(d) Petunia leaves
Answer:
(b) Vanilla essence
Vanilla essence is an olfactory indicator. So, its smell is different in acidic and basic media which can be detected easily by a visually impaired student. It has a characteristic pleasant smell. If a basic solution like sodium hydroxide solution is added to it, an acidic solution like hydrochloric acid, however, does not destroy the smell of Vanilla extract.
Question 4.
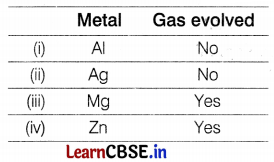
A student performed the following four experiments,
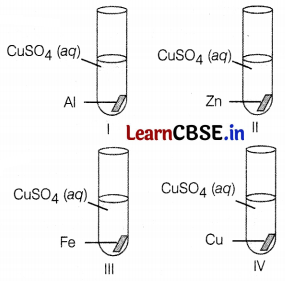
Which of the following observations is correct?
(a) The displacement reaction occurs in all experiments except III.
(b) The solid deposition is observed in the I, II, and III experiments.
(c) The solid deposition is observed in the I and II experiments.
(d) The displacement reaction is observed in the IV experiment.
Answer:
(b) The solid deposition is observed in the I, II, and III experiments.
In I, II, and III experiments, he found the formation of a solid deposition because Al, Zn, and Fe are more reactive than Cu. They will replace Cu from its sulphate solution and Cu will deposit as solid.
Question 5.
The reaction between potassium bromide and silver nitrate is an example of
(a) combination reaction
(b) decomposition reaction
(c) double displacement reaction
(d) displacement reaction
Answer:
(c) double displacement reaction
The reaction between potassium bromide and silver nitrate forms two products: silver bromide and potassium nitrate.
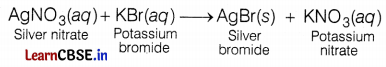
This reaction is an example of a double displacement reaction as Ag and K are displaced by each other.
Question 6.
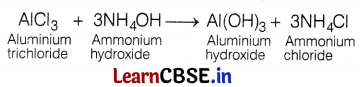
Which of the following represents the correct balanced chemical equation?
(a) AlCl3 + NH4OH → Al(OH)3 + NH4Cl
(b) AlCl3 + 3NH4OH → Al(OH)3 + 3NH4Cl
(c) 2AlCl3 + 3NH4OH → 2Al(OH)3 + 2NH4Cl
(d) 3AlCl3 + 4NH4OH → Al(OH)3 + NH4Cl
Answer:
(b) AlCl3 + 3NH4OH → Al(OH)3 + 3NH4Cl
Chemical equation (b) is a balanced one. The balanced chemical equation is one in which the total number of atoms of each element is equal on both sides of the equation.
Question 7.

In the above-given reaction, alkaline KMnO4 acts as
(a) reducing agent
(b) oxidizing agent
(c) catalyst
(d) dehydrating agent
Answer:
(b) oxidizing agent
KMnO4 acts as an oxidizing agent because it removes hydrogen from CH3CH2OH and adds one oxygen to it.
![]()
Question 8.
Consider the following figure that represents the food web.
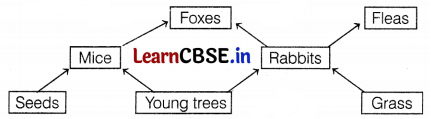
Which of the following options depicts the primary consumer in the food web?
(a) Rabbits and Mice
(b) Foxes and Grass
(c) Rabbits and Fleas
(d) Seeds and Foxes
Answer:
(a) Rabbits and Mice
The primary consumers are the organisms who directly feed on the producers. In the given food web, rabbits and mice are the primary consumers.
Question 9.
The graph shows, how the amount of carbon dioxide taken by a plant.
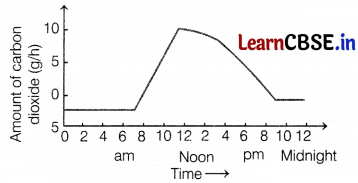
Why is the uptake of CO2 higher during day time?
(a) due to the large number of structures in the daytime
(b) due to photosynthesis
(c) due to transpiration
(d) due to more water absorption
Answer:
(b) due to photosynthesis
Plants take in CO2 from the atmosphere for preparing food. At night or early morning, photosynthesis does not occur hence, they take in oxygen and give out CO2.
Question 10.
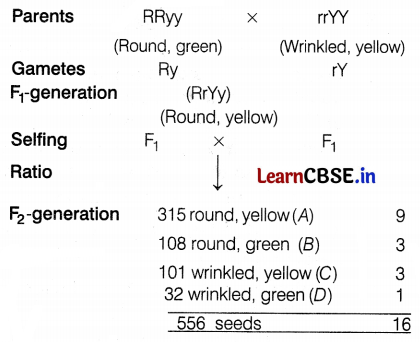
The result of a dihybrid cross between two individuals is recorded in the table as
| Phenotypes of progeny | Number of seeds obtained |
| Round, A | 315 |
| Round, B | 108 |
| Yellow, C | 101 |
| Wrinkled, D | 32 |
Choose the correct option for A, B, C, and D.
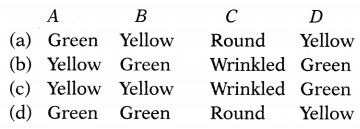
Answer:
(b) A – Yellow, B – Green, C – Wrinkled, D – Green
The dihybrid cross between the round, green plant, and the wrinkled, yellow plant is shown as
Question 11.
Which part of the human brain controls body temperature?
(a) Pituitary
(b) Diencephalon
(c) Hypothalamus
(d) None of the above
Answer:
(c) Hypothalamus
The hypothalamus controls and regulates the temperature of the body, the urge to eat, drink, sleep, etc.
![]()
Question 12.
Which of the following is not a functional component of an ecosystem?
(a) Ecological pyramids
(b) Decomposers
(c) Sunlight
(d) Energy flow
Answer:
(a) Ecological pyramids
Ecological pyramids are not a functional component of an ecosystem.
Question 13.
Four students set up the circuit for the experiment of Ohm’s law as shown. Which of the following options is correct?
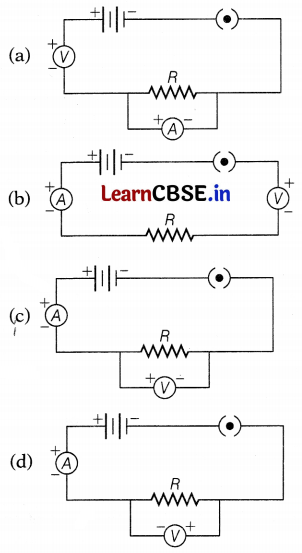
Answer:
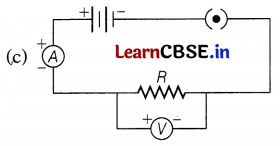
In the circuit, the ammeter should always be connected in series and the voltmeter should always be connected in parallel combination. Hence, the correct circuit set-up is shown below.
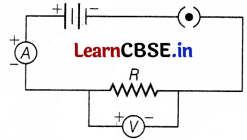
Question 14.
Which of the following can make a parallel beam of light when light from a point source is incident on it?
(a) Concave mirror as well as convex lens
(b) Convex mirror as well as concave lens
(c) Two plane mirrors placed at 90° to each other
(d) Concave mirror as well as concave lens
Answer:
(a) Concave mirror as well as convex lens
Both convex lenses and concave mirrors can make a parallel beam of light when light from a point source is incident on it.
Question 15.
A food web is the
(a) food that a spider collects using its web
(b) network of interlinked trophic levels
(c) network of interlinked food chains
(d) display of food items on a website
Answer:
(c) network of interlinked food chains
A food web is a network of inter-linked food chains operating at various trophic levels.
![]()
Question 16.
The law of dominance states that
(a) a dominant trait always suppresses the recessive trait.
(b) the recessive trait is always expressed.
(c) the dominant trait is always suppressed.
(d) the recessive trait dominates over the dominant trait.
Answer:
(a) a dominant trait always suppresses the recessive trait.
Directions (Q. Nos. 17-20) Consist of two statements – Assertion (A) and Reason (R). Answer these questions by selecting the appropriate option given below.
(a) Both A and R are true and R is the correct explanation of A.
(b) Both A and R are true but R is not the correct explanation of A.
(c) A is true but R is false.
(d) A is false but R is true.
Question 17.
Assertion (A): Diamond does not conduct electricity.
Reason (R): Diamond has a high refractive index.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
The correct reason is, that due to the absence of free electrons, diamond does not conduct electricity.
Question 18.
Assertion (A): Positive phototropism means movement towards light.
Reason (R): When sunlight falls on one side of the plant, the auxin diffuses towards the sunny side of the shoot. Auxin concentration stimulates cells to grow longer and the stem appears bending towards sunlight.
Answer:
(c) A is true, but R is false.
It can be corrected as When sunlight falls on one side of the plant, the auxin diffuses towards the shady side of the shoot.
Question 19.
Assertion (A): DNA copying is necessary during reproduction.
Reason (R): DNA copying leads to the transmission of characters from parents to offspring.
Answer:
(a) Both A and R are true and R is the correct explanation of A.
DNA copying is necessary during reproduction because it leads to the transmission of characters from parents to offspring and brings about variation.
Question 20.
Assertion (A): The stars twinkle while the planet does not.
Reason (R): The stars are much bigger than the planets.
Answer:
(b) Both A and R are true but R is not the correct explanation of A.
As planets are much closer to the earth than stars, planets can be considered as a collection of a large number of point-sized sources of light. The total variation in the amount of light entering our eye from all there individual point-sized sources will average out to zero which nullify the twinkling effect of each other. Therefore, planets do not twinkle.
Section B
Questions No. 21 to 26 are Very Short Answer Questions.
Question 21.
What is a covalent bond? What type of bond exists in CCl4 and CaCl2?
Answer:
The chemical bonds formed between two atoms by the sharing of electrons between them is known as a covalent bond. (1)
- CCl4 – Covalent bond
- CaCl2 – Ionic bond (1)
Question 22.
In a pea plant, find the contrasting trait if the flower is white and the shape of the pod is constricted.
Answer:
Contrasting traits of pea plants were used by Mendel and were classified as dominant or recessive.
| Characters | Given traits | Contrasting traits |
| (a) Colour of flower | White | Violet |
| (b) Shape of the pod | Constricted | Full |
![]()
Question 23.
Goitre is caused by which endocrine gland? Write one primary prevention.
Or
Explain the functions of a neuron.
Answer:
Goitre is caused by the deficiency of thyroxine hormone secreted by the thyroid gland. Uptaking an optimum amount of iodine helps in preventing this disease. In hilly areas, iodine is deficient in water which causes goitre disease in most of the population in that area. Iodized salt uptake can help in the prevention of this disease. (2)
Or
Functions of a neuron The neuron receives information from receptors as electrical impulses, at its dendritic end. The impulse then travels from the dendrite to the cell body and further along the axon to its end. At the end of the axon, an electrical impulse leads to the release of some chemicals. These chemicals cross the synapse and reach the next neuron. This is how nerve impulses travel through the body. Thus, neurons are important in receiving information from the surroundings and in sending it to the effector. (2)
Question 24.
The given data shows the focal lengths of three concave mirrors A, B, and C, and the respective distances of different objects from these mirrors.
| Concave mirrors | Object distances (in cm) | Focal length (in cm) |
| A | 45 | 20 |
| B | 30 | 15 |
| C | 20 | 30 |
In the given positions of objects from the mirrors, which of them will form diminished images of the object?
Answer:
Mirror A will form a diminished image of the object. Because the object is very far from the mirror. [More than twice it’s focal length 2f = 2 × 20 = 40)] (2)
Question 25.
What are magnetic field lines? Justify the following statements.
(a) Two magnetic field lines never intersect each other.
(b) Magnetic field lines are closed curves.
Or
You have two electric lamps having ratings 40 W, 220 V and 60 W, 220 V. Which of the two has a higher resistance? Give a reason for your answer. If these two lamps are connected to a source of 220 V, which will glow brighter?
Answer:
The imaginary lines representing the magnetic field around a magnet are known as magnetic field lines.
(a) If two field lines intersect each other, this would mean that at the point of intersection, the direction of the magnetic field is in two directions, which is not possible. (1)
(b) The direction of field lines outside a magnet is from the North pole to the South pole while it is from the South to the North pole inside the magnet and thus forms closed curves. (1)
Or
We know that, power, P = \(\frac{V^2}{R}\)
Thus, resistance is inversely proportional to power, i.e., higher power less will be resistance and vice-versa. So, the electric lamp with a power rating of 40 W will have a higher resistance as compared to a 60 W lamp. Lamps with higher power will glow brighter. Hence, a lamp with 60 W power will glow brighter.
Question 26.
(a) Give a reason, why the ozone layer in the stratosphere is considered useful. What destructive effect do chlorofluorocarbons bring about in the atmosphere?
(b) Explain why, it is better to use paper bags than plastic bags.
Answer:
(a) The ozone layer is very important for the existence of life on Earth because it prevents harmful ultraviolet (UV) radiations coming from the sun from reaching the Earth. CFCs deplete ozone, resulting in increasing the passage of harmful UV radiation to the earth. (1)
(b) It is better to use paper bags than plastic bags because
- Paper is recyclable whereas plastic bags can not be recycled.
- Plastic cannot be degraded and products made up of plastic pollute the environment, whereas paper is biodegradable and does not cause by pollution.
- Moreover, even on burning, the paper will not cause much pollution but plastic on burning will release toxic chemicals into the environment thus, polluting the environment. (Any two) (1)
Section C
Questions No. 27 to 33 are Short Answer Questions.
Question 27.
Explain the reactions of different metals with hot water, cold water, and steam. Give one example with a properly balanced chemical equation. Name of the two metals which do not react with any form of water.
Or
How is the method of extraction of metals high up in the reactivity series different from that for metals in the middle? Why can be same process not be applied for them? Name the process used for the extraction of these metals.
Answer:
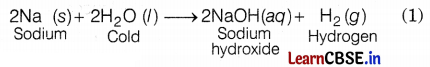
Reaction of metal with water
(a) With cold water
(b) With hot water
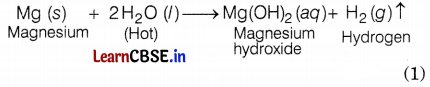
(c) With steam
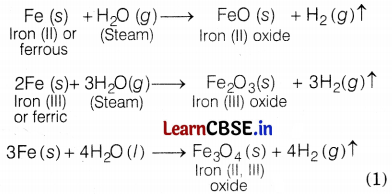
Gold and silver do not react with any form of water.
Or
The metals in the middle of the reactivity series (such as iron, zinc, lead, copper, etc.) are moderately reactive. Thus, to obtain such metals from their compounds, their sulphides and carbonates are first converted into their oxides by the process of roasting and calcination respectively and then the metal oxides are reduced to corresponding metal by using suitable reducing agents such as carbon. (2)
On the other hand, metals which are high up in the reactivity series (such as sodium, magnesium, calcium, aluminium, etc.) are very reactive and cannot be obtained from their compound by heating with carbon. Therefore, such metals are obtained by electrolytic reduction of their molten salt. (1)
![]()
Question 28.
The pH of a salt used to make tasty and crispy pakoras is 14.
(a) Examine the salt used.
(b) Write a chemical equation for its formation.
(c) List its two uses.
Answer:
The pH of a salt used to make tasty and crispy pakoras is 14.
(a) The salt is sodium hydrogen carbonate. Its formula is NaHCO3. This is also known as baking soda. (1)
(b) The chemical reaction involved as
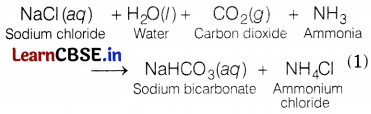
(c) Uses:
(i) For making baking powder.
(ii) Also used as an ingredient in antacids. (1)
Question 29.
Explain how voluntary actions and reflex actions are different from each other.
Answer:
The difference between involuntary actions and reflex actions are as follows:
| Involuntary Actions | Reflex Actions |
| They involve the autonomic nervous system. | They involve all parts of the voluntary nervous system though they are not under our control. |
| They involve the functioning of the internal body parts. | They are concerned with emergencies. |
| The nervous system controlling involuntary actions has two divisions, sympathetic and parasympathetic. | There are no such divisions. |
| They occur in response to internal stimuli. | They commonly operate against harmful stimuli, which are generally external. |
| Most of the involuntary functions occur all the time. | Reflex actions occur occasionally. |
| Sometimes gap occurs between stimulus and response. | They are almost instant. |
| They are carried out by smooth muscles. | They are performed by striated muscles. |
| The beating of the heart and peristalsis. | Closing of eyes when light is flashed on them. |
Question 30.
Shyam wants to have twice in size of a real image of an object placed at 25 cm from a convex lens.
(a) Specify the distance of the image from the lens.
(b) Find the focal length of the lens.
(c) Find the power of the convex lens.
Answer:

Question 31.
Two resistors with resistances 5 Ω and 10 Ω respectively, are to be connected to a battery of emf 6 V. How will you connect the resistances to obtain
(a) maximum current?
(b) Calculate the strength of the total current in the circuit in the two cases.
Answer:
(a) For obtaining maximum current, the two resistors should be connected in parallel. (1)
(b) ∴ Total current in the circuit, (parallel combination)
I = \(\frac{V}{R}=\frac{6 \times 3}{10}\) = 1.8 A
∴ Total current in the circuit, (series combination)
I = \(\frac{V}{R}=\frac{6}{15}\) = 0.4 A (2)
![]()
Question 32.
Rishi went to a palmist to show his palm. The palmist used a special lens for this purpose.
(a) Where should the palmist place/hold the lens to have a real and magnified image of an object?
(b) If the focal length of this lens is 10 cm and the lens is held at a distance of 5 cm from the palm, use the lens formula to find the position and size of the image.
Answer:
(a) A palmist uses a convex lens to see the magnified image of palm lines. The palmist will hold the lens where the palm is in between the focus and pole of the lens. (1)
(b) Given, focal length f = 10 cm and object distance, u = -5 cm
From lens formula, \(\frac{1}{v}-\frac{1}{u}=\frac{1}{f}\)
⇒ \(\frac{1}{v}=\frac{1}{10}+\frac{1}{-5}=\frac{1}{10}-\frac{1}{5}\)
⇒ v = -10 cm
Hence, the magnification,
m = \(\frac{v}{u}=\frac{-10}{-5}\) = 2
Hence, the image is on the same side of the lens as an object (palm) and it is virtually erect and magnified. (2)
Question 33.
Give the basic features of the mechanism of inheritance.
Answer:
The basic features of inheritance are as follows:
- Characters are controlled by genes.
- Each gene controls one character.
- There may be two or more forms of gene.
- One form may be dominant over the other.
- Genes are present on chromosomes.
- An individual has two forms of gene whether similar or dissimilar. (\(\frac{1}{2}\) × 6)
Section D
Questions No. 34 to 36 are Long Answer Questions.
Question 34.
Neha observed the following cases and some questions arose in her mind listed below. Help her to find them out. Why does calcium start floating when added to water? Also, why most of the metals do not give hydrogen while reacting with nitric acid? Write an equation for the reaction of iron with steam. Name the compound of iron obtained.
Or
(a) What is the difference between combustion and oxidation? Under what condition an oxidation reaction can be called combustion? Illustrate your answer with one example in each case.
(b) Yashi took a thin zinc plate and kept it in a glass container having CuSO4 solution. On examination, it was found that the blue colour of the solution was getting lighter and lighter. After a few days when the zinc plate was taken out of the solution, several small holes were noticed in it. State the reason and write the chemical equation of the reaction.
Answer:
The reaction of calcium with water is less violent. The heat evolved is not sufficient for hydrogen to catch fire.
Ca (s) + 2H2O (l) → Ca(OH)2 (aq) + H2 (g) (2)
Calcium starts floating because the bubbles of hydrogen gas formed stick to the surface of the metal. Most of the metals do not give hydrogen while reacting with nitric acid because HNO3 is a strong oxidizing agent. It oxidizes the H2 produced to water and itself gets reduced to any of the nitrogen oxides (N2O, NO, NO2). (2)
Iron reacts with steam to form iron oxide and hydrogen.
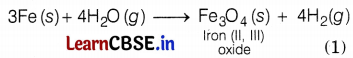
Or
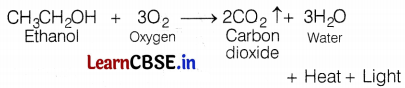
(a) The addition of oxygen or removal of hydrogen from any substance is called oxidation.
e.g. When ethanol is burnt in the air, it produces CO2 and water.
However, when ethanol is heated with alk. KMnO4 or acidified K2Cr2O7, gives ethanoic acid.
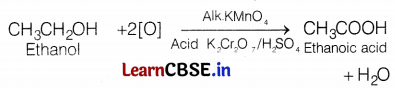
Both these reactions are oxidation reactions. In both cases, oxygen has been added or hydrogen has been removed. However, the complete oxidation of an organic compound to form CO2 and H2O is called combustion. Therefore, the oxidation of ethanol to form CO2 and H2O can also be called a combustion reaction. Thus, complete oxidation of an organic compound to form CO2 and H2O is also called combustion while partial oxidation of a compound to give a compound other than CO2 and H2O is called oxidation.
(b) A strip of zinc metal is put in a copper sulphate solution. The blue colour of copper sulphate solution fades gradually due to the formation of colorless, zinc sulphate solution. Since zinc is more reactive than copper, it displaces copper from copper sulphate solution. Zinc metal is consumed and hence several small holes were formed in the zinc strip.
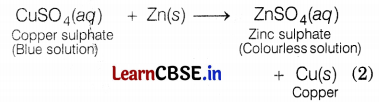
Question 35.
(a) Marriage between cousins is a taboo in most of the countries of the world except in some Asian countries. Why should marriage between close cousins be prevented and which measures will you take to prevent such practices?
(b) Describe the process of fertilization in a flower.
Or
(a) Explain the events of double circulation.
(b) Differentiate between fermentation in yeast and aerobic respiration based on the end products formed.
Answer:
(a) Marriage between close cousins should be prevented as the recessive traits with the genetic defects present in such families will become homozygous and cause harmful diseases Children should be educated about the ill effects so that they come to know about the defects in marriages among close cousins. These are the measures that can be taken to prevent such practices. (2)
(b) It is the process of fusion of male germ cells with the female gametes. It gives rise to a zygote. As soon as the pollen lands on a suitable stigma, it reaches the female germ cells in the ovary. This occurs via pollen tube. The pollen tube grows out of the pollen grain, travels through the style, and finally reaches the ovary. The fertilization In the flowering plant is shown in the given figure.
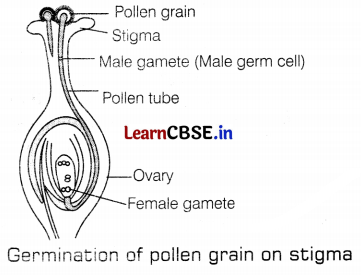
After fertilization, the zygote divides many times and forms an embryo within the ovule. This ovule then develops a tough coat and gets converted into a seed. The ovary rapidly grows and ripens as fruit. The seed contains the future embryo that develops into a seedling under suitable conditions. This process is known as germination. (3)
Or
(a) During double circulation in human beings, the blood passes twice through the heart during one complete cycle of circulation. The double circulation includes the following processes. (1)
- Pulmonary circulation: In this, the deoxygenated blood is pushed by the right ventricle to the lungs for oxygenation. The oxygenated blood then comes back to the left atrium of the heart through the pulmonary veins. (1)
- Systemic circulation: In this circulation, oxygenated blood from the left ventricle of the heart is passed to the different parts of the body through the aorta. Such a separation allows a highly efficient supply of oxygen to the body. This is useful in animals that have high energy needs such as birds and mammals including human beings, which constantly use energy to maintain their body temperature.
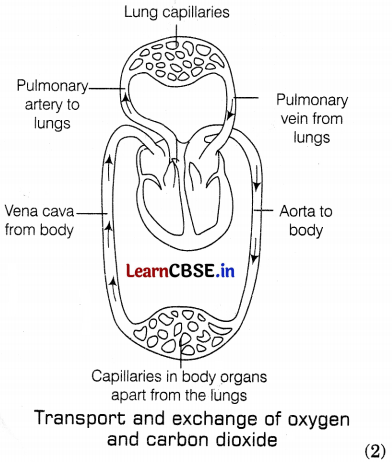
(b) During fermentation in yeast, ethanol is formed, while in aerobic respiration CO2 and water are formed. The energy released in the process of fermentation is also much less as compared to energy released in aerobic respiration. (1)
![]()
Question 36.
The diagram shown below is an electric circuit in which a 7.2 Ω resistor is connected in series with the parallel combination of 8 Ω and 12 Ω resistors. Power is supplied to the circuit with a 6V battery.
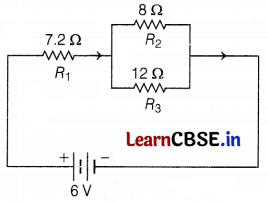
In the circuit diagram given above, find the
(a) the total resistance of the circuit,
(b) the total current flowing in the circuit and
(c) the potential difference across R1.
Or
(a) Heating elements of electrical heating devices are made up of an alloy rather than a pure metal. Give two reasons.
(b) Four resistors of 4 Ω each are joined end to end to form a square. Calculate the equivalent resistance of the combination between two adjacent corners.
(c) Define resistance and 1 ohm.
Answer:
(a) Resistances R2 and R3 are connected in parallel.

Now, resistances 7.2 Ω and 4.8 Ω are connected in series.
So, total resistance = 7.2 + 4.8 = 12.0 Ω (2)
(b) From Ohm’s law,
R = \(\frac{V}{I}\)
I = \(\frac{V}{R}\) = \(\frac{6}{12}\)
Current, I = 0.5A (1)
(c) Current through R1 = 0.5 A
Resistance of R1 = 7.2 Ω
By Ohm’s Law,
R = \(\frac{V}{I}\)
⇒ \(\frac{V}{0.5}\) = 7.2
⇒ V = 7.2 × 0.5 = 3.6 V
The potential difference across the ends of the resistance R1 is 3.6V. (2)
Or
(a) Alloys are used for making electrical heating devices due to the following reasons:
(i) They have a higher resistivity as compared to pure metals.
(ii) They do not oxidize readily at high temperatures. (1)
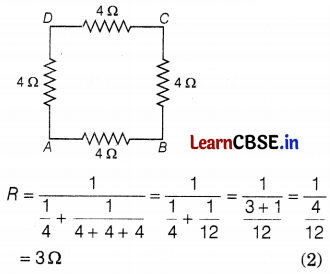
(b) The resistance between points A and B is given by
(c) The property of a conductor due to which it opposes the flow of current through it is called resistance. The unit of resistance is ohm. One ohm is the resistance of a conductor such that when a potential difference of 1V is applied to its ends, a current of 1A flows through it. (2)
Section E
Questions No. 37 to 39 are case-based/data-based questions with 2 to 3 short sub-parts. Internal choice is provided in one of these sub-parts.
Question 37.
Abhishika observed that the cooking utensils of her kitchen are becoming black and the flame of her burner becomes yellowish. She complained about this to the gas company and they repaired it.
(a) What was the reason for this type of flame?
(b) How can this problem affect our environment?
Or
What precautions should be taken to avoid this process?
Answer:
(a) The inlets present in the burner get blocked due to which proper combustion does not take place. This results in the yellow flame. (2)
(b) This process is known as incomplete combustion and is very harmful to our environment. This leads to the formation of oxides which are major pollutants. (2)
Or
To prevent this situation, burners of gas or stores should be cleaned timely so that inlets do not block. (2)
Question 38.
Married women ‘Sita’ used device X made of common metal for preventing pregnancy. This device was put into her uterus by some trained medical professional. Unfortunately, she got pregnant after two months of insertion of the device. She was in shock to learn that her birth control device had failed. Answer the following questions based on the above passage.
(a) Name the birth control device used by the ‘Sita’.
(b) Which metal is commonly used for making device X?
(c) What do you think Sita got pregnant even after using device X?
Or
How does device ‘X’ prevent pregnancy?
Answer:
(a) Copper-T, an intra-uterine device is placed inside the uterus by a doctor or a trained nurse. (1)
(b) Copper metal is commonly used for making device X e.g. copper-T. (1)
(c) Intra-uterine devices are highly effective in preventing unwanted pregnancies. But they came with one disadvantage, they can get expelled anytime without the knowledge of women. (2)
Or
(d) The device ‘X’ namely copper-T releases copper ions that prevent pregnancy by inhibiting the movement of sperm. It is because the copper ion-containing fluid is toxic to sperm. Thus, fertilization is also prevented. (2)
![]()
Question 39.
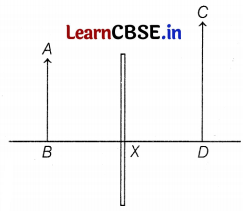
The above diagram shows that two students P and Q experimented on finding the image formation by a mirror. They experimented with different positions of objects and obtained different positions of images. AB, CD, and X represent objects, images, and the optical device respectively.
(a) Based on the text and data given in the above paragraph, identify the type of optical device X.
(b) What will be the position of the image? If the object is placed between the focus and center of curvature.
(c) If the object is placed at a distance of 10 cm in front of the mirror X, then what will be the distance of the image formed? [Take, magnification = -3]
Or
What will happen, if the upper half of the concave mirror ‘X’ is covered?
Answer:
(a) In this case, the image formed is virtual, erect, and magnified. The image is formed behind the optical device X. Hence, device X is a concave mirror. When the object is placed between the pole and the focus of a concave mirror, the image formed will be virtual, erect, magnified, and behind the mirror. (1)
(b) Concave mirror X will form the magnified image of the object as the object is placed between the focus and centre of curvature of the mirror. (1)
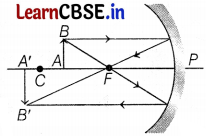
(c) Given, object distance, u = -10 cm
Magnification, m = -3
Now, m = \(-\frac{v}{u}\)
⇒ -3 = \(-\frac{v}{(-10)}\)
⇒ v = -30 cm
The negative sign shows that the image is real and in front of the mirror. (2)
Or
If the upper half of the concave mirror is covered, the focal length of the mirror and object distance do not change, then the brightness of the image will be reduced. (1)
This is because the whole image will be formed by the rays passing through the upper half portion but the intensity of the image formed is proportional to the number of rays and hence the image will be less brighter. (1)